

Real Life Bipolar Disorder: A Case Study of Susan
Bipolar disorder is a complex and often misunderstood mental health condition that affects millions of individuals worldwide. For those living with bipolar disorder, the highs and lows of life can be dizzying, as they navigate through periods of intense mania and debilitating depression. To truly grasp the impact of this disorder, it’s crucial to explore real-life experiences and the stories of those who have dealt firsthand with its challenges.
In this article, we delve into the fascinating case study of Susan, a woman whose life has been profoundly shaped by her bipolar disorder diagnosis. By examining Susan’s journey, we aim to shed light on the realities of living with this condition and the strategies employed to manage and treat it effectively.
But before we plunge deeper into Susan’s story, let’s first gain a comprehensive understanding of bipolar disorder itself. We’ll explore the formal definition, the prevalence of the condition, and its impact on both individuals and society as a whole. This groundwork will set the stage for a more insightful exploration of Susan’s experience and provide valuable context for the subsequent sections of this article.
Bipolar disorder is more than just mood swings; it is a condition that can significantly disrupt an individual’s life, relationships, and overall well-being. By studying a real-life case like Susan’s, we can gain a personal insight into the multifaceted challenges faced by those with bipolar disorder and the importance of effective treatment and support systems. In doing so, we hope to foster empathy, inspire early diagnosis, and contribute to the advancement of knowledge about bipolar disorder’s complexities.
The Case of Susan: A Real Life Experience with Bipolar Disorder
Susan’s story provides a compelling illustration of the impact that bipolar disorder can have on an individual’s life. Understanding her background, symptoms, and the effects of the disorder on her daily life can provide valuable insights into the challenges faced by those with bipolar disorder.
Background Information on Susan
Susan, a thirty-eight-year-old woman, was diagnosed with bipolar disorder at the age of twenty-five. Her early experiences with the disorder were characterized by periods of extreme highs and lows, often resulting in strained relationships and an inability to maintain steady employment. Susan’s episodes of mania frequently led to impulsive decision-making, excessive spending sprees, and risky behaviors. On the other hand, her depressive episodes left her feeling hopeless, fatigued, and unmotivated.
Symptoms and Diagnosis of Bipolar Disorder in Susan
To receive an accurate diagnosis, Susan underwent a thorough examination by mental health professionals. The criteria for diagnosing bipolar disorder include significant and persistent mood swings, alternating between periods of mania and depression. Susan exhibited classic symptoms of bipolar disorder, such as elevated mood, increased energy, racing thoughts, decreased need for sleep, and reckless behavior during her manic episodes. These episodes were interspersed with periods of deep sadness, loss of interest in activities, and changes in appetite and sleep patterns during depressive phases.
Effects of Bipolar Disorder on Susan’s Daily Life
Living with bipolar disorder presents unique challenges for Susan. The unpredictable shifts in her mood and energy levels significantly impact her ability to function in both personal and professional spheres. During manic phases, Susan experiences heightened productivity, creativity, and confidence, often leading her to take on excessive responsibilities and projects. However, these periods are eventually followed by crashes into depressive episodes, leaving her unable to complete tasks, maintain relationships, or even perform routine self-care. The constant fluctuations in her emotional state make it difficult for Susan to establish a sense of stability and predictability in her life.
Susan’s struggle with bipolar disorder is not uncommon. Many individuals with this condition face similar obstacles in their daily lives, attempting to manage the debilitating highs and lows while striving for a sense of normalcy. By understanding the real-life implications of bipolar disorder, we can more effectively tailor our support systems and treatment options to address the needs of individuals like Susan. In the next section, we will explore the various approaches to treating and managing bipolar disorder, providing potential strategies for improving the quality of life for those living with this condition.
Treatment and Management of Bipolar Disorder in Susan
Managing bipolar disorder requires a multifaceted approach that combines psychopharmacological interventions, psychotherapy, counseling, and lifestyle modifications. Susan’s journey towards finding effective treatment and management strategies highlights the importance of a comprehensive and tailored approach.
Psychopharmacological Interventions
Pharmacological interventions play a crucial role in stabilizing mood and managing symptoms associated with bipolar disorder. Susan’s treatment plan involved medications such as mood stabilizers, antipsychotics, and antidepressants. These medications aim to regulate the neurotransmitters in the brain associated with mood regulation. Susan and her healthcare provider closely monitored her medication regimen and made adjustments as needed to achieve symptom control.
Psychotherapy and Counseling
Psychotherapy and counseling provide individuals with bipolar disorder a safe space to explore their thoughts, emotions, and behaviors. Susan engaged in cognitive-behavioral therapy (CBT), which helped her identify and challenge negative thought patterns and develop healthy coping mechanisms. Additionally, psychoeducation in the form of group therapy or support groups allowed Susan to connect with others facing similar challenges, fostering a sense of community and reducing feelings of isolation.
Lifestyle Modifications and Self-Care Strategies
In addition to medical interventions and therapy, lifestyle modifications and self-care strategies play a vital role in managing bipolar disorder. Susan found that maintaining a stable routine, including regular sleep patterns, exercise, and a balanced diet, helped regulate her mood. Avoiding excessive stressors and implementing stress management techniques, such as mindfulness meditation or relaxation exercises, also supported her overall well-being. Engaging in activities she enjoyed, nurturing her social connections, and setting realistic goals further enhanced her quality of life.
Striving for stability and managing bipolar disorder is an ongoing process. What works for one individual may not be effective for another. It is crucial for individuals with bipolar disorder to work closely with their healthcare providers and engage in open communication about treatment options and progress. Fine-tuning the combination of psychopharmacological interventions, therapy, and self-care strategies is essential to optimize symptom control and maintain stability.
Understanding the complexity of treatment and management helps foster empathy for individuals like Susan, who face the daily challenges associated with bipolar disorder. It underscores the importance of early diagnosis, accessible mental health care, and ongoing support systems to enhance the lives of individuals living with this condition. In the following section, we will explore the various support systems available to individuals with bipolar disorder, including family support, peer support groups, and the professional resources that contribute to their well-being.
Support Systems for Individuals with Bipolar Disorder
Navigating the challenges of bipolar disorder requires a strong support system that encompasses various sources of assistance. From family support to peer support groups and professional resources, these networks play a significant role in helping individuals manage their condition effectively.
Family Support
Family support is vital for individuals with bipolar disorder. Understanding and empathetic family members can provide emotional support, monitor medication adherence, and help identify potential triggers or warning signs of relapse. In Susan’s case, her family played a crucial role in her recovery journey, providing a stable and nurturing environment. Education about bipolar disorder within the family helps foster empathy, reduces stigma, and promotes open communication.
Peer Support Groups
Peer support groups provide individuals with bipolar disorder an opportunity to connect with others who share similar experiences. Sharing personal stories, strategies for coping, and offering mutual support can be empowering and validating. In these groups, individuals like Susan can find solace in knowing that they are not alone in their struggles. Peer support groups may meet in-person or virtually, allowing for easier access to support regardless of physical proximity.
Professional Support and Resources
Professional support is crucial in the management of bipolar disorder. Mental health professionals, such as psychiatrists, psychologists, and therapists, provide expertise and guidance in developing comprehensive treatment plans. Regular therapy sessions allow individuals like Susan to explore emotional challenges and develop healthy coping mechanisms. Psychiatrists closely monitor medication effectiveness and make necessary adjustments. Additionally, case managers or social workers can assist with navigating the healthcare system, accessing resources, and connect individuals with other community services.
Beyond direct professional support, there are resources and organizations dedicated to bipolar disorder education, advocacy, and support. Online forums, websites, and helplines provide information, guidance, and a sense of community. These platforms allow individuals to access information at any time and connect with others who understand their unique experiences.
Support systems for bipolar disorder are crucial in empowering individuals and enabling them to lead fulfilling lives. They contribute to reducing stigma, providing emotional support, and ensuring access to resources and education. Through these support systems, individuals with bipolar disorder can gain self-confidence, develop effective coping strategies, and improve their overall well-being.
In the next section, we explore the significance of case studies in understanding bipolar disorder and how they contribute to advancing research and knowledge in the field. Specifically, we will examine how Susan’s case study serves as a valuable contribution to furthering our understanding of this complex disorder.
The Importance of Case Studies in Understanding Bipolar Disorder
Case studies play a vital role in advancing our understanding of bipolar disorder and its complexities. They offer valuable insights into individual experiences, treatment outcomes, and the overall impact of the condition on individuals and society. Susan’s case study, in particular, provides a unique perspective that contributes to broader research and knowledge in the field.
How Case Studies Contribute to Research
Case studies provide an in-depth examination of specific individuals and their experiences with bipolar disorder. They allow researchers and healthcare professionals to observe patterns, identify commonalities, and gain valuable insights into the factors that influence symptom presentation, treatment response, and prognosis. By analyzing various case studies, researchers can generate hypotheses and refine treatment approaches to optimize outcomes for individuals with bipolar disorder.
Case studies are particularly helpful in documenting rare or atypical presentations of bipolar disorder. They shed light on lesser-known subtypes, such as rapid-cycling bipolar disorder or mixed episodes, contributing to a more comprehensive understanding of the condition. Case studies also provide opportunities for clinicians and researchers to discuss unique challenges and discover innovative interventions to improve treatment outcomes.
Susan’s Case Study in the Context of ATI Bipolar Disorder
Susan’s case study is an example of how individual experiences can inform the development of Assessment Technologies Institute (ATI) for bipolar disorder. By examining her journey, researchers can analyze treatment approaches, evaluate the effectiveness of various interventions, and develop evidence-based guidelines for managing bipolar disorder.
Susan’s case study provides rich information about the impact of medication, psychotherapy, and lifestyle modifications on symptom control and overall well-being. It offers valuable insights into the benefits and limitations of specific interventions, highlighting the importance of personalized treatment plans tailored to individual needs. Additionally, Susan’s case study can contribute to ongoing discussions about the role of support systems and the integration of peer support groups in managing and enhancing the lives of individuals with bipolar disorder.
The detailed documentation of Susan’s experiences serves as a powerful tool for healthcare providers, researchers, and individuals living with bipolar disorder. It highlights the complexities and challenges associated with the condition while fostering empathy and understanding among various stakeholders.
Case studies, such as Susan’s, play a crucial role in enhancing our understanding of bipolar disorder. They provide insights into individual experiences, treatment approaches, and the impact of the condition on individuals and society. Through these case studies, we can cultivate empathy for individuals with bipolar disorder, advocate for early diagnosis and effective treatment, and contribute to advancements in research and knowledge.
By illuminating the realities of living with bipolar disorder, we acknowledge the need for accessible mental health care, support systems, and evidence-based interventions. Susan’s case study exemplifies the importance of a comprehensive approach to managing bipolar disorder, integrating psychopharmacological interventions, psychotherapy, counseling, and lifestyle modifications.
Moving forward, it is essential to continue studying cases like Susan’s and explore the diverse experiences within the bipolar disorder population. By doing so, we can foster empathy, encourage early intervention and personalized treatment, and contribute to advancements in understanding bipolar disorder, ultimately improving the lives of individuals affected by this complex condition.
Empathy and Understanding for Individuals with Bipolar Disorder
Developing empathy and understanding for individuals with bipolar disorder is crucial in fostering a supportive and inclusive society. By recognizing the unique challenges they face and the complexity of their experiences, we can better advocate for their needs and provide the necessary resources and support.
It is important to understand that bipolar disorder is not simply a matter of mood swings or being “moody.” It is a chronic and often debilitating mental health condition that affects individuals in profound ways. The extreme highs of mania and the lows of depression can disrupt relationships, employment, and overall quality of life. Developing empathy means acknowledging that these struggles are real and offering support and understanding to those navigating them.
Encouraging Early Diagnosis and Effective Treatment
Early diagnosis and effective treatment are key factors in managing bipolar disorder and reducing the impact of its symptoms. Encouraging individuals to seek help and reducing the stigma associated with mental illness are crucial steps toward achieving early diagnosis. Increased awareness campaigns and education can empower individuals to recognize the signs and symptoms of bipolar disorder in themselves or their loved ones, facilitating timely intervention.
Once diagnosed, providing access to quality mental health care and ensuring individuals receive appropriate treatment is essential. Bipolar disorder often requires a combination of pharmacological interventions, psychotherapy, and lifestyle modifications. By advocating for comprehensive treatment plans and promoting ongoing care, we can help individuals with bipolar disorder achieve symptom control and improve their overall well-being.
The Role of Case Studies in Advancing Knowledge about Bipolar Disorder
Case studies, like Susan’s, play a significant role in advancing knowledge about bipolar disorder. They provide unique insights into individual experiences, treatment outcomes, and the wider impact of the condition. Researchers and healthcare providers can learn from these individual cases, developing evidence-based guidelines and refining treatment approaches.
Additionally, case studies contribute to reducing stigma by providing personal narratives that humanize the disorder. They showcase the challenges faced by individuals with bipolar disorder and highlight the importance of support systems, empathy, and understanding. By sharing these stories, we can help dispel misconceptions and promote a more compassionate approach toward mental health as a whole.
In conclusion, developing empathy and understanding for individuals with bipolar disorder is essential. By recognizing the complexity of their experiences, advocating for early diagnosis and effective treatment, and valuing the insights provided by case studies, we can create a society that supports and uplifts those with bipolar disorder. It is through empathy and education that we can reduce stigma, promote accessible mental health care, and improve the lives of those affected by this condition.In conclusion, gaining a comprehensive understanding of bipolar disorder is crucial in order to support individuals affected by this complex mental health condition. Through the real-life case study of Susan, we have explored the numerous facets of bipolar disorder, including its background, symptoms, and effects on daily life. Susan’s journey serves as a powerful reminder of the challenges individuals face in managing the highs and lows of bipolar disorder and emphasizes the importance of effective treatment and support systems.
We have examined the various approaches to treating and managing bipolar disorder, including psychopharmacological interventions, psychotherapy, and lifestyle modifications. Understanding the role of these treatments and the need for personalized care can significantly improve the quality of life for individuals like Susan.
Support systems also play a crucial role in helping those with bipolar disorder navigate the complexities of the condition. From family support to peer support groups and access to professional resources, fostering a strong network of assistance can provide the necessary emotional support, education, and guidance needed for individuals to effectively manage their symptoms.
Furthermore, case studies, such as Susan’s, contribute to advancing our knowledge about bipolar disorder. By delving into individual experiences, researchers gain valuable insights into treatment outcomes, prognosis, and the impact of the condition on individuals and society as a whole. These case studies foster empathy, reduce stigma, and contribute to the development of evidence-based guidelines and interventions that can improve the lives of individuals with bipolar disorder.
In fostering empathy and promoting early diagnosis, effective treatment, and ongoing support, we create a society that actively embraces and supports individuals with bipolar disorder. By encouraging understanding, reducing stigma, and prioritizing mental health care, we can ensure that those affected by bipolar disorder receive the support and resources necessary to lead fulfilling and meaningful lives. Through empathy, education, and continued research, we can work towards a future where individuals with bipolar disorder are understood, valued, and empowered to thrive.
Similar Posts

Income Requirements to Be a Foster Parent: A Comprehensive Guide
Becoming a foster parent is a noble and rewarding endeavor that can profoundly impact the lives of children in need. However, it’s essential to understand the various requirements and responsibilities that come with this role, including financial considerations. This comprehensive guide will explore the income requirements for foster parents, as well as other crucial aspects…

Family Access Berkley: A Guide to Depression Treatment and Support
Darkness can engulf not just individuals but entire families, yet Berkley’s comprehensive guide to depression treatment and support offers a beacon of hope for those seeking to reclaim the light. Depression is a complex mental health condition that affects millions of people worldwide, and its impact extends far beyond the individual experiencing it. In Berkley,…

Understanding Bipolar Codependency: The Relationship Between Codependency and Bipolar Disorder
Imagine being in a relationship where your sense of identity becomes intertwined with someone else’s emotions, needs, and desires. You constantly find yourself putting their well-being above your own, sacrificing your own happiness for theirs. This unhealthy and often destructive pattern is known as codependency. Now, imagine experiencing extreme mood swings that take you on…

The Complex Relationship Between Codependency and Depression: Understanding, Recognizing, and Healing
The intricate relationship between codependency and depression is a complex web that often leaves individuals feeling trapped in a cycle of emotional turmoil. These two conditions, while distinct, frequently intertwine, creating a challenging landscape for those affected and their loved ones. Understanding this connection is crucial for recognizing the signs and taking steps towards healing…

Most Common Reasons for Teenage Breakups: Understanding the Link with Depression
Teenage relationships are a significant part of adolescent development, often marked by intense emotions and experiences. However, these relationships can be fragile, and breakups are a common occurrence during this formative period. The prevalence of teenage breakups is high, with many adolescents experiencing the end of a romantic relationship before they reach adulthood. These breakups…

A Comprehensive Guide for Bipolar Caregivers
Caring for someone with bipolar disorder can be an emotional and challenging journey. As a caregiver, you play a crucial role in supporting your loved one through the ups and downs of this complex mental health condition. But where do you begin? How do you navigate the unfamiliar territory of bipolar caregiving? In this comprehensive…
Leave a Reply Cancel reply
Your email address will not be published. Required fields are marked *
Save my name, email, and website in this browser for the next time I comment.

Patient Case: 30-Year-Old Male With Bipolar Disorder
Nidal Moukaddam, MD, PhD, presents the case of a 30-year-old male diagnosed with bipolar 1 disorder and shares her initial impressions on diagnosis.

EP: 1 . Patient Case: 30-Year-Old Male With Bipolar Disorder
Ep: 2 . approaching the treatment of bipolar disorder, ep: 3 . treatment selection for bipolar disorder, ep: 4 . takeaways for bipolar disorder management.
Nidal Moukaddam, MD, PhD: Today, we’re going to talk about a new case. A 30-year-old man has taken short-term disability leave from work due to the progression of a depressive episode. He received a diagnosis of bipolar I disorder about 10 years ago. He had his first episode of mania at the age of 20 and 2 subsequent episodes of mania between the ages of 21 and 29. He was treated with lithium, which was highly effective, but he experienced excessive thirst and developed hyperthyroidism. His lithium level at the time was in the therapeutic range of 0.8 mEq/L. He was switched to valproate; however, valproate lacked the efficacy of lithium and caused adverse effects of sedation and weight gain. During his third manic episode, he started on olanzapine but experienced excessive weight gain. He was then cross-titrated to quetiapine, which improved his manic symptoms. However, weight gain again became an adverse effect, and he also complained of sedation. The patient reported sleeplessness and made unnecessary online purchases when unable to sleep, but the quetiapine sleepiness was unacceptable. Despite these adverse effects, he continued taking] quetiapine until he decompensated into his third depressive episode. The quetiapine was then augmented with lamotrigine, which was titrated up to 300 mg per day but demonstrated no efficacy. At the time of presentation, the patient was adhering to the medications. He did not have a substance use disorder, which was confirmed by a negative toxicology screen. His TSH [thyroid-stimulating hormone] level was in the middle of the normal range, and he had no suicidal ideations or psychotic symptoms.
I think the most important thing to do when somebody comes to you, even if they tell you they have a diagnosis, is to confirm the diagnosis. You want to start by making up your own mind, and sometimes the patient is not a good source of information. But in the case of bipolar disorder without psychosis, you expect the patient to be able to give you a solid history. Typically, the part of the history that’s hardest to nail down is mania. When people experience mania, they have excessive energy and excessive activation that creates the need for sleep, and sometimes they like it. They feel that this is the way it should be, so they don’t point it out as pathological. Now, the DSM-5 [ Diagnostic and Statistical Manual of Mental Disorders, Fifth Edition ] criteria tell us that mania that leads to hospitalization or some negative consequence like incarceration is problematic no matter what the duration is. Assuming the patient did not end up in the hospital or in prison, we want to verify the story of mania. In the current case presentation, I can see many of my colleagues saying, “Hey, you’re not giving us enough symptoms of mania. He’s a bit sleepless. He makes frivolous purchases. That’s bipolar disorder but not bipolar I; maybe it’s bipolar II.”
Thus, my first step would be to explain that this patient had at least a week without sleep. During that week, he was spacing, had pressured speech, and was talking fast to the point that others around him commented about it. He became more impulsive, and buying things was the tip of the iceberg. He also became more sexual to the point where it got him in trouble in his relationships, he spent more money than he had planned, etc. These examples of impulsivity often nail down the diagnosis of bipolar disorder. Of course, these symptoms change with the time that we live in. For example, before unlimited plans on cell phones, you would have been taught to ask: “Do you get a very high bill on your phone when you’re manic?” Because patients with mania talk a lot, and the bills would be higher when they call across state lines or internationally. First, I would recommend verifying the diagnosis. My impression of the patient is that this is somebody with a set diagnosis of bipolar I. Three manic episodes is a lot. He has impairment because of it, and it’s affected his job. Thus, my first step is confirming the diagnosis. My second would be a lot of psychoeducation; make sure that the patient understands what he’s up against and why he needs treatment.
Transcript Edited for Clarity

Evaluating the Efficacy of Lumateperone for MDD and Bipolar Depression With Mixed Features

Blue Light Blockers: A Behavior Therapy for Mania

Efficacy of Modafinil for Treatment of Neurocognitive Impairment in Bipolar Disorder

Blue Light, Depression, and Bipolar Disorder

Securing the Future of Lithium Research

An Update on Early Intervention in Psychotic Disorders
2 Commerce Drive Cranbury, NJ 08512
609-716-7777

- Search Menu
Sign in through your institution
- Browse content in Arts and Humanities
- Browse content in Archaeology
- Anglo-Saxon and Medieval Archaeology
- Archaeological Methodology and Techniques
- Archaeology by Region
- Archaeology of Religion
- Archaeology of Trade and Exchange
- Biblical Archaeology
- Contemporary and Public Archaeology
- Environmental Archaeology
- Historical Archaeology
- History and Theory of Archaeology
- Industrial Archaeology
- Landscape Archaeology
- Mortuary Archaeology
- Prehistoric Archaeology
- Underwater Archaeology
- Zooarchaeology
- Browse content in Architecture
- Architectural Structure and Design
- History of Architecture
- Residential and Domestic Buildings
- Theory of Architecture
- Browse content in Art
- Art Subjects and Themes
- History of Art
- Industrial and Commercial Art
- Theory of Art
- Biographical Studies
- Byzantine Studies
- Browse content in Classical Studies
- Classical History
- Classical Philosophy
- Classical Mythology
- Classical Numismatics
- Classical Literature
- Classical Reception
- Classical Art and Architecture
- Classical Oratory and Rhetoric
- Greek and Roman Papyrology
- Greek and Roman Epigraphy
- Greek and Roman Law
- Greek and Roman Archaeology
- Late Antiquity
- Religion in the Ancient World
- Social History
- Digital Humanities
- Browse content in History
- Colonialism and Imperialism
- Diplomatic History
- Environmental History
- Genealogy, Heraldry, Names, and Honours
- Genocide and Ethnic Cleansing
- Historical Geography
- History by Period
- History of Emotions
- History of Agriculture
- History of Education
- History of Gender and Sexuality
- Industrial History
- Intellectual History
- International History
- Labour History
- Legal and Constitutional History
- Local and Family History
- Maritime History
- Military History
- National Liberation and Post-Colonialism
- Oral History
- Political History
- Public History
- Regional and National History
- Revolutions and Rebellions
- Slavery and Abolition of Slavery
- Social and Cultural History
- Theory, Methods, and Historiography
- Urban History
- World History
- Browse content in Language Teaching and Learning
- Language Learning (Specific Skills)
- Language Teaching Theory and Methods
- Browse content in Linguistics
- Applied Linguistics
- Cognitive Linguistics
- Computational Linguistics
- Forensic Linguistics
- Grammar, Syntax and Morphology
- Historical and Diachronic Linguistics
- History of English
- Language Evolution
- Language Reference
- Language Acquisition
- Language Variation
- Language Families
- Lexicography
- Linguistic Anthropology
- Linguistic Theories
- Linguistic Typology
- Phonetics and Phonology
- Psycholinguistics
- Sociolinguistics
- Translation and Interpretation
- Writing Systems
- Browse content in Literature
- Bibliography
- Children's Literature Studies
- Literary Studies (Romanticism)
- Literary Studies (American)
- Literary Studies (Asian)
- Literary Studies (European)
- Literary Studies (Eco-criticism)
- Literary Studies (Modernism)
- Literary Studies - World
- Literary Studies (1500 to 1800)
- Literary Studies (19th Century)
- Literary Studies (20th Century onwards)
- Literary Studies (African American Literature)
- Literary Studies (British and Irish)
- Literary Studies (Early and Medieval)
- Literary Studies (Fiction, Novelists, and Prose Writers)
- Literary Studies (Gender Studies)
- Literary Studies (Graphic Novels)
- Literary Studies (History of the Book)
- Literary Studies (Plays and Playwrights)
- Literary Studies (Poetry and Poets)
- Literary Studies (Postcolonial Literature)
- Literary Studies (Queer Studies)
- Literary Studies (Science Fiction)
- Literary Studies (Travel Literature)
- Literary Studies (War Literature)
- Literary Studies (Women's Writing)
- Literary Theory and Cultural Studies
- Mythology and Folklore
- Shakespeare Studies and Criticism
- Browse content in Media Studies
- Browse content in Music
- Applied Music
- Dance and Music
- Ethics in Music
- Ethnomusicology
- Gender and Sexuality in Music
- Medicine and Music
- Music Cultures
- Music and Media
- Music and Religion
- Music and Culture
- Music Education and Pedagogy
- Music Theory and Analysis
- Musical Scores, Lyrics, and Libretti
- Musical Structures, Styles, and Techniques
- Musicology and Music History
- Performance Practice and Studies
- Race and Ethnicity in Music
- Sound Studies
- Browse content in Performing Arts
- Browse content in Philosophy
- Aesthetics and Philosophy of Art
- Epistemology
- Feminist Philosophy
- History of Western Philosophy
- Metaphysics
- Moral Philosophy
- Non-Western Philosophy
- Philosophy of Language
- Philosophy of Mind
- Philosophy of Perception
- Philosophy of Science
- Philosophy of Action
- Philosophy of Law
- Philosophy of Religion
- Philosophy of Mathematics and Logic
- Practical Ethics
- Social and Political Philosophy
- Browse content in Religion
- Biblical Studies
- Christianity
- East Asian Religions
- History of Religion
- Judaism and Jewish Studies
- Qumran Studies
- Religion and Education
- Religion and Health
- Religion and Politics
- Religion and Science
- Religion and Law
- Religion and Art, Literature, and Music
- Religious Studies
- Browse content in Society and Culture
- Cookery, Food, and Drink
- Cultural Studies
- Customs and Traditions
- Ethical Issues and Debates
- Hobbies, Games, Arts and Crafts
- Natural world, Country Life, and Pets
- Popular Beliefs and Controversial Knowledge
- Sports and Outdoor Recreation
- Technology and Society
- Travel and Holiday
- Visual Culture
- Browse content in Law
- Arbitration
- Browse content in Company and Commercial Law
- Commercial Law
- Company Law
- Browse content in Comparative Law
- Systems of Law
- Competition Law
- Browse content in Constitutional and Administrative Law
- Government Powers
- Judicial Review
- Local Government Law
- Military and Defence Law
- Parliamentary and Legislative Practice
- Construction Law
- Contract Law
- Browse content in Criminal Law
- Criminal Procedure
- Criminal Evidence Law
- Sentencing and Punishment
- Employment and Labour Law
- Environment and Energy Law
- Browse content in Financial Law
- Banking Law
- Insolvency Law
- History of Law
- Human Rights and Immigration
- Intellectual Property Law
- Browse content in International Law
- Private International Law and Conflict of Laws
- Public International Law
- IT and Communications Law
- Jurisprudence and Philosophy of Law
- Law and Politics
- Law and Society
- Browse content in Legal System and Practice
- Courts and Procedure
- Legal Skills and Practice
- Legal System - Costs and Funding
- Primary Sources of Law
- Regulation of Legal Profession
- Medical and Healthcare Law
- Browse content in Policing
- Criminal Investigation and Detection
- Police and Security Services
- Police Procedure and Law
- Police Regional Planning
- Browse content in Property Law
- Personal Property Law
- Restitution
- Study and Revision
- Terrorism and National Security Law
- Browse content in Trusts Law
- Wills and Probate or Succession
- Browse content in Medicine and Health
- Browse content in Allied Health Professions
- Arts Therapies
- Clinical Science
- Dietetics and Nutrition
- Occupational Therapy
- Operating Department Practice
- Physiotherapy
- Radiography
- Speech and Language Therapy
- Browse content in Anaesthetics
- General Anaesthesia
- Clinical Neuroscience
- Browse content in Clinical Medicine
- Acute Medicine
- Cardiovascular Medicine
- Clinical Genetics
- Clinical Pharmacology and Therapeutics
- Dermatology
- Endocrinology and Diabetes
- Gastroenterology
- Genito-urinary Medicine
- Geriatric Medicine
- Infectious Diseases
- Medical Toxicology
- Medical Oncology
- Pain Medicine
- Palliative Medicine
- Rehabilitation Medicine
- Respiratory Medicine and Pulmonology
- Rheumatology
- Sleep Medicine
- Sports and Exercise Medicine
- Community Medical Services
- Critical Care
- Emergency Medicine
- Forensic Medicine
- Haematology
- History of Medicine
- Browse content in Medical Skills
- Clinical Skills
- Communication Skills
- Nursing Skills
- Surgical Skills
- Browse content in Medical Dentistry
- Oral and Maxillofacial Surgery
- Paediatric Dentistry
- Restorative Dentistry and Orthodontics
- Surgical Dentistry
- Medical Ethics
- Medical Statistics and Methodology
- Browse content in Neurology
- Clinical Neurophysiology
- Neuropathology
- Nursing Studies
- Browse content in Obstetrics and Gynaecology
- Gynaecology
- Occupational Medicine
- Ophthalmology
- Otolaryngology (ENT)
- Browse content in Paediatrics
- Neonatology
- Browse content in Pathology
- Chemical Pathology
- Clinical Cytogenetics and Molecular Genetics
- Histopathology
- Medical Microbiology and Virology
- Patient Education and Information
- Browse content in Pharmacology
- Psychopharmacology
- Browse content in Popular Health
- Caring for Others
- Complementary and Alternative Medicine
- Self-help and Personal Development
- Browse content in Preclinical Medicine
- Cell Biology
- Molecular Biology and Genetics
- Reproduction, Growth and Development
- Primary Care
- Professional Development in Medicine
- Browse content in Psychiatry
- Addiction Medicine
- Child and Adolescent Psychiatry
- Forensic Psychiatry
- Learning Disabilities
- Old Age Psychiatry
- Psychotherapy
- Browse content in Public Health and Epidemiology
- Epidemiology
- Public Health
- Browse content in Radiology
- Clinical Radiology
- Interventional Radiology
- Nuclear Medicine
- Radiation Oncology
- Reproductive Medicine
- Browse content in Surgery
- Cardiothoracic Surgery
- Gastro-intestinal and Colorectal Surgery
- General Surgery
- Neurosurgery
- Paediatric Surgery
- Peri-operative Care
- Plastic and Reconstructive Surgery
- Surgical Oncology
- Transplant Surgery
- Trauma and Orthopaedic Surgery
- Vascular Surgery
- Browse content in Science and Mathematics
- Browse content in Biological Sciences
- Aquatic Biology
- Biochemistry
- Bioinformatics and Computational Biology
- Developmental Biology
- Ecology and Conservation
- Evolutionary Biology
- Genetics and Genomics
- Microbiology
- Molecular and Cell Biology
- Natural History
- Plant Sciences and Forestry
- Research Methods in Life Sciences
- Structural Biology
- Systems Biology
- Zoology and Animal Sciences
- Browse content in Chemistry
- Analytical Chemistry
- Computational Chemistry
- Crystallography
- Environmental Chemistry
- Industrial Chemistry
- Inorganic Chemistry
- Materials Chemistry
- Medicinal Chemistry
- Mineralogy and Gems
- Organic Chemistry
- Physical Chemistry
- Polymer Chemistry
- Study and Communication Skills in Chemistry
- Theoretical Chemistry
- Browse content in Computer Science
- Artificial Intelligence
- Computer Architecture and Logic Design
- Game Studies
- Human-Computer Interaction
- Mathematical Theory of Computation
- Programming Languages
- Software Engineering
- Systems Analysis and Design
- Virtual Reality
- Browse content in Computing
- Business Applications
- Computer Security
- Computer Games
- Computer Networking and Communications
- Digital Lifestyle
- Graphical and Digital Media Applications
- Operating Systems
- Browse content in Earth Sciences and Geography
- Atmospheric Sciences
- Environmental Geography
- Geology and the Lithosphere
- Maps and Map-making
- Meteorology and Climatology
- Oceanography and Hydrology
- Palaeontology
- Physical Geography and Topography
- Regional Geography
- Soil Science
- Urban Geography
- Browse content in Engineering and Technology
- Agriculture and Farming
- Biological Engineering
- Civil Engineering, Surveying, and Building
- Electronics and Communications Engineering
- Energy Technology
- Engineering (General)
- Environmental Science, Engineering, and Technology
- History of Engineering and Technology
- Mechanical Engineering and Materials
- Technology of Industrial Chemistry
- Transport Technology and Trades
- Browse content in Environmental Science
- Applied Ecology (Environmental Science)
- Conservation of the Environment (Environmental Science)
- Environmental Sustainability
- Environmentalist Thought and Ideology (Environmental Science)
- Management of Land and Natural Resources (Environmental Science)
- Natural Disasters (Environmental Science)
- Nuclear Issues (Environmental Science)
- Pollution and Threats to the Environment (Environmental Science)
- Social Impact of Environmental Issues (Environmental Science)
- History of Science and Technology
- Browse content in Materials Science
- Ceramics and Glasses
- Composite Materials
- Metals, Alloying, and Corrosion
- Nanotechnology
- Browse content in Mathematics
- Applied Mathematics
- Biomathematics and Statistics
- History of Mathematics
- Mathematical Education
- Mathematical Finance
- Mathematical Analysis
- Numerical and Computational Mathematics
- Probability and Statistics
- Pure Mathematics
- Browse content in Neuroscience
- Cognition and Behavioural Neuroscience
- Development of the Nervous System
- Disorders of the Nervous System
- History of Neuroscience
- Invertebrate Neurobiology
- Molecular and Cellular Systems
- Neuroendocrinology and Autonomic Nervous System
- Neuroscientific Techniques
- Sensory and Motor Systems
- Browse content in Physics
- Astronomy and Astrophysics
- Atomic, Molecular, and Optical Physics
- Biological and Medical Physics
- Classical Mechanics
- Computational Physics
- Condensed Matter Physics
- Electromagnetism, Optics, and Acoustics
- History of Physics
- Mathematical and Statistical Physics
- Measurement Science
- Nuclear Physics
- Particles and Fields
- Plasma Physics
- Quantum Physics
- Relativity and Gravitation
- Semiconductor and Mesoscopic Physics
- Browse content in Psychology
- Affective Sciences
- Clinical Psychology
- Cognitive Psychology
- Cognitive Neuroscience
- Criminal and Forensic Psychology
- Developmental Psychology
- Educational Psychology
- Evolutionary Psychology
- Health Psychology
- History and Systems in Psychology
- Music Psychology
- Neuropsychology
- Organizational Psychology
- Psychological Assessment and Testing
- Psychology of Human-Technology Interaction
- Psychology Professional Development and Training
- Research Methods in Psychology
- Social Psychology
- Browse content in Social Sciences
- Browse content in Anthropology
- Anthropology of Religion
- Human Evolution
- Medical Anthropology
- Physical Anthropology
- Regional Anthropology
- Social and Cultural Anthropology
- Theory and Practice of Anthropology
- Browse content in Business and Management
- Business Ethics
- Business Strategy
- Business History
- Business and Technology
- Business and Government
- Business and the Environment
- Comparative Management
- Corporate Governance
- Corporate Social Responsibility
- Entrepreneurship
- Health Management
- Human Resource Management
- Industrial and Employment Relations
- Industry Studies
- Information and Communication Technologies
- International Business
- Knowledge Management
- Management and Management Techniques
- Operations Management
- Organizational Theory and Behaviour
- Pensions and Pension Management
- Public and Nonprofit Management
- Social Issues in Business and Management
- Strategic Management
- Supply Chain Management
- Browse content in Criminology and Criminal Justice
- Criminal Justice
- Criminology
- Forms of Crime
- International and Comparative Criminology
- Youth Violence and Juvenile Justice
- Development Studies
- Browse content in Economics
- Agricultural, Environmental, and Natural Resource Economics
- Asian Economics
- Behavioural Finance
- Behavioural Economics and Neuroeconomics
- Econometrics and Mathematical Economics
- Economic History
- Economic Systems
- Economic Methodology
- Economic Development and Growth
- Financial Markets
- Financial Institutions and Services
- General Economics and Teaching
- Health, Education, and Welfare
- History of Economic Thought
- International Economics
- Labour and Demographic Economics
- Law and Economics
- Macroeconomics and Monetary Economics
- Microeconomics
- Public Economics
- Urban, Rural, and Regional Economics
- Welfare Economics
- Browse content in Education
- Adult Education and Continuous Learning
- Care and Counselling of Students
- Early Childhood and Elementary Education
- Educational Equipment and Technology
- Educational Strategies and Policy
- Higher and Further Education
- Organization and Management of Education
- Philosophy and Theory of Education
- Schools Studies
- Secondary Education
- Teaching of a Specific Subject
- Teaching of Specific Groups and Special Educational Needs
- Teaching Skills and Techniques
- Browse content in Environment
- Applied Ecology (Social Science)
- Climate Change
- Conservation of the Environment (Social Science)
- Environmentalist Thought and Ideology (Social Science)
- Management of Land and Natural Resources (Social Science)
- Natural Disasters (Environment)
- Pollution and Threats to the Environment (Social Science)
- Social Impact of Environmental Issues (Social Science)
- Sustainability
- Browse content in Human Geography
- Cultural Geography
- Economic Geography
- Political Geography
- Browse content in Interdisciplinary Studies
- Communication Studies
- Museums, Libraries, and Information Sciences
- Browse content in Politics
- African Politics
- Asian Politics
- Chinese Politics
- Comparative Politics
- Conflict Politics
- Elections and Electoral Studies
- Environmental Politics
- Ethnic Politics
- European Union
- Foreign Policy
- Gender and Politics
- Human Rights and Politics
- Indian Politics
- International Relations
- International Organization (Politics)
- Irish Politics
- Latin American Politics
- Middle Eastern Politics
- Political Behaviour
- Political Economy
- Political Institutions
- Political Methodology
- Political Communication
- Political Philosophy
- Political Sociology
- Political Theory
- Politics and Law
- Politics of Development
- Public Policy
- Public Administration
- Qualitative Political Methodology
- Quantitative Political Methodology
- Regional Political Studies
- Russian Politics
- Security Studies
- State and Local Government
- UK Politics
- US Politics
- Browse content in Regional and Area Studies
- African Studies
- Asian Studies
- East Asian Studies
- Japanese Studies
- Latin American Studies
- Middle Eastern Studies
- Native American Studies
- Scottish Studies
- Browse content in Research and Information
- Research Methods
- Browse content in Social Work
- Addictions and Substance Misuse
- Adoption and Fostering
- Care of the Elderly
- Child and Adolescent Social Work
- Couple and Family Social Work
- Direct Practice and Clinical Social Work
- Emergency Services
- Human Behaviour and the Social Environment
- International and Global Issues in Social Work
- Mental and Behavioural Health
- Social Justice and Human Rights
- Social Policy and Advocacy
- Social Work and Crime and Justice
- Social Work Macro Practice
- Social Work Practice Settings
- Social Work Research and Evidence-based Practice
- Welfare and Benefit Systems
- Browse content in Sociology
- Childhood Studies
- Community Development
- Comparative and Historical Sociology
- Disability Studies
- Economic Sociology
- Gender and Sexuality
- Gerontology and Ageing
- Health, Illness, and Medicine
- Marriage and the Family
- Migration Studies
- Occupations, Professions, and Work
- Organizations
- Population and Demography
- Race and Ethnicity
- Social Theory
- Social Movements and Social Change
- Social Research and Statistics
- Social Stratification, Inequality, and Mobility
- Sociology of Religion
- Sociology of Education
- Sport and Leisure
- Urban and Rural Studies
- Browse content in Warfare and Defence
- Defence Strategy, Planning, and Research
- Land Forces and Warfare
- Military Administration
- Military Life and Institutions
- Naval Forces and Warfare
- Other Warfare and Defence Issues
- Peace Studies and Conflict Resolution
- Weapons and Equipment

- < Previous chapter
- Next chapter >
5 Psychiatric Treatment of Bipolar Disorder: The Case of Janice
- Published: February 2013
- Cite Icon Cite
- Permissions Icon Permissions
Chapter 5 covers the psychiatric treatment of bipolar disorder, including a case history, key principles, assessment strategy, differential diagnosis, case formulation, treatment planning, nonspecific factors in treatment, potential treatment obstacles, ethical considerations, common mistakes to avoid in treatment, and relapse prevention.
Personal account
- Sign in with email/username & password
- Get email alerts
- Save searches
- Purchase content
- Activate your purchase/trial code
- Add your ORCID iD
Institutional access
Sign in with a library card.
- Sign in with username/password
- Recommend to your librarian
- Institutional account management
- Get help with access
Access to content on Oxford Academic is often provided through institutional subscriptions and purchases. If you are a member of an institution with an active account, you may be able to access content in one of the following ways:
IP based access
Typically, access is provided across an institutional network to a range of IP addresses. This authentication occurs automatically, and it is not possible to sign out of an IP authenticated account.
Choose this option to get remote access when outside your institution. Shibboleth/Open Athens technology is used to provide single sign-on between your institution’s website and Oxford Academic.
- Click Sign in through your institution.
- Select your institution from the list provided, which will take you to your institution's website to sign in.
- When on the institution site, please use the credentials provided by your institution. Do not use an Oxford Academic personal account.
- Following successful sign in, you will be returned to Oxford Academic.
If your institution is not listed or you cannot sign in to your institution’s website, please contact your librarian or administrator.
Enter your library card number to sign in. If you cannot sign in, please contact your librarian.
Society Members
Society member access to a journal is achieved in one of the following ways:
Sign in through society site
Many societies offer single sign-on between the society website and Oxford Academic. If you see ‘Sign in through society site’ in the sign in pane within a journal:
- Click Sign in through society site.
- When on the society site, please use the credentials provided by that society. Do not use an Oxford Academic personal account.
If you do not have a society account or have forgotten your username or password, please contact your society.
Sign in using a personal account
Some societies use Oxford Academic personal accounts to provide access to their members. See below.
A personal account can be used to get email alerts, save searches, purchase content, and activate subscriptions.
Some societies use Oxford Academic personal accounts to provide access to their members.
Viewing your signed in accounts
Click the account icon in the top right to:
- View your signed in personal account and access account management features.
- View the institutional accounts that are providing access.
Signed in but can't access content
Oxford Academic is home to a wide variety of products. The institutional subscription may not cover the content that you are trying to access. If you believe you should have access to that content, please contact your librarian.
For librarians and administrators, your personal account also provides access to institutional account management. Here you will find options to view and activate subscriptions, manage institutional settings and access options, access usage statistics, and more.
Our books are available by subscription or purchase to libraries and institutions.
| Month: | Total Views: |
|---|---|
| October 2022 | 4 |
| November 2022 | 1 |
| December 2022 | 5 |
| January 2023 | 6 |
| February 2023 | 4 |
| March 2023 | 6 |
| April 2023 | 1 |
| May 2023 | 1 |
| June 2023 | 3 |
| July 2023 | 2 |
| August 2023 | 2 |
| September 2023 | 4 |
| October 2023 | 4 |
| November 2023 | 3 |
| December 2023 | 4 |
| January 2024 | 14 |
| February 2024 | 6 |
| March 2024 | 3 |
| April 2024 | 3 |
| May 2024 | 6 |
| June 2024 | 5 |
| July 2024 | 2 |
- About Oxford Academic
- Publish journals with us
- University press partners
- What we publish
- New features
- Open access
- Rights and permissions
- Accessibility
- Advertising
- Media enquiries
- Oxford University Press
- Oxford Languages
- University of Oxford
Oxford University Press is a department of the University of Oxford. It furthers the University's objective of excellence in research, scholarship, and education by publishing worldwide
- Copyright © 2024 Oxford University Press
- Cookie settings
- Cookie policy
- Privacy policy
- Legal notice
This Feature Is Available To Subscribers Only
Sign In or Create an Account
This PDF is available to Subscribers Only
For full access to this pdf, sign in to an existing account, or purchase an annual subscription.

- Presidential Message
- Nominations/Elections
- Past Presidents
- Member Spotlight
- Fellow List
- Fellowship Nominations
- Current Awardees
- Award Nominations
- SCP Mentoring Program
- SCP Book Club
- Pub Fee & Certificate Purchases
- LEAD Program
- Introduction
- Find a Treatment
- Submit Proposal
- Dissemination and Implementation
- Diversity Resources
- Graduate School Applicants
- Student Resources
- Early Career Resources
- Principles for Training in Evidence-Based Psychology
- Advances in Psychotherapy
- Announcements
- Submit Blog
- Student Blog
- The Clinical Psychologist
- CP:SP Journal
- APA Convention
- SCP Conference
CASE STUDY Richard (bipolar disorder, substance use disorder)
Case study details.
Richard is a 62-year-old single man who says that his substance dependence and his bipolar disorder both emerged in his late teens. He says that he started to drink to “feel better” when his episodes of depression made it hard for him to interact with his peers. He also states that alcohol and cocaine are a natural part of his manic episodes. He also notes that coming off the cocaine and binge drinking contribute to low mood, but he has not responded well to referrals to AA and past inpatient stays have led to only temporary abstinence. Yet, Richard is now trying to forge a closer relationship to his adult children, and he says he is especially motivated to get a better handle on both his bipolar disorder and his substance use. He has been more compliant with his mood stabilizing and antidepressant medication, and his psychiatrist would like his dual diagnoses addressed with psychotherapy.
- Alcohol Use
- Elevated Mood
- Impulsivity
- Mania/Hypomania
- Mood Cycles
- Substance Abuse
Diagnoses and Related Treatments
1. bipolar disorder, 2. mixed substance use/dependence.
Thank you for supporting the Society of Clinical Psychology. To enhance our membership benefits, we are requesting that members fill out their profile in full. This will help the Division to gather appropriate data to provide you with the best benefits. You will be able to access all other areas of the website, once your profile is submitted. Thank you and please feel free to email the Central Office at [email protected] if you have any questions
Case Study 1: A 55-Year-Old Woman With Progressive Cognitive, Perceptual, and Motor Impairments
Information & authors, metrics & citations, view options, case presentation, what are diagnostic considerations based on the history how might a clinical examination help to narrow the differential diagnosis.
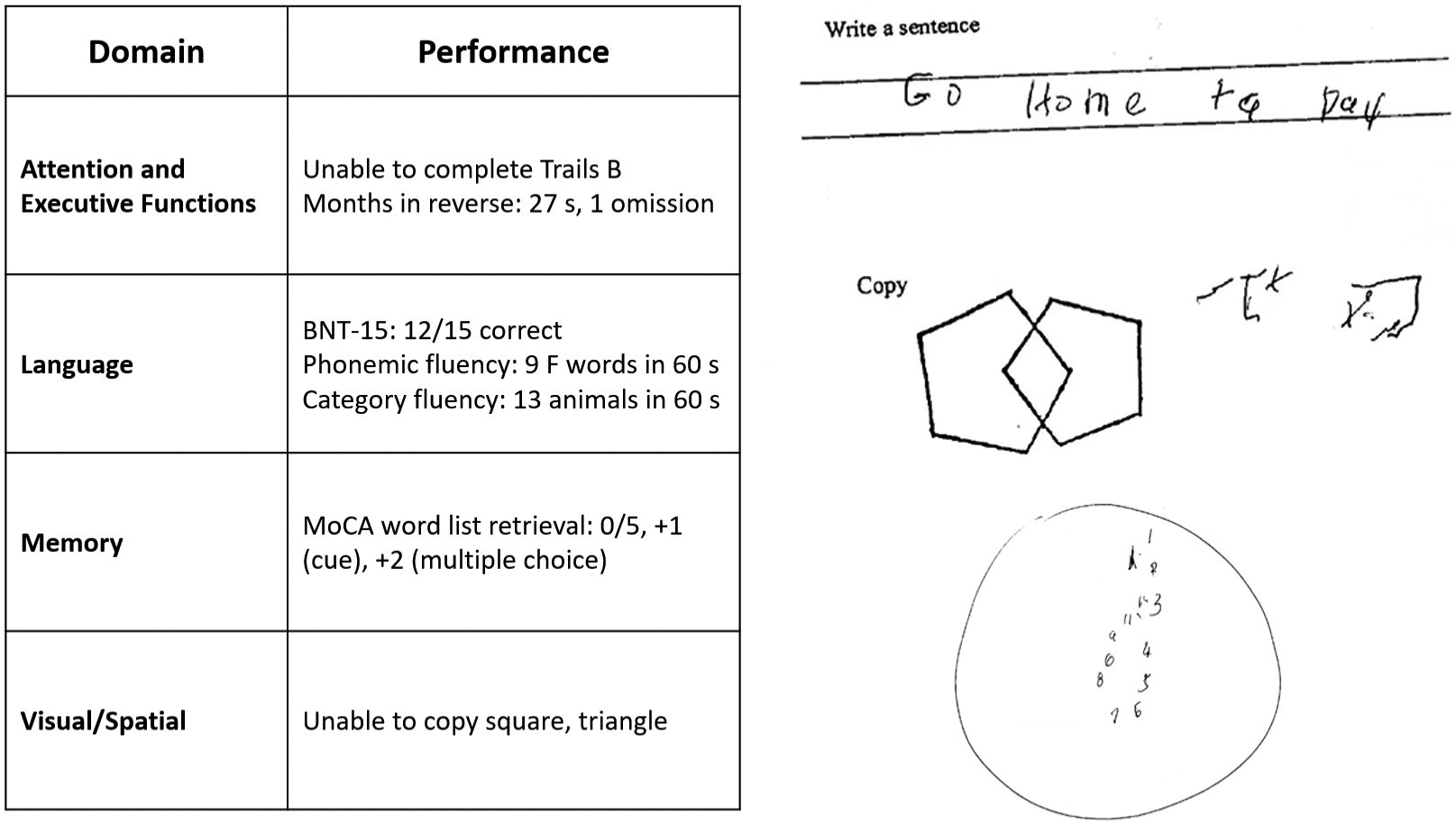
How Does the Examination Contribute to Our Understanding of Diagnostic Considerations? What Additional Tests or Studies Are Indicated?
| Feature | Posterior cortical atrophy | Corticobasal syndrome |
|---|---|---|
| Cognitive and motor features | Visual-perceptual: space perception deficit, simultanagnosia, object perception deficit, environmental agnosia, alexia, apperceptive prosopagnosia, and homonymous visual field defect | Motor: limb rigidity or akinesia, limb dystonia, and limb myoclonus |
| Visual-motor: constructional dyspraxia, oculomotor apraxia, optic ataxia, and dressing apraxia | ||
| Other: left/right disorientation, acalculia, limb apraxia, agraphia, and finger agnosia | Higher cortical features: limb or orobuccal apraxia, cortical sensory deficit, and alien limb phenomena | |
| Imaging features (MRI, FDG-PET, SPECT) | Predominant occipito-parietal or occipito-temporal atrophy, and hypometabolism or hypoperfusion | Asymmetric perirolandic, posterior frontal, parietal atrophy, and hypometabolism or hypoperfusion |
| Neuropathological associations | AD>CBD, LBD, TDP, JCD | CBD>PSP, AD, TDP |
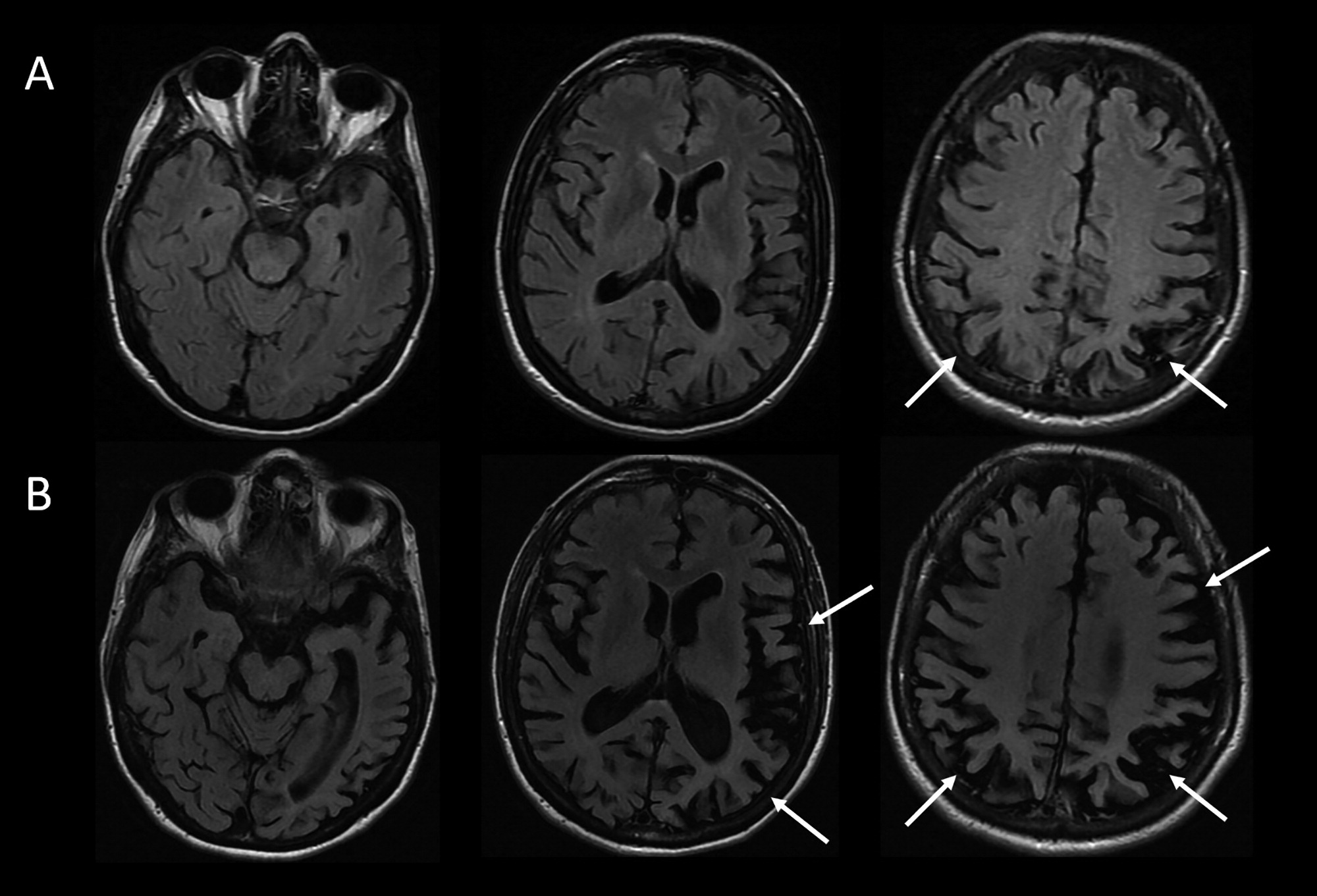
Considering This Additional Data, What Would Be an Appropriate Diagnostic Formulation?
Does information about the longitudinal course of her illness alter the formulation about the most likely underlying neuropathological process, neuropathology.
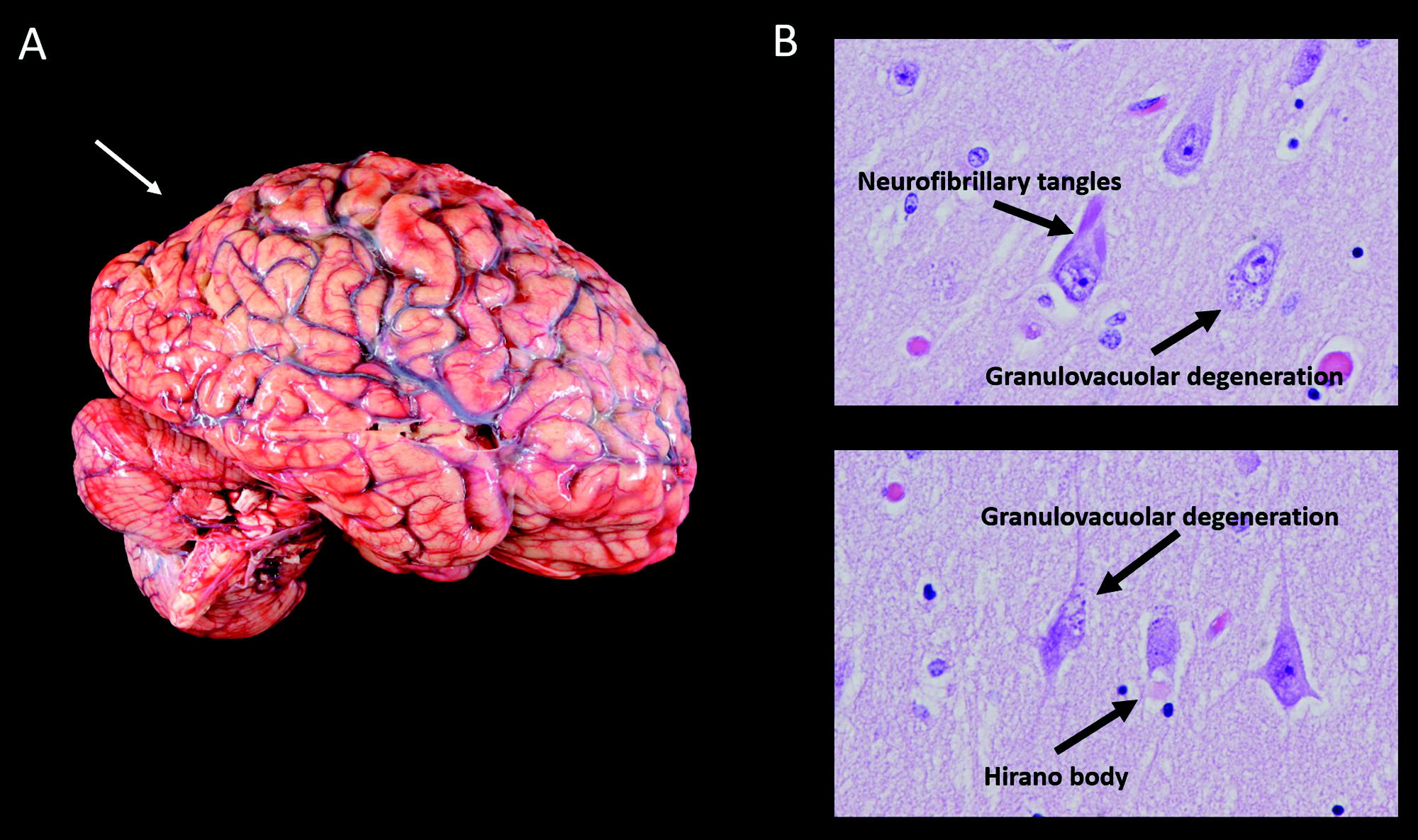
| Feature | Case of PCA/CBS due to AD | Exemplar case of CBD |
|---|---|---|
| Macroscopic findings | Cortical atrophy: symmetric, mild | Cortical atrophy: often asymmetric, predominantly affecting perirolandic cortex |
| Substantia nigra: appropriately pigmented | Substantia nigra: severely depigmented | |
| Microscopic findings | Tau neurofibrillary tangles and beta-amyloid plaques | Primary tauopathy |
| No tau pathology in white matter | Tau pathology involves white matter | |
| Hirano bodies, granulovacuolar degeneration | Ballooned neurons, astrocytic plaques, and oligodendroglial coiled bodies | |
| (Lewy bodies, limbic) |
Information
Published in.

- Posterior Cortical Atrophy
- Corticobasal Syndrome
- Atypical Alzheimer Disease
- Network Degeneration
Competing Interests
Funding information, export citations.
If you have the appropriate software installed, you can download article citation data to the citation manager of your choice. Simply select your manager software from the list below and click Download. For more information or tips please see 'Downloading to a citation manager' in the Help menu .
| Format | |
|---|---|
| Citation style | |
| Style | |
To download the citation to this article, select your reference manager software.
There are no citations for this item
View options
Login options.
Already a subscriber? Access your subscription through your login credentials or your institution for full access to this article.
Purchase Options
Purchase this article to access the full text.
PPV Articles - Journal of Neuropsychiatry and Clinical Neurosciences
Not a subscriber?
Subscribe Now / Learn More
PsychiatryOnline subscription options offer access to the DSM-5-TR ® library, books, journals, CME, and patient resources. This all-in-one virtual library provides psychiatrists and mental health professionals with key resources for diagnosis, treatment, research, and professional development.
Need more help? PsychiatryOnline Customer Service may be reached by emailing [email protected] or by calling 800-368-5777 (in the U.S.) or 703-907-7322 (outside the U.S.).
Share article link
Copying failed.
PREVIOUS ARTICLE
Next article, request username.
Can't sign in? Forgot your username? Enter your email address below and we will send you your username
If the address matches an existing account you will receive an email with instructions to retrieve your username
Create a new account
Change password, password changed successfully.
Your password has been changed
Reset password
Can't sign in? Forgot your password?
Enter your email address below and we will send you the reset instructions
If the address matches an existing account you will receive an email with instructions to reset your password.
Your Phone has been verified
As described within the American Psychiatric Association (APA)'s Privacy Policy and Terms of Use , this website utilizes cookies, including for the purpose of offering an optimal online experience and services tailored to your preferences. Please read the entire Privacy Policy and Terms of Use. By closing this message, browsing this website, continuing the navigation, or otherwise continuing to use the APA's websites, you confirm that you understand and accept the terms of the Privacy Policy and Terms of Use, including the utilization of cookies.

Transforming the understanding and treatment of mental illnesses.
Información en español
Celebrating 75 Years! Learn More >>
- Science News
- Meetings and Events
- Social Media
- Press Resources
- Email Updates
- Innovation Speaker Series
Genomic Data From More Than 41,000 People Shed New Light on Bipolar Disorder
September 29, 2021 • Research Highlight
In the largest genome-wide association study of bipolar disorder to date, researchers found about twice as many genetic locations associated with bipolar disorder as reported in previous studies. These and other genome-wide findings help improve our understanding of the biological origins of bipolar disorder and suggest some promising genes for further research.
The study, led by the Psychiatric Genomics Consortium bipolar disorder working group, is published in Nature Genetics . The Psychiatric Genomics Consortium is a global collaborative effort consisting of more than 800 investigators, including researchers in the National Institute of Mental Health (NIMH) Intramural Research Program and extramural scientists conducting NIMH-supported research.
Bipolar disorder is a mental illness characterized by episodes of mania and depression that can seriously impair day-to-day functioning. Affecting up to 50 million people worldwide, bipolar disorder is a major public health concern. Although evidence suggests that genes play an important role in the development of bipolar disorder, researchers still do not have a clear understanding of the disorder’s specific biological causes. Examining common genetic variations in the genomes (or complete set of DNA) of people with bipolar disorder is a way that scientists can home in on the genetic factors that are likely to play a causal role in the disorder.
For this study, the researchers analyzed genomic data from 57 groups of participants across Europe, North America, and Australia. These cohorts included individuals receiving clinical care for bipolar disorder and individuals classified as having bipolar disorder based on data from health registries, electronic health records, or repositories. The total combined sample included 41,917 individuals with bipolar disorder and 371,549 individuals without bipolar disorder.
The researchers used an approach known as a genome-wide association study (GWAS) , which allowed them to identify common genetic variations that are more likely to occur in people with bipolar disorder. Identifying these variations can provide important clues about the biological pathways and processes that are likely to be involved in the disorder.
According to the GWAS results, a total of 64 genomic locations, or risk loci , were associated with bipolar disorder even after accounting for all the variations studied across the genome. These 64 risk loci included 33 that had not been reported in previous bipolar disorder studies. Among the novel loci, the researchers found that bipolar disorder was associated with the major histocompatibility complex, which is a large group of genes involved in immune function. They also found a correlation between bipolar disorder and loci linked to other psychiatric disorders, including schizophrenia, major depression, and childhood-onset disorders such as attention-deficit/hyperactivity disorder (ADHD).
The study findings also revealed genome-wide genetic overlaps, or correlations, between bipolar disorder and certain traits. For example, the results showed a genetic correlation between bipolar disorder and both alcohol use and smoking, as well as genetic correlations with some aspects of sleep (daytime sleepiness, insomnia, and sleep duration).
The researchers also compared genetic overlap between the two types of bipolar disorder: bipolar I disorder (which includes manic episodes and, typically, depressive episodes) and bipolar II disorder (which includes depressive episodes and hypomanic episodes). As expected, the results indicated a substantial but incomplete genetic overlap between the two types. Comparing the two types and their associations with other psychiatric disorders, the researchers found that bipolar I disorder showed a stronger genetic correlation with schizophrenia, whereas bipolar II disorder was more closely correlated with major depression. Additional studies with detailed trait data for large cohorts will be essential for further understanding the genetic components of these bipolar disorder types.
Drawing from the GWAS results, the researchers found that the 64 risk loci contained at least 161 individual genes. Some of these genes play a role in how neurons signal to each other in the brain. Some of these genes are also known to be targets for certain types of drugs currently used to treat bipolar disorder, such as antipsychotics, mood stabilizers, and antiepileptics. And some genes are known to be targets for other drug types, including calcium channel blockers (typically used to treat high blood pressure) and certain anesthetics.
The researchers then used an analytic technique called “fine-mapping” to connect risk loci with specific genes that are most likely to play a causal role in bipolar disorder. This technique identified 15 genes with the strongest evidence, which suggests they are promising candidates for further study.
Overall, the study findings confirmed many of the risk loci and genetic correlations reported in previous studies. But the study also represents an advance for the field, as a 1.5-fold increase in the number of participants effectively doubled the number of loci identified as associated with bipolar disorder. According to the researchers, this marks an “inflection point” in discovery because it indicates that the number of loci identified will continue to increase rapidly with the addition of new cohorts.
Taken together, these findings establish a more detailed picture of the genetic factors that underlie bipolar disorder and suggest possible biological targets for new treatments.
Mullins, N., Forstner, A. J., O’Connell, K. S., Coombes, B., Coleman, J. R., Qiao, Z., Als, T. D., Bigdeli, T. B., Børte, S., Bryois, J., Charney, A. W., Drange, O. K., Gandal, M. J., Hagenaars, S. P., Ikeda, M., Kamitaki, N., Kim, M., Krebs, K., Panagiotaropoulou, G.,…Andreassen, O.A. (2021). Genome-wide association study of more than 40,000 bipolar disorder cases provides new insights into the underlying biology. Nature Genetics, 53, 817–829. https://doi.org/10.1038/s41588-021-00857-4
MH109528 , MH094421 , MH085520
An official website of the United States government
The .gov means it’s official. Federal government websites often end in .gov or .mil. Before sharing sensitive information, make sure you’re on a federal government site.
The site is secure. The https:// ensures that you are connecting to the official website and that any information you provide is encrypted and transmitted securely.
- Publications
- Account settings
Preview improvements coming to the PMC website in October 2024. Learn More or Try it out now .
- Advanced Search
- Journal List
- Cambridge Open

Genetic contributions to bipolar disorder: current status and future directions
Kevin s. o'connell.
1 Division of Mental Health and Addiction, NORMENT Centre, Institute of Clinical Medicine, University of Oslo, Oslo University Hospital, 0407Oslo, Norway
Brandon J. Coombes
2 Department of Health Sciences Research, Mayo Clinic, Rochester, Minnesota, USA
Bipolar disorder (BD) is a highly heritable mental disorder and is estimated to affect about 50 million people worldwide. Our understanding of the genetic etiology of BD has greatly increased in recent years with advances in technology and methodology as well as the adoption of international consortiums and large population-based biobanks. It is clear that BD is also highly heterogeneous and polygenic and shows substantial genetic overlap with other psychiatric disorders. Genetic studies of BD suggest that the number of associated loci is expected to substantially increase in larger future studies and with it, improved genetic prediction of the disorder. Still, a number of challenges remain to fully characterize the genetic architecture of BD. First among these is the need to incorporate ancestrally-diverse samples to move research away from a Eurocentric bias that has the potential to exacerbate health disparities already seen in BD. Furthermore, incorporation of population biobanks, registry data, and electronic health records will be required to increase the sample size necessary for continued genetic discovery, while increased deep phenotyping is necessary to elucidate subtypes within BD. Lastly, the role of rare variation in BD remains to be determined. Meeting these challenges will enable improved identification of causal variants for the disorder and also allow for equitable future clinical applications of both genetic risk prediction and therapeutic interventions.
Definition of illness
Affective disorders are classified along a spectrum from unipolar depression to bipolar disorder (BD) type II and type I (Carvalho, Firth, & Vieta, 2020 ; Grande, Berk, Birmaher, & Vieta, 2016 ). The presence of recurring manic or hypomanic episodes alternating with euthymia or depressive episodes distinguishes BD from other affective disorders (American Psychiatric Association, 2013 ; World Health Organization et al., 1992 ). BD type I (BDI) is characterized by alternating manic and depressive episodes ( Fig. 1 ). Psychotic symptoms also occur in a majority of these patients which may lead to compromised functioning and hospitalization. The Diagnostic and Statistical Manual of Mental Disorder, Fifth Edition (DSM-5) also allows for individuals impaired by manic episodes without depression to still be diagnosed with BDI (American Psychiatric Association, 2013 ). In comparison, a diagnosis of BD type II (BDII) is based on the occurrence of at least one depressive and one hypomanic episode during the lifetime, but no manic episodes ( Fig. 1 ). A diagnosis of BD not otherwise specified or BD unspecified may be given when a patient has bipolar symptoms that do not fit within these major subtype categories. The DSM-5 also includes specifiers which define the clinical features of episodes and the course of the disorder, namely, anxious distress, mixed features, rapid cycling, melancholic features, atypical features, psychotic features (mood-congruent and mood-incongruent), catatonia, peripartum onset, and seasonal pattern (American Psychiatric Association, 2013 ). In addition, the DSM-5 includes schizoaffective BD as a distinct diagnosis wherein individuals suffer from psychotic symptoms as well as episodes of mania or depression (American Psychiatric Association, 2013 ).

Polarity of symptoms for bipolar disorder subtypes. Bipolar disorder type I is characterized by at least one manic episode. Bipolar disorder type II is characterized by at least one depressive and one hypomanic episode during the lifetime, but no manic episodes. Major depressive disorder does not include episodes of hypomania or mania.
Epidemiology
BD is projected to affect about 50 million people worldwide (GBD 2016 Disease and Injury Incidence and Prevalence Collaborators, 2017 ). The BD subtypes each have an estimated lifetime prevalence of approximately 1% (Merikangas et al., 2007 , 2011 ) although large ranges in lifetime prevalence have been reported (BDI: 0.1–1.7%, BDII: 0.1–3.0%) (Angst, 1998 ; Merikangas et al., 2007 , 2011 ). Most studies report no gender differences in the prevalence of BD; however, women may be at increased risk for BDII, rapid cycling, and mixed episodes (Diflorio & Jones, 2010 ; Nivoli et al., 2011 ). The mean age of onset of BD is at approximately 20 years. An earlier age of onset is associated with poorer prognosis, increased comorbidity, onset beginning with depression, and more severe depressive episodes, as well as longer treatment delays (Joslyn, Hawes, Hunt, & Mitchell, 2016 ). Additionally, initial depressive episodes may lead to a misdiagnosis of major depressive disorder until the onset of manic or hypomanic episodes necessary to confirm BD (Zimmerman, Ruggero, Chelminski, & Young, 2008 ).
BD is often comorbid with other psychiatric (Eser, Kacar, Kilciksiz, Yalçinay-Inan, & Ongur, 2018 ; Frías, Baltasar, & Birmaher, 2016 ; Salloum & Brown, 2017 ) and non-psychiatric disorders (Bortolato, Berk, Maes, McIntyre, & Carvalho, 2016 ; Correll et al., 2017 ; Vancampfort et al., 2016 ). It is estimated that >90% of BD patients have at least one lifetime comorbid disorder, and >70% present with three or more comorbid disorders during their lifetime (Merikangas et al., 2007 ). Such ubiquitous comorbidity within BD suggests the disturbance of multiple systems and pathways, and the presence of comorbidities is associated with increased premature mortality in BD when compared to the general population (Kessing, Vradi, McIntyre, & Andersen, 2015 ; Roshanaei-Moghaddam & Katon, 2009 ).
Classical genetic epidemiology
Family studies.
Genetic factors for BD were first investigated using twin, family, and adoption studies. These studies established that family history of BD is an important clinical predictor of The onset of mood disorders in a patient and that the risk of mood disorder decreases as the genetic distance from the proband increases (Craddock & Sklar, 2013 ; Merikangas & Yu, 2002 ). A large family-based Swedish study showed the risk of BD was as much as 7.9, 3.3, and 1.6 times higher for first-, second-, and third-degree relatives of BD probands, respectively, than those without a proband in their family (Song et al., 2015 ). In the largest family study to date in the Swedish cohort with over 2 million individuals, the transmission of BD was found to be statistically homogenous across family type (intact family, families without fathers, and adoptive families) (Kendler, Ohlsson, Sundquist, & Sundquist, 2020 ). This family-based study also estimated the heritability of BD, which is a measure of the proportion of variation in a given trait attributed to genetic variation, to be 44% (95% CI 36–48%). Estimates of heritability from twin studies, which compare the concordance of disease between monozygotic and dizygotic twins, were between 60% and 90% (Craddock & Sklar, 2013 ; Merikangas & Yu, 2002 ; Smoller & Finn, 2003 ). Furthermore, by comparing estimates from twin studies, the heritability of BD is among the highest of all other psychiatric and behavioral disorders (Bienvenu, Davydow, & Kendler, 2011 ).
It has also been well-established that familial risk of BD correlates with increased familial risk of other psychiatric disorders such as schizophrenia, depression, anxiety, drug abuse, attention-deficit hyperactivity disorder (ADHD), personality disorders, and autism spectrum disorder (ASD) (Craddock & Sklar, 2013 ; Kendler et al., 2020 ; Song et al., 2015 ). Schizophrenia, ASD, and depression have the strongest genetic correlations with BD as identified through family studies.
Cohort and population studies
Sample sizes of studies have rapidly increased as genetic studies of BD have moved from family-based designs to cohort and population-based designs. With these, computationally-efficient methods, such as linkage disequilibrium score regression (LDSC), have been developed to estimate both heritability and genetic correlation captured by the single nucleotide polymorphisms (SNPs) which are common locations in the genome where variation occurs between individuals and are measured on a genotyping array (Bulik-Sullivan et al., 2015a ).
Using this technique, the largest study of BD estimated an SNP-based heritability ( h 2 SNP ), which is a measure of the proportion of variation in a given trait attributed to the genetic variation captured by a genotyping array, of 18.6% (Mullins et al., 2020 ). Figure 2 compares the latest estimates of the twin-based heritability and h 2 SNP for BD to a range of psychiatric, behavioral, and neurological disorders (Bienvenu et al., 2011 ; Browne, Gair, Scharf, & Grice, 2014 ; Cross-Disorder Group of the Psychiatric Genomics Consortium, 2019 ; Demontis et al., 2019a ; Faraone & Larsson, 2019 ; Gatz et al., 2006 ; Jansen et al., 2019 ; Purves et al., 2020 ; Walters et al., 2018 ; Zilhão et al., 2017 ). BD has the greatest twin-based heritability estimate and, similar to other traits, also has a substantial proportion not captured by common variations.

Estimated heritability of psychiatric, behavioral, and neurological disorders. Mean twin-based and SNP-based (on liability scale) heritability for different psychiatric (BIP, bipolar disorder; SCZ, schizophrenia; ADHD, attention-deficit/hyperactivity disorder; MD, major depression; ANX, generalized anxiety disorder), behavioral (AN, anorexia nervosa; AUD, alcohol use disorder; CUD, cannabis use disorder), or neurological (ASD, autism spectrum disorder; AD, Alzheimer's disorder; OCD, obsessive-compulsive disorder; TS, Tourette's syndrome) disorders. Error bars are shown for SNP-based estimates from LDSC.
New methodologies have also allowed the genetic correlation between different traits to be estimated using the summary statistics from overlapping sets of common SNPs in different samples. A recent cross-disorder analysis of eight different psychiatric and neurological disorders found that BD is most strongly genetically correlated with schizophrenia ( r g = 0.70) but also shares substantial genetic overlap with major depression ( r g = 0.36), obsessive-compulsive disorder (OCD; r g = 0.31), anorexia nervosa ( r g = 0.21), ADHD ( r g = 0.14), and ASD ( r g = 0.14) (Cross-Disorder Group of the Psychiatric Genomics Consortium, 2019 ). Furthermore, the high comorbidity of substance use in BD has been linked to substantial genetic correlation with substance use: cigarette use ( r g = 0.1), cannabis use ( r g = 0.27), and alcohol use disorder ( r g = 0.30) (Jang et al., 2020 ; Kranzler et al., 2019 ).
Molecular genetic epidemiology
Common variants.
Genome-wide association studies (GWASs) have been the most successful strategy for identifying specific genetic variants associated with BD. Unlike linkage studies, a GWAS can be performed on a set of unrelated cases and controls by testing for the association of genetic variants across the genome with a trait. The first GWAS of BD in 2007 included 2000 cases and 3000 controls and only identified one independent association signal at p < 5 × 10 –7 (Wellcome Trust Case Control Consortium, 2007 ). This association was below the now established p < 5 × 10 –8 threshold for genome-wide significance and further, this association was not supported in an expanded reference group analysis nor in an independent replication. Since then, dozens of additional GWASs of case–control cohorts, consisting of samples from mostly European ancestries, have been published ( Table 1 ). Until the sample size of the GWASs increased to over 10 K, these GWASs identified very few genome-wide significant loci. The most recent GWAS was based on a meta-analysis of 52 case–control cohorts and five large population-based cohorts and included over 40 K cases and 350 K controls identified 64 independent loci across the genome associated with BD (Mullins et al., 2020 ). Thirty-three of these loci were novel for BD, including the region containing the major histocompatibility complex which is also strongly associated with schizophrenia (Mullins et al., 2020 ; Schizophrenia Working Group of the Psychiatric Genomics Consortium, 2014 ). Furthermore, additional novel loci identified in this GWAS are also reported as risk loci for schizophrenia, depression, childhood-onset psychiatric disorders, and problematic alcohol use, highlighting shared underlying genetic architecture between BD and these other psychiatric disorders (Mullins et al., 2020 ). Still though, the SNPs in this large study only explain 15–18% of the variance in the trait (Mullins et al., 2020 ).
Summary of bipolar disorder GWAS
| Reference | Discovery sample ( case + control) | Replication sample ( case + control) | Number of genome-wide significant loci | Ethnicity |
|---|---|---|---|---|
| (Wellcome Trust Case Control Consortium, ) | 2000 + 3000 | – | 0 | European |
| (Baum et al., ; Ferreira et al., ) | 461 + 562 | 772 + 876 | 1 | European |
| (Ferreira et al., ) | 4387 + 6209 | – | 1 | European |
| (Sklar et al., ) | 1461 + 2008 | 365 + 351 | 0 | European |
| (Hattori et al., ) | 107 + 107 | 395 + 409 | 0 | Japanese |
| (Scott et al., ) | 2076 + 1676 | 1868 + 12 831 | 0 | European |
| (Smith et al., ) | 1001 + 1033 | – | 0 | European |
| 345 + 670 | – | 0 | African-American | |
| (Djurovic et al., ) | 194 + 336 | 435 + 10 258 | 0 | European |
| (Cichon et al., ) | 682 + 1300 | 1729 + 2313 6030 + 31 749 | 1 | European |
| (Kerner, Lambert, &Muthén, ) | 1000 + 1034 | – | 2 | European |
| (Psychiatric GWAS Consortium Bipolar Disorder Working Group, ) – PGC1 BD | 7481 + 9250 | 4496 + 42 422 | 2 | European |
| (Smith et al., ) | 1190 + 401 | 2191 + 1434 | 0 | European |
| (Yosifova et al., ) | 188 + 376 | 122 + 328 | 0 | European |
| (Bergen et al., ) | 836 + 2093 | – | 0 | European |
| (Chen et al., ) | 6658 + 8187 | 1115 + 2728 | 5 | European + Taiwanese |
| (Lee, Woo, Greenwood, Kripke, & Kelsoe, ) | 2191 + 1434 | – | 0 | European |
| (Kuo et al., ) | 200 + 200 | 351 + 341 | 0 | Taiwanese |
| (Mühleisen et al., ) | 9747 + 14 278 | – | 5 | European |
| (Xu et al., ) | 950 + 950 | – | 0 | European |
| (Hou et al., ) | 7647 + 27 303 | 2137 + 3168 | 6 | European |
| (Kao et al., ) | 189 + 1773 | 283 + 500 | 0 | Taiwanese |
| (Charney et al., ) | 13 902 + 19 279 | – | 8 | European |
| (Ikeda et al., ) | 2964 + 61 887 | – | 1 | Japanese |
| 10 445 + 71 137 | – | 5 | Japanese + European | |
| (Fiorica & Wheeler, ) | 359 + 686 | – | 0 | African-American |
| (Stahl et al., ) – PGC2 BD | 20 352 + 31 358 | 9412 + 137 760 | 30 | European |
| (Mullins et al., ) – PGC3 BD | 41 917 + 371 549 | – | 64 | European |
Furthermore, as shown in Fig. 3 , univariate causal mixture modeling suggests that we can expect to see substantial increases in identified genome-wide significant loci and consequently in the proportion of h 2 SNP explained by these variants as GWAS sample sizes increase (Holland et al., 2020 ). This is particularly relevant for BD where GWAS studies have now reached the ‘inflection’ point where the significant associations begin to accumulate with smaller increases in sample size (Mullins et al., 2020 ). As such, international collaborations in large-scale GWAS remain imperative for the continued identification of common variants underlying BD etiology, and the plan of the PGC Bipolar Working Group to further increase GWAS sample sizes is encouraging (Sullivan et al., 2018 ).

Statistical power calculations for current and future GWAS. The variance explained by genome-wide significant variants ( y -axis) is calculated for increasing GWAS sample sizes ( x -axis) using the univariate causal mixture model (Holland et al., 2020 ). The legend describes the estimated GWAS sample sizes (SE) needed to capture 50% of the genetic variance (horizontal dashed line) associated with each trait. Stars indicate the sample sizes of currently available GWAS, and circles indicate the estimated sample sizes needed to capture 50% of the genetic variance for each trait. Traits include attention-deficit/hyperactivity disorder (ADHD) (Demontis et al., 2019b ), autism spectrum disorder (ASD) (Grove et al., 2019 ), bipolar disorder (BD) (Mullins et al., 2020 ), depression (MDD) (Howard et al., 2019 ), and schizophrenia (SCZ) (Pardiñas et al., 2018 ). Height is included as a somatic control (no genetic correlation exists between height and bipolar disorder) (Yengo et al., 2018 ). s.e. , standard error.
Genetic overlap
In addition to genetic correlation (Bulik-Sullivan et al., 2015b ) (described above), the most common approach for assessing genetic overlap at the genome-wide level is polygenic risk score (PRS) analysis (International Schizophrenia Consortium et al., 2009 ). The PRS for a given trait is typically a weighted sum of genetic variants where the variants used and their weights are defined by effects measured by previous GWASs of the trait. The genetic liability for BD has been used to predict a number of other psychiatric disorders as well as creativity, educational attainment (Mistry, Harrison, Smith, Escott-Price, & Zammit, 2018 ), addiction (Reginsson et al., 2018 ), as well as psychopathology (Mistry, Escott-Price, Florio, Smith, & Zammit, 2019a ), cognitive functioning (Mistry, Escott-Price, Florio, Smith, & Zammit, 2019b ), progression of unipolar to bipolar depression, and depression onset (Musliner et al., 2019 , 2020 ).
PRSs for BD and other traits have also been used to explain common comorbidities within BD. Suicide attempts by people with BD have been associated with higher genetic liability for depression (Mullins et al., 2019 ) as well as an interaction between trauma and bipolar genetic liability (Wilcox et al., 2017 ). Previous childhood ADHD diagnosis in those with BD was associated with higher genetic liability for ADHD (Grigoroiu-Serbanescu et al., 2020 ; Wilcox et al., 2017 ).
In addition to PRS analysis, cross-disorder GWAS meta-analyses have also been performed for BD and ADHD (Bipolar Disorder and Schizophrenia Working Group of the Psychiatric Genomics Consortium, 2018 ; van Hulzen et al., 2017 ), SCZ (Bipolar Disorder and Schizophrenia Working Group of the Psychiatric Genomics Consortium, 2018 ), and MDD (Coleman, Gaspar, Bryois, & Breen, 2020 ), identifying two, 114, and 73 genome-wide significant loci associated with these phenotype pairs, respectively. Moreover, numerous genes mapped to BD risk loci are also linked to schizophrenia, ASD, and OCD (O'Connell, McGregor, Lochner, Emsley, & Warnich, 2018 ) further highlighting common genetic architecture across psychiatric disorders.
The conditional/conjunctional false discovery rate statistical tool has also been used to identify BD risk loci as well as shared risk loci between BD and a number of phenotypes. This method leverages the power of two GWAS to boost discovery by re-adjusting the GWAS test statistics in a primary phenotype and allows for the discovery of loci significantly associated with two phenotypes simultaneously (Andreassen, Thompson, & Dale, 2014 ; Smeland et al., 2020b ). Utilizing this method, shared genetic loci have been identified between BD and ADHD ( n = 5) (O'Connell et al., 2019 ), schizophrenia ( n = 14) (Andreassen et al., 2013 ), Alzheimer's disease ( n = 2) (Drange et al., 2019 ), intelligence ( n = 12) (Smeland et al., 2020a ), body mass index ( n = 17) (Bahrami et al., 2020 ), and lifespan ( n = 8) (Muntané et al., 2021 ). This method is agnostic to the effect directions of genetic variants and so shared loci were identified between BD and Alzheimer's disease, intelligence, body mass index, and lifespan despite observed null and non-significant genetic correlations with these phenotypes.
Most recently (Mullins et al., 2020 ), the genetic relationship between BD and 10 clinically and epidemiologically associated traits (daytime sleepiness, morningness, sleep duration, insomnia, mood instability, educational attainment, problematic alcohol use, drinks per week, smoking initiation, and cigarettes per day) were assessed using the MiXeR tool (Frei et al., 2019 ), to identify trait-specific and shared genetic components, and Mendelian randomization (Zhu et al., 2018 ), to establish ‘causal’ relationships. Extensive genetic overlap was identified between all traits and BD, most notably that >90% of the genetic variants estimated to influence BD were also estimated to influence educational attainment. Moreover, bidirectional relationships were identified between BD and sleep duration, mood instability, educational attainment, and problematic alcohol use, while BD was identified as ‘causal’ for morningness and drinks per week and smoking initiation was ‘causal’ for BD (Mullins et al., 2020 ).

Rare variants
In addition to genetic interactions, the difference in heritability could also be explained by rare variants in the genome which are often unmeasured and thus not included in GWASs. While the cost of whole-exome sequencing (WES) and whole-genome sequencing (WGS) has decreased, these technologies are still substantially more expensive than common genotyping arrays. As a result, WGS/WES studies of BD have been limited to small studies consisting mostly of large pedigrees to potentially enrich the sample with causal rare variants and increase power (Forstner et al., 2020 ; Goes et al., 2016 , 2019 ; Maaser et al., 2018 ; Sul et al., 2020 ; Toma et al., 2018 ). While these studies have found evidence of higher rare deleterious burden in cases (Sul et al., 2020 ), higher disruptive variant burden in early-onset cases (Toma et al., 2018 ), evidence of rare variant segregation in pedigrees (Forstner et al., 2020 ; Goes et al., 2016 ; Maaser et al., 2018 ), and evidence of de novo variation (Goes et al., 2019 ), much larger sample sizes will be required to definitively identify rare variants conferring risk for BD.
Copy number variants
Copy number variants (CNVs) refer to regions of the genome where a duplication (three or more copies are present) or deletion (only one copy remains) has occurred such that more or less than the expected two copies in the diploid human genome are present. Carriers of certain CNVs are known to be at considerably elevated risk for developing neurodevelopmental (e.g. ASDs) and mental disorders (e.g. schizophrenia) (Kirov, Rees, & Walters, 2015 ) as well as somatic conditions (e.g. diabetes and hypertension) (Crawford et al., 2019 ). The frequency of CNVs in BD is less than that observed for neurodevelopmental disorders or schizophrenia (Kirov, 2015 ), and correspondingly their role in the disorder appears less with only one CNV robustly associated with BD to date. A 650 kb duplication at 16p11.2 was first described as a de novo CNV for BD (Kirov, 2015 ; Malhotra et al., 2011 ) and this association was replicated in a larger genome-wide analysis (Green et al., 2016 ). This CNV is also implicated in schizophrenia, autism, and intellectual disability (Kirov, 2015 ). Two additional CNVs, at 1q21.1 and 3q29, are also implicated in BD; however, these associations fail to pass the genome-wide significance threshold (Green et al., 2016 ). Interestingly, these two CNVs are also associated with schizophrenia (Kirov, 2015 ). One further study identified enrichment of genic CNVs in schizoaffective BD, but not between BD cases and controls or other BD subtypes (Charney et al., 2019 ).
These findings highlight that the genetic overlap between BD and schizophrenia extends beyond common variation, but suggests a difference in underlying mechanisms. One possible explanation for the smaller role of CNVs in BD is that patients with BD exhibit less cognitive deficits than patients with schizophrenia who can exhibit substantial cognitive deficits, since the same CNVs which are implicated in schizophrenia are also known to cause cognitive problems (Kirov, 2015 ).
Genetic interactions
Other than increasing the sample size of GWAS, the difference between observed twin-based and h 2 SNP ( Fig. 2 ) may also be explained by unaccounted for moderated genetic effects such as interactions between genes and the environment (G×E) or gene–gene interactions (epistatic effects). The role of G×E in BD remains an under-researched area, however, but some interactions have been identified (Aas et al., 2014 , 2020 ; Hosang, Fisher, Cohen-Woods, McGuffin, & Farmer, 2017 ; Oliveira et al., 2016 ; Winham et al., 2014 ). Although these studies highlight the potential role of G×E in the etiology of BD, the lack of replication studies and small sample sizes suggest that they should be interpreted with caution. As with G×E, studies of epistasis in BD are in their infancy and lack replication (Judy et al., 2013 ). As the ability of GWAS to identify risk variants with small effects increases, further study of how implicated genes interact with environmental or other genetic factors to modulate the risk of BD are required.
Clinical implications
Pharmacogenomics.
Lithium, anti-epileptic drug mood stabilizers (such as valproate/divalproex, lamotrigine, and carbamazepine), antipsychotics, and antidepressants are commonly prescribed treatments for BD. However, response to these medications can widely vary between individuals, and some patients may cycle through different medications before they find an effective treatment with minimal side effects. Pharmacogenomic studies aim to use genetics to predict treatment response. A particular challenge to pharmacogenomics in BD has been the measurement of treatment response which can be limited by the length of follow-up, adherence to medication, and confounding due to the multi-drug treatment strategy common to the illness. Consequently, a systematic rating system with a high inter-rater reliability, the Alda score, was developed to quantify the clinical improvement of BD during treatment while also accounting for potential confounders of treatment response (Nunes, Trappenberg, & Alda, 2020 ). However, obtaining large samples with reliable measures has limited the statistical power to discover clinically-informative genetic variants associated with treatment response. Furthermore, heterogeneity between study designs and the samples included have yielded limited replication of any findings. While not yet replicable, promising pharmacogenomic findings for BD were summarized in a recent review (Gordovez & McMahon, 2020 ). Most of the previous pharmacogenomic studies have been focused on either lithium treatment response or HLA haplotypes predicting serious adverse reactions related to carbamazepine, phenytoin, and lamotrigine. A recent study tested for genetic association with treatment response to anti-epileptic drug mood stabilizers, an alternative to lithium, and identified two SNP-level associations in THSD7A and SLC35F3 as well as two gene-level associations with ABCC1 and DISP1 (Ho et al., 2020 ).
With the exception of genetic predictors of adverse reactions to medication, no large genetic effects on treatment response have been identified. However, current pharmacogenomic testing has already been shown to be useful by providing clinicians support in reaching effective and well-tolerated treatments of BD (Ielmini et al., 2018 ). Additionally, as the sample size of pharmacogenomic studies increases, PRSs derived from these studies could further enable a precision medicine approach to BD treatment. In addition to pharmacogenomic PRSs, PRSs derived from large case–control studies could also improve the genetic prediction of treatment response. For example, increased genetic liability for depression and schizophrenia was associated with worse response to lithium (Amare et al., 2020 ; International Consortium on Lithium Genetics (ConLi+Gen) et al., 2018 ). These PRSs could be explaining some of the clinical heterogeneity in the sample as discussed below and thus improve the identification of certain BD clinical profiles that respond best to lithium.
Finally, there is potential application of repurposing drugs and focusing on different drug targets based on recent genetic findings. For example, calcium channel blockers (CBBs), which have been widely used to treat hypertension and other cardiovascular conditions, were also once considered as a treatment in psychiatry (Harrison, Tunbridge, Dolphin, & Hall, 2020 ). However, because CACNA1C has now been implicated as one of the strongest associations with BD (Gordovez & McMahon, 2020 ), there is renewed interest in CBBs as a treatment for the disorder (Cipriani et al., 2016 ).
Risk prediction
In addition to therapeutic intervention, PRSs may also provide clinical utility to inform disease screening (Torkamani, Wineinger, & Topol, 2018 ). While the PRS derived from the latest GWAS of BD only explains about 4.75% of the phenotypic variance, the latest PRS could still be useful for risk stratification (Mullins et al., 2020 ). Compared to individuals with average genetic risk for BD, individuals in the top decile risk had an odds ratio of 3.62 (95% CI 1.7–7.9) of being a case. An important caveat to note about PRSs, however, is that prediction performance is worse when applied to ancestries not included in the training GWAS (Martin et al., 2019 ). For instance, the current BD PRS, estimated using individuals with European ancestries, explains only around 2% and 1% of the phenotypic variance in individuals with East Asian or admixed African American ancestry, respectively (Mullins et al., 2020 ). Encouragingly though, the trans-ethnic prediction accuracy of the PRS has improved as the sample size has increased. Furthermore, the PRS prediction accuracy will also improve as new non-European ancestries are included in future training GWASs.
Clinical heterogeneity
PRSs can also help dissect the high clinical heterogeneity (i.e. bipolar type, psychosis, rapid cycling) present in the disorder (Coombes et al., n.d.). For example, higher genetic liability for schizophrenia is associated with bipolar type I (Charney et al., 2017 ). This finding could be driven by the increased prevalence of psychosis among those with BDI as multiple studies have shown that higher genetic risk of schizophrenia is associated with psychosis in BD, particularly during mania (Allardyce et al., 2018 ; Bipolar Disorder and Schizophrenia Working Group of the Psychiatric Genomics Consortium, 2018 ; Charney et al., 2019 ; Coombes et al., 2020 ; Markota et al., 2018 ). Other studies of bipolar subtypes have shown positive associations between BDII and insomnia PRS, rapid cycling and ADHD PRS, as well as early age-of-onset of BD and PRSs for risk-taking and anhedonia (Coombes et al., n.d.; Lewis et al., 2019 ). While no individual PRS is able to explain a large amount of variation among bipolar subtypes, these findings give insight into the genetic contributions to clinical heterogeneity and could help classify the disorder more accurately as well as identify the risk of suicide, psychosis, and other adverse outcomes in patients with BD.
Future directions
Significant advances in our understanding of the genetic architecture of BD have been made, from initial linkage and family studies to current large consortia-driven genome-wide studies. Moreover, integration of these genetic discoveries with other -omic and imaging data will be key to comprehending the role of genetic variation in the etiology of BD. However, distinct shortcomings and limitations to genetic discovery highlight key areas to be prioritized in future studies.
Diverse phenotype ascertainment
Identification of novel loci for BD, and other polygenic complex phenotypes, requires increasing sample sizes ( Fig. 3 ), which remains a challenging and costly task (Lu, Campeau, & Lee, 2014 ). The majority of samples included in the PGC-BD were clinically ascertained, with the inclusion of external biobank samples only in the most recent discovery GWAS (Mullins et al., 2020 ). Numerous efforts have been made to combine electronic health record and registry data with genetic data to facilitate large population-based studies, such as the Electronic Medical Records and Genomics network ( https://emerge-network.org/ ), the UK Biobank ( https://www.ukbiobank.ac.uk/ ), All of Us ( https://allofus.nih.gov/ ), the Million Veterans Program ( https://www.research.va.gov/mvp/ ), and iPsych ( https://ipsych.dk/en/ ). Furthermore, GWAS summary statistics of self-reported phenotypes for thousands to millions of individuals may be obtained through collaboration with the personal genetics company 23andMe, Inc. ( https://research.23andme.com/research-innovation-collaborations/ ). The data generated by such population studies and 23andMe provide a means by which to drastically increase sample size without the costs associated with clinical ascertainment. This approach was shown to be successful for depression, where PGC cohorts were meta-analyzed with data from the UK Biobank and summary statistics from 23andMe, increasing the number of identified associated risk loci from 44 (Wray et al., 2018 ) to 102 (Howard et al., 2019 ). However, a limitation to this use of ‘minimal phenotyping’ data is that the loci identified, especially when based on self-report data, were non-specific for depression highlighting potential differences in genetic architecture when compared to clinically ascertained depression (Cai et al., 2020 ). In line with this, the h 2 SNP estimates of the biobank samples included in the latest PGC BD GWAS are less than that observed for clinically ascertained samples which may reflect more heterogeneous clinical presentations or less severe illness (Mullins et al., 2020 ).
Data generated from ‘minimal phenotyping’ are likely to include other psychopathological features which may underlie self-reported BD such as personality disorders or mild temperamental traits, thereby increasing heterogeneity in the sample and leading to the possibility of non-specific or false-positive results. However, true self-reported BD may reflect the non-hospitalized, non-psychotic part of the BD spectrum, more typical of BDII, which is under-represented in the current PGC BD sample. Moreover, expanding genetic studies to include the full spectrum of BD in population-based non-clinical samples increases the potential for novel discoveries with important implications for clinical management and further research, and is therefore of high interest to both clinicians and the pharmaceutical industry.
Thus, while adopting the ‘minimal phenotyping’ approach for BD will allow GWAS to reach sample sizes not currently feasible by clinical ascertainment and will likely identify numerous novel risk loci, similar post-hoc analyses as that performed for depression (Cai et al., 2020 ), will be required to determine the specificity of identified loci to BD.
Increased deep phenotyping
The high levels of heterogeneity amongst patients with BD, including disorder type, features of episodes, and the course of the disorder, contribute to the difficulty in identifying underlying genetic risk factors. BDI ( h 2 SNP = 25%) is shown to be more heritable than BDII ( h 2 SNP = 11%), and the genetic correlation ( r g = 0.89) between these types suggests that they are closely related, yet distinct, phenotypes (Stahl et al., 2019 ). In support of this, the most recent PGC GWAS for BD identified novel and distinct loci specifically associated with BDI or BDII, which were not identified when all bipolar cases were analyzed together (Mullins et al., 2020 ). Genetic studies of the features and course of BD have predominantly employed a PRS approach, as outlined above, and GWAS data for these subtypes is lacking due to small sample sizes [data from the PGC indicate that none of these subtypes include more than 10 K samples (Bipolar Disorder and Schizophrenia Working Group of the Psychiatric Genomics Consortium, 2018 )]. Thus, larger deeply phenotyped samples are required in order to conduct a thorough investigation of the genetic architecture of these subtypes within BD. Doing so would aid subtype-specific discoveries, and may inform on nosology, diagnostic practices, and drug development for BD.
In addition, the potential inclusion of ‘minimal phenotyping’ data, as described above, further emphasizes the need for increased deep phenotyping. Results generated from deep phenotyped samples will serve as standards against which to compare the specificity of results generated from the inclusion of ‘minimal phenotyping’.
Increased ancestral-diversity
The majority of individuals included in GWASs for any trait have overwhelmingly been of European descent and the lack of diversity is even more pronounced in genetic studies of psychiatric disorders (Martin et al., 2019 ; Peterson et al., 2019 ; Sirugo, Williams, & Tishkoff, 2019 ). In BD, the largest GWAS includes only individuals from European ancestries (Mullins et al., 2020 ). This ‘missing diversity’ can greatly hinder our understanding of the etiology of BD. For example, the inclusion of non-European ancestries could substantially improve fine-mapping of disease-associated loci (Peterson et al., 2019 ). Furthermore, the current Eurocentric approach has the potential to exacerbate health disparities already seen in BD (Akinhanmi et al., 2018 ) by limiting the therapeutic advances gained by pharmacogenomics and improved genetic risk predictions to those of European descent (Duncan et al., 2019 ; Martin et al., 2019 ; Sirugo et al., 2019 ). Future inclusion of diverse samples will come with new ethical, technological, and methodological challenges (Peterson et al., 2019 ). Some of these considerations include choosing ancestry-specific genotyping platforms to improve genomic coverage, increasing sample sizes of diverse reference panels to improve imputation accuracy, and improving statistical methods to control for population stratification and estimate ancestry-specific PRSs. Thus, the PGC Bipolar Working Group has committed to expanding the future GWAS to include non-European ancestries.
Larger sequencing efforts
As mentioned above, sequencing efforts in BD are currently in their infancy (Forstner et al., 2020 ; Goes et al., 2016 ; Maaser et al., 2018 ; Sul et al., 2020 ; Toma et al., 2018 ). Although studies provide evidence that rare variants might contribute to the etiology of BD, weak statistical power due to small sample sizes remains an issue. The Bipolar Sequencing Consortium (BSC) was established to facilitate combining existing exome and WGS studies of BD ( http://metamoodics.org/bsc/consortium/ ), and includes approximately 4500 BD cases and 9000 controls, as well as 1200 affected relatives from 250 families. Moreover, a collaboration between the Dalio Initiative in BD ( https://www.daliophilanthropies.org/initiatives/mental-health-and-wellness/ ), the Stanley Centre ( https://www.broadinstitute.org/stanley ), and iPSYCH ( https://ipsych.dk/en/ ) aims to generate WES data from approximately 7000 BD cases and 10 000 matched controls. However, it is estimated that as many as 25 000 cases might be necessary in order to identify significant rare variant associations with BD (Zuk et al., 2014 ), confirmed by recent analyses in schizophrenia (Singh et al., 2020 ), and so continued expansion of these, or similar, efforts will be crucial to determine the role of rare variation in BD.
Our knowledge of the genetic etiology of BD has rapidly accelerated in recent years with advances in technology and methodology as well as the adoption of international consortiums and large population-based biobanks. It is now clear that BD is highly heritable but also highly heterogeneous and polygenic with substantial genetic overlap with other psychiatric disorders. Encouragingly, genetic studies of BD have reached an ‘inflection point’ ( Fig. 3 ). Thus, the number of associated loci is expected to substantially increase in larger future studies and with it, improved genetic prediction of the disorder. Incorporation of ancestrally-diverse samples in these studies will enable improved identification of causal variants for the disorder and also allow for equitable future clinical applications of both genetic risk prediction and therapeutic interventions.
Acknowledgements
We would like to thank the research participants and members of the Bipolar Disorder Working Group of the Psychiatric Genomics Consortium, and other studies reported in this review, for making this research possible.
Financial support
We acknowledge the support from the Research Council of Norway (229129, 213837, 223273), the South-East Norway Regional Health Authority (2017-112), and the PGC US Norway Collaboration (RCN# 248980).
Conflict of interest
- Get new issue alerts Get alerts
Secondary Logo
Journal logo.
Colleague's E-mail is Invalid
Your message has been successfully sent to your colleague.
Save my selection
Manic episode in patient with bipolar disorder and recent multiple sclerosis diagnosis
A case report.
Editor(s): Saranathan., Maya
a University of Minnesota Medical School
b Department of Psychiatry, University of Minnesota, Minneapolis, MN.
∗Correspondence: Simon Yang, University of Minnesota, 420 Delaware St. SE, Minneapolis MN 55455 (e-mail: [email protected] ).
Abbreviations: BD = bipolar disorder, MS = multiple sclerosis.
How to cite this article: Yang S, Wichser L. Manic episode in patient with bipolar disorder and recent multiple sclerosis diagnosis: a case report. Medicine . 2020;99:42(e22823).
Patient information was de-identified. Received written consent to use patient information as well.
The authors have no conflicts of interest to disclose.
Data sharing not applicable to this article as no datasets were generated or analyzed during the current study.
This is an open access article distributed under the Creative Commons Attribution License 4.0 (CCBY), which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited. http://creativecommons.org/licenses/by/4.0
Introduction/Rationale:
Multiple sclerosis (MS) is associated with a higher prevalence of mood and psychiatric disorders, such as bipolar disorder (BD). While mania is most often associated with BD, MS can also induce manic symptoms. However, it is crucial to distinguish which condition is causing mania since medical management is different based on its etiology. Herein, we report a case of a manic episode in a middle-aged female with a prolonged history of BD who received a recent diagnosis of MS 1 year ago.
Patient Concerns:
A 56-year-old female presented with an episode of mania and psychosis while receiving a phenobarbital taper for chronic lorazepam use. She had a prolonged history of bipolar type 1 disorder and depression. She showed optic neuritis and was diagnosed with MS a year prior.
Diagnoses:
The patient was diagnosed with BD-induced mania based on the absence of increased demyelination compared to previous MRI and lack of new focal or lateralizing neurologic findings of MS.
Interventions:
Lithium was given for mood stabilization and decreased dosage of prior antidepressant medication. Risperidone was given for ongoing delusions.
Outcomes:
After 8 days of hospitalization, patient's mania improved but demonstrated atypical features and ongoing delusions. She was discharged at her request to continue treatment in an outpatient setting.
Conclusion/Lesson:
In BD patients with an episode of mania, MS should be included in the differential, since both conditions can cause manic symptoms. The origin of mania should be delineated through a detailed neurological exam, neuroimaging, and thorough patient-family psychiatric history for appropriate clinical treatment.
1 Introduction
Multiple sclerosis (MS) is an inflammatory autoimmune disease that focally damages the white matter in the brain and spinal cord. [1] It affects 1 in 1000 people and is the most common central nervous system disease for young adults in the Western world. [2] Initially, neurological symptoms are transient due to remyelination, but repeated demyelination progressively leads to diffuse and chronic neurodegeneration. Furthermore, previous studies have shown increased psychiatric symptoms and higher prevalence of psychiatric and mood disorders. [3]
Bipolar disorder (BD) is a mood disorder characterized by extreme mood fluctuations with episodes of mania or hypomania and depression. Mania, a hallmark of BD, is when the patient is in a state of elevated mood and energy, during which the patient reports symptoms such as euphoria or irritable mood, racing thoughts, overactivity, and reduced need for sleep. BD affects more than 1 in 100 people worldwide. [4]
The prevalence of BD in MS patients has been reported to be twice than that of the general population. [5] For patients diagnosed with BD and MS, there is no clear method to distinguish whether mania was induced from BD or from a MS flare-up. However, it is important to discern the cause of manic episode since management is different for BD-induced mania vs MS-induced mania. Herein, we describe a patient diagnosed with BD that later developed MS who presented to us during a manic episode. Through this case, we aim to examine the BD versus MS origins of manic episodes and discuss relevant literature.
2 Case Presentation
The patient was a 56-year-old female who came to us during an episode of mania and psychosis while receiving treatment at an addiction treatment center where she was taking a phenobarbital taper for chronic lorazepam use. She displayed symptoms of aggressive posturing, verbal abuse to staff, delayed response, and racing thoughts. She did not describe suicidal thoughts. She had 4 prior psychiatric hospitalizations. At age 33, she exhibited depression, anxiety, and paranoia that lead to her first hospitalization. At age 44, she attempted suicide via acetaminophen overdose. Her first reported manic episode was at age 45, during which bipolar type 1 disorder was considered as her differential and subsequently diagnosed. Her symptoms accompanied delusions during this episode, without suicidal ideation. Her most recent hospitalization was at age 49 for depression and paranoia with delusions of being wiretapped and people reading her mind. At age 55, the patient presented with optic neuritis and diagnosed with MS after a lumbar puncture showed oligoclonal bands.
Family history revealed depression in father and alcohol use disorder in mother. Past medical history described an acute onset dizziness when moving eyes left to right or vice versa and when standing up from a lying position.
Neurology consult found no focal or lateralizing findings. MRI analysis showed greater than 15 foci of T2 hyperintensity within white matter where some lesions were within periventricular and juxtacortical white matter of both cerebral hemispheres, consistent with a demyelinating disease. A single focus of enhancement in the posterior corona radiata was suggestive of active demyelination. No demyelinating signs were seen in the thoracic spine. However, no significant difference was seen compared to previous MRI.
During the present hospitalization, patient's prior bupropion was reduced due to concern for further mania activation. Lithium 600 mg twice a day was prescribed for mood stabilization. Risperidone 0.5 mg at bedtime was prescribed for ongoing delusions. Patient was not taking scheduled steroids prior to admission. After 8 days of hospitalization, patient's mania improved but demonstrated atypical features, such as absence of pressured speech, grandiosity, risk taking or sleep pattern changes. Per a family member's report, patient stated that she was in a movie and that everyone else was acting around her. Patient requested discharge to continue treatment in an outpatient setting.
3 Discussion
Although neurological symptoms of MS have been extensively studied, the psychiatric effects of MS are relatively less elucidated, despite the fact that the association of MS and psychiatric symptoms observed as early as 1872 by Jean-Martin Charcot. [6] In 1986, Schiffer et al suggested an association between BD and MS after identifying 10 patients with both BD and MS, out of more than 700,000 individuals, when epidemiologic data expected to find only 5.4 patients. [7] Co-occurrences of BD and MS have been reported infrequently through case studies. Recently, Carta et al conducted a case control study with 201 MS patients that examined the risk of BD in MS patients and reported OR of 44.4 for bipolar spectrum disorders. Specifically, bipolar type 2 diagnoses (7.5%) was more frequent than bipolar type 1 diagnoses (0.99%). [8]
The exact underlying mechanism and pathophysiology of BD and MS co-presentation is yet to be established. It is unknown whether BD is an early manifestation of MS or if both diseases share a common underlying cause presenting at similar timelines. More recent studies have shown genetic associations between BD and MS in human leukocyte antigen (HLA) DR2 gene and mitochondrial transcriptomes. [9,10] Further understanding of the etiology of this association may elucidate whether there are synergistic effects or crosstalk between MS and BD therapeutics.
While mania is a hallmark symptom of BD, MS can also exhibit a range of psychiatric symptoms including mania, euphoria, depression, hallucinations, and episodes of pathologic laughing and weeping, which is coined as ‘pseudobulbar effects.’ [11] Focal neuronal demyelination in MS patients may interfere with communication between frontal lobe brain regions responsible for emotion and manifests as emotional lability and exaggerated emotions, common symptoms in a manic or depressive episode. [12] Features of MS flare-up mania are no different than those of non-MS mania. However, the incidence of psychosis has been reported to be less common in MS. [13]
Differentiating the cause of the manic episode is of clinical significance as the treatment plan differs between a MS flare-up and a BD manic episode. For instance, while lithium and sodium valproate have been shown to be effective in treating mania in BD, no controlled trials of its efficacy in mania in MS patient has been published. [14] Additionally, manic episodes due to medications cannot be precluded. Steroid treatment in MS patient may often cause a moderate degree of mania. [15] Patients with a family history of alcohol use disorder or other affective disorder are more vulnerable to this cause. [15] Other medications, such as tizanidine, baclofen, and dantrolene, can also cause hypomania following their use. [16] Manic symptoms due to medications are often dose-dependent and manifest soon after initiating the medication. [16]
Detailed neurologic tests or neuroimaging can often help differentiate the cause of a manic episode. MS flare-ups often manifest with increased focal neurological symptoms including visual loss, fatigue, urinary incontinence, and cognitive impairment, in addition to any of the afore-mentioned mood symptoms. Additionally, MS flare-ups may show an increased degree of demyelination on MRI compared to prior images.
Both MS and BD-onset mania have been reported to show white matter changes on MRI by Young et al. [17,18] Especially, MS patients with mania and psychotic symptoms were shown to have plaques located in the bilateral temporal horn areas. [14] Neuroimaging of BD patients without MS has been more complex. Several studies proposed increased white matter and periventricular hyperintensities in these patients. [19,20] McDonald et al reported increased subcortical hyperintensities in T2 weighted MRI in late-onset BD patients. [19,21] Dupont et al reported increased white matter hyperintensities in early-onset BD patients. [19,22] Altshuler et al reported no significant difference white matter hyperintensities, but increased periventricular hyperintensity in BD type 1 patients. [19,23]
In our case, the absence of aforementioned focal or lateralizing finding in MS during the neurological exam, absence of increased demyelination compared to previous MRI, and family history of psychiatric disorders decreased the likelihood of her current symptoms representing a MS flare-up and was more consistent with BD-induced mania. Additionally, patient was not taking mania-inducing medications such as steroids, tizanidine, baclofen, or dantrolene. Patient's symptoms improved with lithium treatment. The patient's MRI showed increased white matter and periventricular T2 hyperintensity. However, no plaques at bilateral temporal horn areas were identified. Considering that her symptom onset was during a phenobarbital taper for chronic benzodiazepine use, her mania may have been a BD manic episode triggered by her benzodiazepine withdrawal directly or exacerbated from withdrawal symptoms, such as poor sleep and increased anxiety.
The ages at which this patient's illnesses presented - BD type I onset at age 45 preceding MS onset at age 55, is of particular note in relation to previous case reports. Marangoni et al identified case reports of 26 patients who had BD onset clearly preceding MS, via a PubMed search from inception to 2014. [24] The study showed an average of 5 years difference between BD and MS onset. The majority of these patients were found to have BD type I, where 25 patients had BD type I and 1 patient had BD type II with rapid cycling. Three cases reported family history of MS and 6 cases reported psychiatric family history. The study also noticed increased white matter lesions in periventricular and subcortical white matter – which was consistent with our case - as well as in the centrum semiovale, frontal, parietal, and temporal lobes. However, it did not identify association between certain BD type to MS types nor association between certain BD types with patterns of white matter lesions.
While the study had insufficient data to formulate a valid hypothesis, the study found that BD-preceded-MS had a higher age of both BD and MS onset compared to the age of onset of the combined pool of patients with BD and MS regardless of onset order. The study also suggested that later onset of MS may be associated with co-occurrence with BD. This case report, where the patient was diagnosed with BD and MS relatively later than the common age of onset of 20s or 30s, substantiates these trends found in previous case reports by Marangoni et al and speculates that late onset of BD or MS may be associated with BD-MS comorbidity. Past reports showed cases where acute psychotic symptoms led to MS diagnosis, which were coined as “inaugural manifestations” to MS. [25] Future research into the timing of onset can elucidate whether late diagnosis of mood or psychotic disorders can be early signs of comorbidity with MS.
4 Conclusion
In patients with co-occurrence of BD and MS, there is currently no clear guideline to discern the origin of manic episodes. However, it is important to attempt to discern the predominant cause of the manic episode through detailed patient history, neurologic exam, and neuroimaging, as it can affect treatment plans. Additionally, the presented case, along with previous cases of BD-preceding-MS correlating with generally later age of onset of BD and MS, may be a future direction for further investigation.
Author contributions
Conceptualization: Simon Yang.
Supervision: Lora Wichser.
Writing – original draft: Simon Yang.
Writing – review & editing: Simon Yang, Lora Wichser.
- Cited Here |
- PubMed | CrossRef |
- Google Scholar
- View Full Text | PubMed | CrossRef |
bipolar disease; mania; multiple sclerosis; neuroimaging; mood disorder
- + Favorites
- View in Gallery
CASE REPORT article
Retrospective case study: ketogenic metabolic therapy in the effective management of treatment-resistant depressive symptoms in bipolar disorder.

- Family Renewal, Inc. DBA Mental Health Keto, Vancouver, WA, United States
This retrospective case study assessed Ketogenic Metabolic Therapy’s (KMT) efficacy in a bipolar disorder patient with treatment-resistant depressive symptoms insufficiently controlled by weekly ketamine treatments. Monitoring included relevant biomarkers of ketone production and macronutrient levels, alongside mood evaluations through the Generalized Anxiety Disorder-7 (GAD-7), Depression Anxiety Stress Scales (DASS), and PTSD Checklist for DSM-5 (PCL-5), showing mood stabilization and improved functionality. Qualitative analysis revealed sub-stantial enhancements in functioning, life quality, and mental well-being. This study enriches the metabolic psychiatry literature, emphasizing KMT’s potential benefits by integrating quantitative data from recognized psychiatric assessment tools and qualitative insights.
1 Introduction
Bipolar II disorder is marked by significant emotional and psychological distress, characterized by periods of depressive episodes and hypomania ( 1 ). This condition not only affects an individual’s psychological well-being but also has profound implications on their social and occupational functioning ( 2 ). The complexity of Bipolar II disorder, especially with treatment-resistant depressive symptoms, presents a substantial challenge in psychiatric care ( 3 ). Current treatments for Bipolar II disorder often include a combination of mood stabilizers, antidepressants, and psychotherapy. However, a notable subset of patients remains resistant to these interventions, experiencing persistent symptoms and a diminished quality of life. Even individuals with bipolar disorder undergoing treatment still spend about 19% of their time in depressive states and an additional 18% in sub-syndromal depressive states ( 4 ). This resistance underscores the urgent need for alternative strategies that can offer relief and improve patient outcomes ( 5 ).
Emerging evidence suggests that metabolic interventions, such as Ketogenic Metabolic Therapy (KMT), also known as the ketogenic diet, may offer favorable treatment outcomes for individuals with psychiatric disorders. Well established in the management of epilepsy ( 6 ), recent studies indicate that the ketogenic diet may have beneficial outcomes for individuals with bipolar disorder, with observations from case studies ( 7 – 9 ) and pilot studies ( 10 – 12 ) reporting notable improvements in symptoms. The diet’s mechanism is believed to involve the modulation of brain energy metabolism and neurotransmitter levels ( 13 – 16 ), providing a compelling rationale for its application in Bipolar II disorder.
This case focuses on an individual diagnosed with Bipolar II disorder, presenting with persistent depressive episodes marked by significant lethargy, low mood, and difficulty in managing daily activities despite standard treatment protocols. By employing both quantitative and qualitative methods, this case study seeks to understand better the treatment potential of KMT with patients for whom standard care has not yielded satisfactory outcomes.
2 Case presentation
2.1 clinical background.
In this case, a 53-year-old female with Bipolar II reported persistent mood instability and depressive episodes resistant to past and current conventional treatments. Psychiatric intervention at time of diet implementation consisted of weekly ketamine treatments for temporary symptom relief. Despite this intervention, the relief from depressive symptoms was short-lived, lasting only 1 to 3 days before the symptoms returned. The patient also experienced migraine headaches. Prior attempts at management included medication, psychotherapy, a Mediterranean diet, physical exercise, and consistent sleep schedules, which yielded limited improvement. Given the limited efficacy of standard treatments and the transient benefits achieved with ketamine therapy, she was open to exploring KMT as a novel intervention. Her history of psychiatric conditions began in childhood and adolescence, leading to subsequent diagnoses of Generalized Anxiety Disorder and Major Depressive Disorder before the eventual identification of Bipolar II as the most recent diagnosis. At the initiation of treatment, the participant was receiving medical care for additional chronic conditions, which included Immune Thrombocytopenia, Migraines, Hypothyroidism, and recurrent shingles (Herpes Zoster).
2.2 Ketogenic metabolic therapy intervention strategy
Macronutrient tracking was initiated using Cronometer, which identified an average baseline carbohydrate consumption of between 200 and 300 g per day. BMI was in a healthy range at diet commencement and remained so throughout treatment. Virtual meetings for KMT support were scheduled twice weekly for 30-min intervals over 3 months and then moved to weekly. Carbohydrate consumption was systematically reduced over 2 weeks to achieve a 30 g total intake per day. Macronutrient ratios were initially set at a 1:1 ratio and later adjusted to a 1.5:1 ratio (154 g Fat, 72 g Protein, 30 g Total Carbohydrates) to increase ketone production. Total carbohydrate measurement was chosen over net to initiate and maintain ketosis at consistent levels. Both ratios used are generally considered Modified-Atkins (MAD). The diet consisted primarily of beef, pork, chicken, eggs, dairy, and salmon, with primary fat sources being MCT oil, avocado oil, and butter. Low-carbohydrate vegetables and minimal amounts of low-carb berries complemented this.
Supplementation provided included a non-methylated B-complex, trace minerals (providing zinc, copper, manganese, chromium, molybdenum, boron, and vandyl sulfate), vitamin D, and electrolytes in the form of sodium, magnesium, and potassium. Testing compliance was 89% complete for daily ketone measures and 91% complete for daily glucose measures over the 21-week period. Blood glucose and BHB level tracking was initiated and showed nutritional ketosis was achieved at 1.0 mmol/L ( Figure 1 ). Approximately 3.5 weeks into the process of carbohydrate restriction, lab work was received showing free carnitine at 16 μmol/L that identified hypocarnitinemia ( 17 ), prompting ongoing L-carnitine supplementation of 3,000 mg in divided doses daily.
Figure 1 . Line graph showing average glucose and ketone levels in mmol/L over 21 weeks.
3 Evaluation of intervention outcomes
3.1 quantitative analysis.
Mood assessments were collected at baseline, one-month, four-month, and five-month intervals. They were selected for their validity in assessing self-reported markers of mood, anxiety, stress, and PTSD symptoms. The Generalized Anxiety Disorder-7 (GAD-7), Depression Anxiety Stress Scales (DASS), and PTSD Checklist for DSM-5 (PCL-5) were used. Although no prior diagnosis of PTSD was given, the PCL-5 includes items that assess symptoms such as trouble sleeping, feeling easily startled, difficulty concentrating, and strong negative emotions, which can overlap with symptoms of Generalized Anxiety Disorder, Major Depressive Disorder, and Bipolar Disorder. As the case study participant had received these diagnoses in the past, its inclusion allowed for the detection of nuanced symptom changes potentially relevant in measuring changes in mental health status.
The Generalized Anxiety Disorder-7 (GAD-7) is a self-reported assessment measuring the severity of anxiety symptoms and is considered a dimensional indicator of Generalized Anxiety Disorder severity ( 18 ). Scores at the onset indicated mild symptoms, which decreased over the course of the intervention, ending in a normal range ( Figure 2 ). A breakdown of these changes is presented ( Supplementary Table S1 ), quantifying the initial severity and subsequent reductions in GAD-7 scores over the 21-week period.
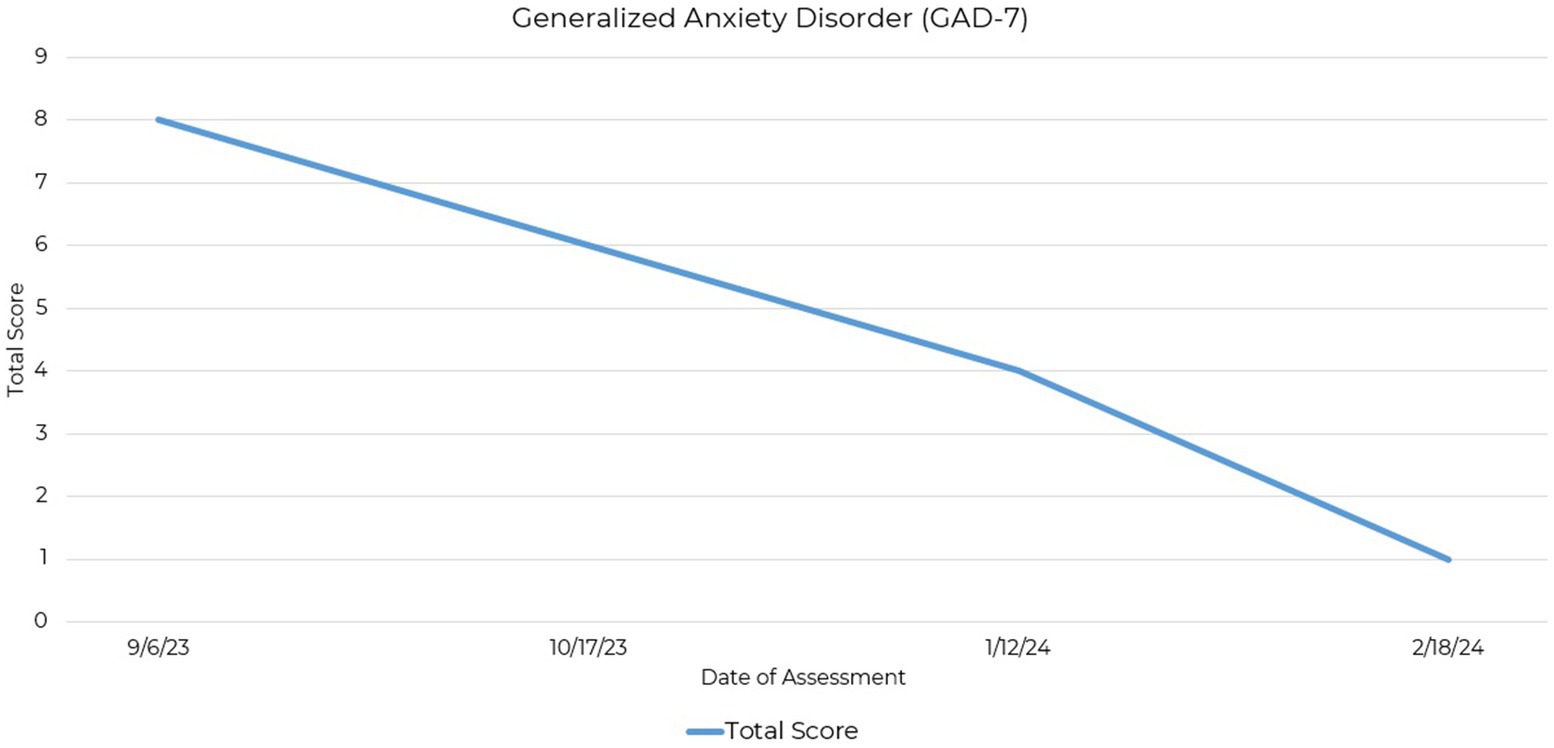
Figure 2 . Line graph depicting the reduction in GAD-7 total scores across four assessment points over a 21-week period.
The Depression Anxiety Stress Scales (DASS) is based on a dimensional rather than a categorical conception of psychological disorders and differentially assesses three negative emotional states: depression, anxiety, and stress ( 19 , 20 ). Initial evaluations showed high levels of these symptoms, especially depression, indicating substantial emotional distress. The 42-item version of the DASS was administered with scores indicating a reduction in symptoms ( Figure 3 ).
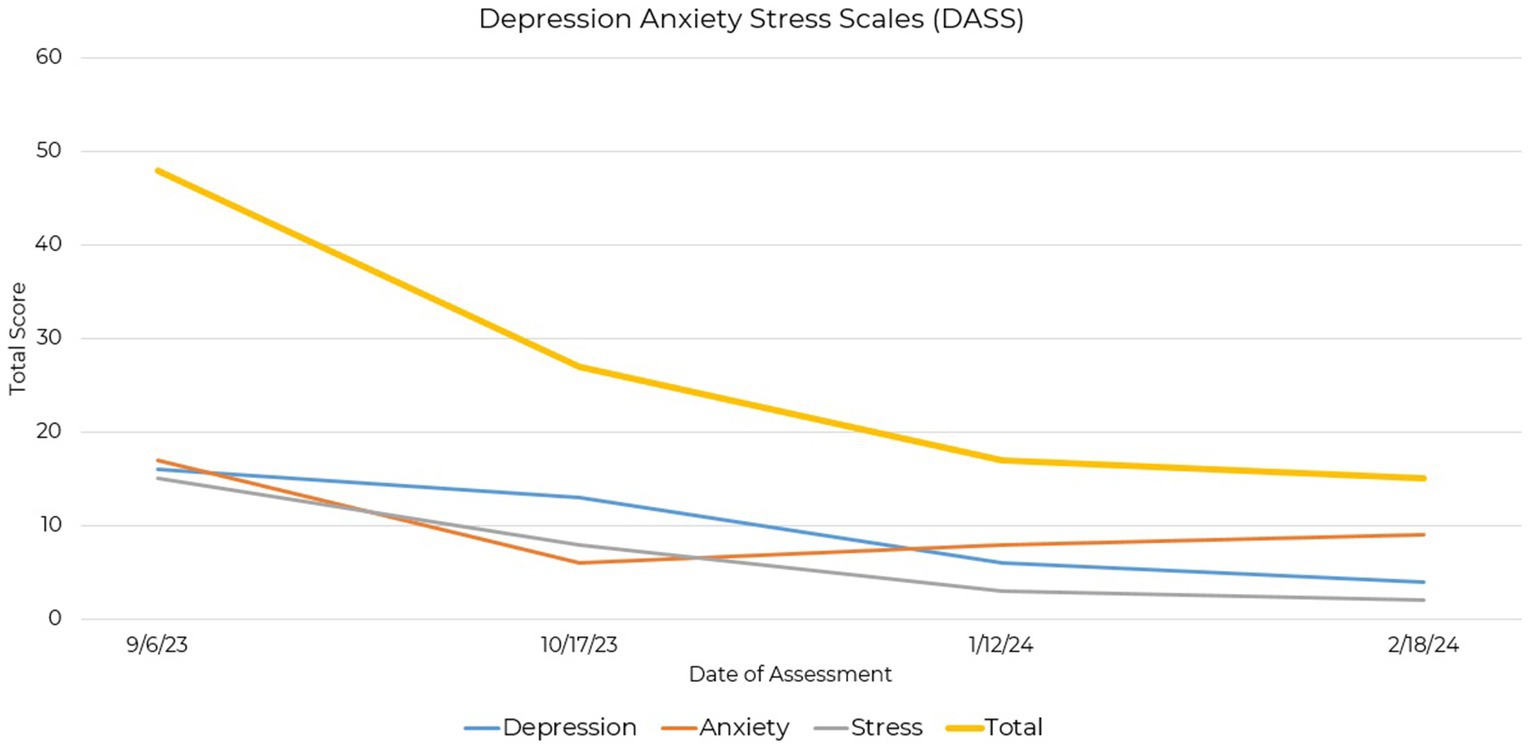
Figure 3 . Line graph depicting the reduction in DASS total and subscale scores across four assessment points over a 21-week period.
Baseline scores indicated moderate to severe levels of depression, anxiety, and stress, with reductions across all three subscales as treatment progressed. Particularly notable was the decrease in depression scores from a moderate level to a normal range. Additionally, anxiety and stress scores showed decreases, indicating a shift towards milder symptomatology ( Supplementary Table S2 ). Differences in initial severity scores between the GAD-7 and DASS anxiety scale could be attributed to the broader assessment coverage provided by the DASS.
The PTSD Checklist for DSM-5 (PCL-5) is a self-report rating scale for assessing the 20 DSM-5 symptoms of post-traumatic stress disorder ( 21 ). Initial assessment revealed endorsement of Criterion D (negative alterations in cognitions and mood), initially exhibiting the highest severity, and Criterion E (alterations in arousal and activity). Subsequent assessments showed a consistent decrease in these scores, with marked improvements observed in both Criterion D and Criterion E ( Figure 4 ).
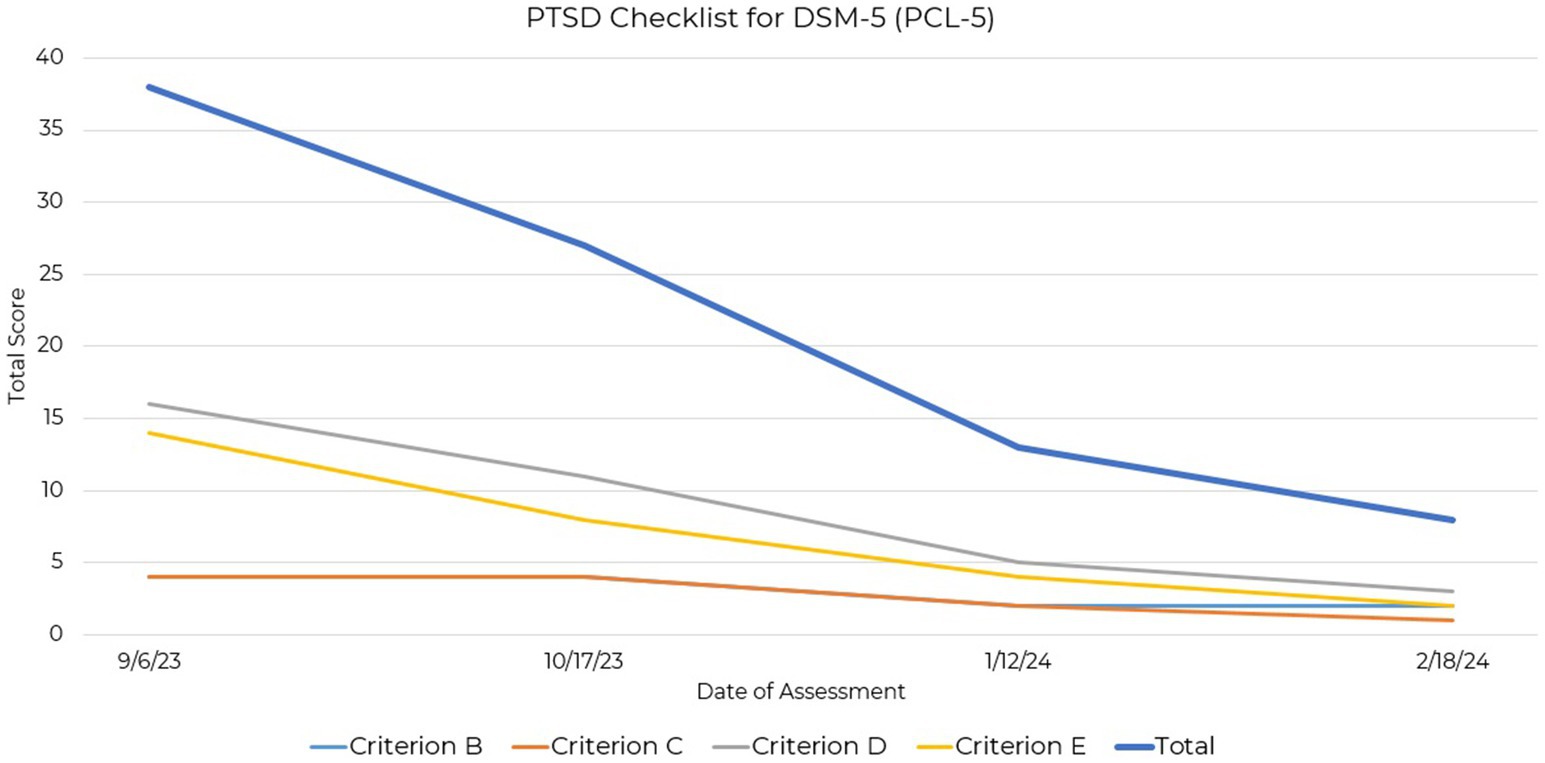
Figure 4 . Line graph depicting the reduction in PCL-5 total and criterio subscales across four assessment points over a 21-week period.
Although there are currently no empirically derived severity ranges for the PCL-5 ( 22 ), reductions in Criteria D and E suggest improvement in mood and arousal symptoms over the assessment period. These criteria, indicative of symptoms seen also in depression and anxiety, may serve as markers of symptom improvement relevant to this case study participant ( Supplementary Table S3 ).
3.2 Qualitative analysis
Qualitative analysis, as delineated by Yin and discussed in Baškarada ( 23 ), was employed to ensure the systematic collection, analysis, and interpretation of data. The qualitative component of data collection centered on the exploration of participant experience using KMT as a treatment for mental illness, recognizing that quantitative assessments may not fully encapsulate the participant’s experience.
Deductive thematic analysis was applied to the case study’s transcript data, focusing on four predefined themes: the personal and emotional journey with KMT, the adoption decision-making process, enhancements in quality of life, and a comparative analysis of conditions before and after KMT. Open-ended, non-leading questions encouraged unbiased responses, developed in line with the Case Report (CARE) guidelines ( 24 ) and as detailed in Supplementary Table S5 . Conducted virtually after informed consent, the interview’s structured approach, conducted by the case study author and guided by these themes, facilitated the categorization of the transcript via a systematic coding procedure. Deductive coding in a single case allows focus on specific theoretical constructs that enable a targeted exploration of the participant’s experiences, as detailed in Supplementary Table S4 , which links the coding strategy directly to the theoretical constructs addressed. Incorporating peer debriefing and soliciting participant feedback on the interview’s comprehensiveness and preliminary findings helped manage researcher bias, ensuring an objective qualitative examination of KMT’s impact in this single case study analysis.
3.2.1 Personal and emotional journey with KMT
The theme ‘Personal and Emotional Journey with KMT’ was used to identify codes for symptom severity, emotional impact, and personal insights. These codes were utilized to document the participant’s mental and physical health fluctuations, emotional responses, and self-reflections on their experience with KMT, focusing on the direct impact of KMT on the individual’s life. Codes developed within this theme identified the experience of a personal and emotional journey with KMT that communicated the transition from a state of profound mental health struggles to a newfound stability and normalcy. Clinically, this reflected a significant shift in self-perception and emotional regulation, which is foundational in the therapeutic process ( 25 , 26 ). The narrative revealed how, for this participant, KMT facilitated a re-engagement with life with movement from a position of vulnerability and isolation to one of agency and connectedness. An example of coded data included the patient stating, “I think everyone has to deal with some anxiety and depression. I feel like the amount that I have in my life at this point is like a normal amount.”
3.2.2 Adoption decision-making process
The “Adoption Decision-Making Process” theme and subsequent code development investigated the participant’s route to choosing the intervention. It examined past treatments, differences between expected and actual effects, factors influencing their choice, intervention tolerability, and the potential impact of earlier access. This distillation attempted clarification of the participant’s decision-making framework. Actual codes applied included ‘Previous Treatments,’ ‘Expectations vs. Reality,’ ‘Journey to KMT,’ and ‘KMT Treatment Availability.’
In this single case, the participant’s decision-making process was driven by frustration with standard-of-care treatments towards the adoption of the KMT approach. This identification of a pivotal decision-making phase was suggestive that active patient engagement in treatment choices might be indicative of the broader search for autonomy and efficacy in treatment strategies among individuals with treatment-resistant conditions. The coded narrative identified the psychological impact of finding new hope after numerous failed attempts with traditional therapies and reflected critical moments of self-determination, where the participant took an active role in their KMT treatment plan. The theme adequately captured that the participant viewed the intervention as sustainable with prolonged continuation as needed to control symptoms. The theme was further able to identify an expression of the participant that they would have preferred earlier introduction to the therapy, indicating that the current substantial relief they experienced may not have been achieved had they not discovered this treatment option on their own. This sentiment highlights the importance of early and proactive consideration of KMT by mental health and other professionals with whom they come in contact. An example of coded data included the patient stating, “I do not think if I had not stumbled upon it myself, and had just a very open and caring practitioner to discuss it with for the first time, that I would be experiencing the sense of relief that I’m experiencing today.”
3.2.3 Enhancements in quality of life
Delineating through deductive analysis, the theme of “Enhancements in Quality of Life” focused on capturing the broad improvements in the participant’s life following KMT adoption. This theme encompassed codes for ‘Lifestyle Adjustments,’ detailing changes in habits and routines, and ‘Life Quality Improvement,’ highlighting overall enhancements in life satisfaction across relationships, work, hobbies, and lifestyle. These codes detailed multifaceted benefits beyond clinical symptom alleviation to identify positive impacts on daily living and well-being.
The findings demonstrated improvements in quality of life post-KMT adoption were suggestive of the therapy’s capacity to effect change beyond symptom relief, touching on aspects of daily functioning, social engagement, and overall well-being. Clinically, this theme highlights the impact of KMT, suggesting that its benefits extend into the psychosocial realm, enhancing patients’ ability to engage in meaningful relationships, pursue interests, and maintain a sense of normalcy. The narratives reveal a restoration of hope and vitality, which is paramount in the recovery process. This enhancement in quality of life can possibly be attributed to the stabilizing effects of KMT on mood, which, in turn, facilitates greater emotional resilience and adaptability in facing life’s challenges. An example of coded data included the patient stating, “I actually made the drive with very little fatigue, no anxiety, great energy. All the things that kind of crop up at those kind of appointments happened, but I felt like I dealt with them just so much more easily. Just easily!”
3.2.4 Conditions before and after KMT
The ‘Conditions Before and After KMT’ theme, through deductive analysis, captured the participant’s experiences pre-and post-KMT adoption, employing codes for detailed comparisons and evaluation of efficacy. Codes within this theme included ‘Before After Comparison’ for specific contrasts in conditions and emotional states and ‘Treatment Efficacy’ assessing KMT’s performance against prior treatments. An example of coded data included the patient stating, “I just spent a lot of time very depressed and feeling very withdrawn,” to describe their prior experience. This structured analysis sought to clarify the participant’s experience of the impacts of KMT on their condition and life, offering a more nuanced understanding of KMT’s effectiveness and its role in altering patient outcomes.
The comparative analysis of conditions before and after implementing KMT for this participant provided a clear contrast between the debilitating effects of bipolar disorder and the empowering influence of effective management through this therapy. This theme is clinically significant as it illustrates the potential of KMT to redefine the treatment landscape for individuals with treatment-resistant bipolar disorder. The narrative highlighted a marked improvement in mood stability, cognitive function, and overall well-being, endorsing the effectiveness of KMT in addressing the complex needs of this population. The theme also reflected the broader implications of KMT for clinical practice, framing KMT as a viable approach for the management of bipolar disorder and enhancing patient outcomes.
From a clinical perspective, the analysis of data from this theme underscored the significance of KMT as a possibly viable intervention for individuals with treatment-resistant bipolar disorder. The collected narrative provided a detailed account of KMT’s impact on personal well-being, decision-making processes related to treatment choices, quality of life improvements, and the condition’s comparative state before and after KMT implementation. These findings offer valuable insights into the potential of KMT to augment clinical practice and patient management.
4 Discussion
The participant further reported that in response to significant reductions in symptoms and under the guidance of their physician, they were able to discontinue the use of some medications and reduce others previously prescribed for the aforementioned chronic conditions. In regards to mood, initial improvements were verbally reported by the patient 2 weeks after diet initiation. Improvements in mood continued and were generally maintained 5 months following the initiation of KMT, offering data on the timeline of symptom improvement. This data may be helpful for aligning the expectations of both patients and clinicians, as well as for informing the design of future research studies. Studies with extended durations or follow-ups may better capture the potential benefits of KMT as a treatment option for mental illnesses.
This participant’s outcome suggests that metabolic health interventions, like ketogenic diets, could offer new directions for treating psychiatric disorders, especially where standard-of-care treatments fall short. The qualitative analysis suggests the possibility that those suffering from Bipolar II disorder may benefit from early introduction to the treatment as an option. While promising, these findings stem from a single case, urging further research to validate these results in broader clinical settings. This work supports further research on the use of KMT as a potential treatment in psychiatry.
5 Conclusion
In this case study, a ketogenic diet significantly improved treatment-resistant depressive symptoms in a patient with bipolar disorder. Both mood assessments and the patient’s experience showed marked improvements. Mood scores moved to normal ranges, indicating stabilized mental health. The patient’s account highlighted improved functioning, better quality of life, and emotional well-being. This case study is of particular interest because it documents the longer-term feasibility of diet implementation, ketone testing compliance, and improvements in relevant symptoms reported by qualitative and quantitative methods. However, any conclusions based on this case study are severely limited by its single-participant sample size and retrospective design, highlighting the need for further research employing randomized controlled trials. Integration of both quantitative and qualitative data may be valuable to adequately represent improvements that researchers are attempting to document as a result of using KMT as a treatment for mental illness.
Data availability statement
The original contributions presented in the study are included in the article/ Supplementary material , further inquiries can be directed to the corresponding author.
Ethics statement
Ethical approval was not required for the studies involving humans because this was a retrospective case study. After the intervention, the participant decided whether or not they wanted to contribute their experience to the research. Informed consent was obtained to use existing quantitative data and collect case study interview data for analysis. The studies were conducted in accordance with the local legislation and institutional requirements. The participants provided their written informed consent to participate in this study. Written informed consent was obtained from the individual(s) for the publication of any potentially identifiable images or data included in this article.
Author contributions
NL: Writing – original draft, Writing – review & editing.
The author(s) declare that no financial support was received for the research, authorship, and/or publication of this article.
Acknowledgments
Special thanks to Erin L. Bellamy, PhD for their critical insights during the peer debriefing process.
Conflict of interest
NL is employed by and owns Family Renewal, Inc. DBA Mental Health Keto.
Publisher’s note
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article, or claim that may be made by its manufacturer, is not guaranteed or endorsed by the publisher.
Supplementary material
The Supplementary material for this article can be found online at: https://www.frontiersin.org/articles/10.3389/fnut.2024.1394679/full#supplementary-material
1. American Psychiatric Association. Diagnostic and statistical manual of mental disorders: DSM-5 . 5th ed Arlington, Virginia: American Psychiatric Publishing (2013).
Google Scholar
2. Ponsoni, A, Branco, LD, Cotrena, C, Shansis, FM, and Fonseca, RP. A longitudinal study of cognition, functional outcome and quality of life in bipolar disorder and major depression. Appl Neuropsychol Adult . (2023) 30:757–63. doi: 10.1080/23279095.2021.1979551
PubMed Abstract | Crossref Full Text | Google Scholar
3. Elsayed, OH, Ercis, M, Pahwa, M, and Singh, B. Treatment-resistant bipolar depression: therapeutic trends, challenges and future directions. Neuropsychiatr Dis Treat . (2022) 18:2927–43. doi: 10.2147/NDT.S273503
4. Levenberg, K, and Cordner, ZA. Bipolar depression: a review of treatment options. Gen Psychiatr . (2022) 35:e100760. doi: 10.1136/gpsych-2022-100760
5. Hu, Y, Zhang, H, Wang, H, Wang, C, Kung, S, and Li, C. Adjunctive antidepressants for the acute treatment of bipolar depression: a systematic review and meta-analysis. Psychiatry Res . (2022) 311:114468. doi: 10.1016/j.psychres.2022.114468
6. Cicek, E, and Sanlier, N. The place of a ketogenic diet in the treatment of resistant epilepsy: a comprehensive review. Nutr Neurosci . (2023) 26:828–41. doi: 10.1080/1028415X.2022.2095819
7. Phelps, JR, Siemers, SV, and El-Mallakh, RS. The ketogenic diet for type II bipolar disorder. Neurocase . (2013) 19:423–6. doi: 10.1080/13554794.2012.690421
Crossref Full Text | Google Scholar
8. Chmiel, I. Ketogenic diet in therapy of bipolar affective disorder -- case report and literature review. Psychiatr Polska . (2022) 56:1. doi: 10.12740/PP/OnlineFirst/136356
9. Saraga, M, Misson, N, and Cattani, E. Ketogenic diet in bipolar disorder. Bipolar Disord . (2020) 22:765–5. doi: 10.1111/bdi.13013
10. Needham, N, Campbell, IH, Grossi, H, Kamenska, I, Rigby, BP, Simpson, SA, et al. Pilot study of a ketogenic diet in bipolar disorder. BJPsych Open . (2023) 9:e176. doi: 10.1192/bjo.2023.568
11. Danan, A, Westman, EC, Saslow, LR, and Ede, G. The ketogenic diet for refractory mental illness: a retrospective analysis of 31 inpatients. Front Psych . (2022) 13:951376. doi: 10.3389/fpsyt.2022.951376
12. Sethi, S, Wakeham, D, Ketter, T, Hooshmand, F, Bjorstead, J, Richards, B, et al. Ketogenic diet intervention on metabolic and psychiatric health in bipolar and schizophrenia: a pilot trial. Psychiatry Res . (2024) 335:115866. doi: 10.1016/j.psychres.2024.115866
13. Norwitz, NG, Dalai, SS, and Palmer, CM. Ketogenic diet as a metabolic treatment for mental illness. Curr Opin Endocrinol Diabetes Obes . (2020) 27:269–74. doi: 10.1097/MED.0000000000000564
14. Campbell, IH, and Campbell, H. The metabolic overdrive hypothesis: hyperglycolysis and glutaminolysis in bipolar mania. Mol Psychiatry . (2024) 29:1521–7. doi: 10.1038/s41380-024-02431-w
15. Choi, J, Kang, J, Kim, T, and Nehs, CJ. Sleep, mood disorders, and the ketogenic diet: potential therapeutic targets for bipolar disorder and schizophrenia. Front Psych . (2024) 15:1358578. doi: 10.3389/fpsyt.2024.1358578
16. Yu, B, Ozveren, R, and Dalai, SS. Ketogenic diet as a metabolic therapy for bipolar disorder: Clinical developments. J Affect Disord Rep . (2022) 11:100457. doi: 10.21203/rs.3.rs-334453/v2
17. Chu, DY, Ravelli, MN, Faltersack, KM, Woods, AL, Almane, D, Li, Z, et al. Hypocarnitinemia and its effect on seizure control in adult patients with intractable epilepsy on the modified Atkins diet. Front Nutr . (2024) 10:1304209. doi: 10.3389/fnut.2023.1304209
18. Rutter, LA, and Brown, TA. Psychometric properties of the generalized anxiety disorder Scale-7 (GAD-7) in outpatients with anxiety and mood disorders. J Psychopathol Behav Assess . (2017) 39:140–6. doi: 10.1007/s10862-016-9571-9
19. Antony, MM, Bieling, PJ, Cox, BJ, Enns, MW, and Swinson, RP. Psychometric properties of the 42-item and 21-item versions of the depression anxiety stress scales in clinical groups and a community sample. Psychol Assess . (1998) 10:176–81. doi: 10.1037/1040-3590.10.2.176
20. Brown, TA, Chorpita, BF, Korotitsch, W, and Barlow, DH. Psychometric properties of the depression anxiety stress scales (DASS) in clinical samples. Behav Res Ther . (1997) 35:79–89. doi: 10.1016/S0005-7967(96)00068-X
21. Forkus, SR, Raudales, AM, Rafiuddin, HS, Weiss, NH, Messman, BA, and Contractor, AA. The posttraumatic stress disorder (PTSD) checklist for DSM–5: a systematic review of existing psychometric evidence. Clin Psychol Sci Pract . (2023) 30:110–21. doi: 10.1037/cps0000111
22. National Center for PTSD. Using the PTSD checklist for DSM-5 (PCL-5). Available at: https://www.ptsd.va.gov/professional/assessment/documents/using-PCL5.pdf (Accessed February 23, 2024).
23. Baskarada, S. Qualitative case study guidelines. (2014) Available at: https://papers.ssrn.com/abstract=2559424 (Accessed February 19, 2024).
24. Riley, DS, Barber, MS, Kienle, GS, Aronson, JK, von Schoen-Angerer, T, Tugwell, P, et al. CARE guidelines for case reports: explanation and elaboration document. J Clin Epidemiol . (2017) 89:218–35. doi: 10.1016/j.jclinepi.2017.04.026
25. Miola, A, Cattarinussi, G, Antiga, G, Caiolo, S, Solmi, M, and Sambataro, F. Difficulties in emotion regulation in bipolar disorder: a systematic review and meta-analysis. J Affect Disord . (2022) 302:352–60. doi: 10.1016/j.jad.2022.01.102
26. Ironside, ML, Johnson, SL, and Carver, CS. Identity in bipolar disorder: self-worth and achievement. J Pers . (2020) 88:45–58. doi: 10.1111/jopy.12461
Keywords: ketogenic diet, bipolar disorder, KMT, ketogenic metabolic therapy, metabolic psychiatry, mood disorders, treatment-refractory depression, clinical psychology
Citation: Laurent N (2024) Retrospective case study: ketogenic metabolic therapy in the effective management of treatment-resistant depressive symptoms in bipolar disorder. Front. Nutr . 11:1394679. doi: 10.3389/fnut.2024.1394679
Received: 01 March 2024; Accepted: 30 July 2024; Published: 12 August 2024.
Reviewed by:
Copyright © 2024 Laurent. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY) . The use, distribution or reproduction in other forums is permitted, provided the original author(s) and the copyright owner(s) are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Nicole Laurent, [email protected]
Disclaimer: All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article or claim that may be made by its manufacturer is not guaranteed or endorsed by the publisher.
- Research article
- Open access
- Published: 15 August 2024
The impact of adverse childhood experiences on multimorbidity: a systematic review and meta-analysis
- Dhaneesha N. S. Senaratne 1 ,
- Bhushan Thakkar 1 ,
- Blair H. Smith 1 ,
- Tim G. Hales 2 ,
- Louise Marryat 3 &
- Lesley A. Colvin 1
BMC Medicine volume 22 , Article number: 315 ( 2024 ) Cite this article
293 Accesses
15 Altmetric
Metrics details
Adverse childhood experiences (ACEs) have been implicated in the aetiology of a range of health outcomes, including multimorbidity. In this systematic review and meta-analysis, we aimed to identify, synthesise, and quantify the current evidence linking ACEs and multimorbidity.
We searched seven databases from inception to 20 July 2023: APA PsycNET, CINAHL Plus, Cochrane CENTRAL, Embase, MEDLINE, Scopus, and Web of Science. We selected studies investigating adverse events occurring during childhood (< 18 years) and an assessment of multimorbidity in adulthood (≥ 18 years). Studies that only assessed adverse events in adulthood or health outcomes in children were excluded. Risk of bias was assessed using the ROBINS-E tool. Meta-analysis of prevalence and dose–response meta-analysis methods were used for quantitative data synthesis. This review was pre-registered with PROSPERO (CRD42023389528).
From 15,586 records, 25 studies were eligible for inclusion (total participants = 372,162). The prevalence of exposure to ≥ 1 ACEs was 48.1% (95% CI 33.4 to 63.1%). The prevalence of multimorbidity was 34.5% (95% CI 23.4 to 47.5%). Eight studies provided sufficient data for dose–response meta-analysis (total participants = 197,981). There was a significant dose-dependent relationship between ACE exposure and multimorbidity ( p < 0.001), with every additional ACE exposure contributing to a 12.9% (95% CI 7.9 to 17.9%) increase in the odds for multimorbidity. However, there was heterogeneity among the included studies ( I 2 = 76.9%, Cochran Q = 102, p < 0.001).
Conclusions
This is the first systematic review and meta-analysis to synthesise the literature on ACEs and multimorbidity, showing a dose-dependent relationship across a large number of participants. It consolidates and enhances an extensive body of literature that shows an association between ACEs and individual long-term health conditions, risky health behaviours, and other poor health outcomes.
Peer Review reports
In recent years, adverse childhood experiences (ACEs) have been identified as factors of interest in the aetiology of many conditions [ 1 ]. ACEs are potentially stressful events or environments that occur before the age of 18. They have typically been considered in terms of abuse (e.g. physical, emotional, sexual), neglect (e.g. physical, emotional), and household dysfunction (e.g. parental separation, household member incarceration, household member mental illness) but could also include other forms of stress, such as bullying, famine, and war. ACEs are common: estimates suggest that 47% of the UK population have experienced at least one form, with 12% experiencing four or more [ 2 ]. ACEs are associated with poor outcomes in a range of physical health, mental health, and social parameters in adulthood, with greater ACE burden being associated with worse outcomes [ 1 , 2 , 3 , 4 , 5 , 6 , 7 , 8 ].
Over a similar timescale, multimorbidity has emerged as a significant heath challenge. It is commonly defined as the co-occurrence of two or more long-term conditions (LTCs), with a long-term condition defined as any physical or mental health condition lasting, or expected to last, longer than 1 year [ 9 ]. Multimorbidity is both common and age-dependent, with a global adult prevalence of 37% that rises to 51% in adults over 60 [ 10 , 11 ]. Individuals living with multimorbidity face additional challenges in managing their health, such as multiple appointments, polypharmacy, and the lack of continuity of care [ 12 , 13 , 14 ]. Meanwhile, many healthcare systems struggle to manage the additional cost and complexity of people with multimorbidity as they have often evolved to address the single disease model [ 15 , 16 ]. As global populations continue to age, with an estimated 2.1 billion adults over 60 by 2050, the pressures facing already strained healthcare systems will continue to grow [ 17 ]. Identifying factors early in the aetiology of multimorbidity may help to mitigate the consequences of this developing healthcare crisis.
Many mechanisms have been suggested for how ACEs might influence later life health outcomes, including the risk of developing individual LTCs. Collectively, they contribute to the idea of ‘toxic stress’; cumulative stress during key developmental phases may affect development [ 18 ]. ACEs are associated with measures of accelerated cellular ageing, including changes in DNA methylation and telomere length [ 19 , 20 ]. ACEs may lead to alterations in stress-signalling pathways, including changes to the immune, endocrine, and cardiovascular systems [ 21 , 22 , 23 ]. ACEs are also associated with both structural and functional differences in the brain [ 24 , 25 , 26 , 27 ]. These diverse biological changes underpin psychological and behavioural changes, predisposing individuals to poorer self-esteem and risky health behaviours, which may in turn lead to increased risk of developing individual LTCs [ 1 , 2 , 28 , 29 , 30 , 31 , 32 ]. A growing body of evidence has therefore led to an increased focus on developing trauma-informed models of healthcare, in which the impact of negative life experiences is incorporated into the assessment and management of LTCs [ 33 ].
Given the contributory role of ACEs in the aetiology of individual LTCs, it is reasonable to suspect that ACEs may also be an important factor in the development of multimorbidity. Several studies have implicated ACEs in the aetiology of multimorbidity, across different cohorts and populations, but to date no meta-analyses have been performed to aggregate this evidence. In this review, we aim to summarise the state of the evidence linking ACEs and multimorbidity, to quantify the strength of any associations through meta-analysis, and to highlight the challenges of research in this area.
Search strategy and selection criteria
We conducted a systematic review and meta-analysis that was prospectively registered in the International Prospective Register of Systematic Reviews (PROSPERO) on 25 January 2023 (ID: CRD42023389528) and reported using the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) guidelines.
We developed a search strategy based on previously published literature reviews and refined it following input from subject experts, an academic librarian, and patient and public partners (Additional File 1: Table S1). We searched the following seven databases from inception to 20 July 2023: APA PsycNET, CINAHL Plus, Cochrane CENTRAL, Embase, MEDLINE, Scopus, and Web of Science. The search results were imported into Covidence (Veritas Health Innovation, Melbourne, Australia), which automatically identified and removed duplicate entries. Two reviewers (DS and BT) independently performed title and abstract screening and full text review. Discrepancies were resolved by a third reviewer (LC).
Reports were eligible for review if they included adults (≥ 18 years), adverse events occurring during childhood (< 18 years), and an assessment of multimorbidity or health status based on LTCs. Reports that only assessed adverse events in adulthood or health outcomes in children were excluded.
The following study designs were eligible for review: randomised controlled trials, cohort studies, case–control studies, cross-sectional studies, and review articles with meta-analysis. Editorials, case reports, and conference abstracts were excluded. Systematic reviews without a meta-analysis and narrative synthesis review articles were also excluded; however, their reference lists were screened for relevant citations.
Data analysis
Two reviewers (DS and BT) independently performed data extraction into Microsoft Excel (Microsoft Corporation, Redmond, USA) using a pre-agreed template. Discrepancies were resolved by consensus discussion with a third reviewer (LC). Data extracted from each report included study details (author, year, study design, sample cohort, sample size, sample country of origin), patient characteristics (age, sex), ACE information (definition, childhood cut-off age, ACE assessment tool, number of ACEs, list of ACEs, prevalence), multimorbidity information (definition, multimorbidity assessment tool, number of LTCs, list of LTCs, prevalence), and analysis parameters (effect size, model adjustments). For meta-analysis, we extracted ACE groups, number of ACE cases, number of multimorbidity cases, number of participants, odds ratios or regression beta coefficients, and 95% confidence intervals (95% CI). Where data were partially reported or missing, we contacted the study authors directly for further information.
Two reviewers (DS and BT) independently performed risk of bias assessments of each included study using the Risk Of Bias In Non-randomized Studies of Exposures (ROBINS-E) tool [ 34 ]. The ROBINS-E tool assesses the risk of bias for the study outcome relevant to the systematic review question, which may not be the primary study outcome. It assesses risk of bias across seven domains; confounding, measurement of the exposure, participant selection, post-exposure interventions, missing data, measurement of the outcome, and selection of the reported result. The overall risk of bias for each study was determined using the ROBINS-E algorithm. Discrepancies were resolved by consensus discussion.
All statistical analyses were performed in R version 4.2.2 using the RStudio integrated development environment (RStudio Team, Boston, USA). To avoid repetition of participant data, where multiple studies analysed the same patient cohort, we selected the study with the best reporting of raw data for meta-analysis and the largest sample size. Meta-analysis of prevalence was performed with the meta package [ 35 ], using logit transformations within a generalised linear mixed model, and reporting the random-effects model [ 36 ]. Inter-study heterogeneity was assessed and reported using the I 2 statistic, Cochran Q statistic, and Cochran Q p -value. Dose–response meta-analysis was performed using the dosresmeta package [ 37 ] following the method outlined by Greenland and Longnecker (1992) [ 38 , 39 ]. Log-linear and non-linear (restricted cubic spline, with knots at 5%, 35%, 65%, and 95%) random effects models were generated, and goodness of fit was evaluated using a Wald-type test (denoted by X 2 ) and the Akaike information criterion (AIC) [ 39 ].
Patient and public involvement
The Consortium Against Pain Inequality (CAPE) Chronic Pain Advisory Group (CPAG) consists of individuals with lived experiences of ACEs, chronic pain, and multimorbidity. CPAG was involved in developing the research question. The group has experience in systematic review co-production (in progress).
The search identified 15,586 records, of which 25 met inclusion criteria for the systematic review (Fig. 1 ) [ 40 , 41 , 42 , 43 , 44 , 45 , 46 , 47 , 48 , 49 , 50 , 51 , 52 , 53 , 54 , 55 , 56 , 57 , 58 , 59 , 60 , 61 , 62 , 63 , 64 ]. The summary characteristics can be found in Additional File 1: Table S2. Most studies examined European ( n = 11) or North American ( n = 9) populations, with a few looking at Asian ( n = 3) or South American ( n = 1) populations and one study examining a mixed cohort (European and North American populations). The total participant count (excluding studies performed on the same cohort) was 372,162. Most studies had a female predominance (median 53.8%, interquartile range (IQR) 50.9 to 57.4%).

Flow chart of selection of studies into the systematic review and meta-analysis. Flow chart of selection of studies into the systematic review and meta-analysis. ACE, adverse childhood experience; MM, multimorbidity; DRMA, dose–response meta-analysis
All studies were observational in design, and so risk of bias assessments were performed using the ROBINS-E tool (Additional File 1: Table S3) [ 34 ]. There were some consistent risks observed across the studies, especially in domain 1 (risk of bias due to confounding) and domain 3 (risk of bias due to participant selection). In domain 1, most studies were ‘high risk’ ( n = 24) as they controlled for variables that could have been affected by ACE exposure (e.g. smoking status) [ 40 , 41 , 43 , 44 , 45 , 46 , 47 , 48 , 49 , 50 , 51 , 52 , 53 , 54 , 55 , 56 , 57 , 58 , 59 , 60 , 61 , 62 , 63 , 64 ]. In domain 3, some studies were ‘high risk’ ( n = 7) as participant selection was based on participant characteristics that could have been influenced by ACE exposure (e.g. through recruitment at an outpatient clinic) [ 45 , 48 , 49 , 51 , 53 , 54 , 58 ]. The remaining studies were deemed as having ‘some concerns’ ( n = 18) as participant selection occurred at a time after ACE exposure, introducing a risk of survivorship bias [ 40 , 41 , 42 , 43 , 44 , 46 , 47 , 50 , 52 , 55 , 56 , 57 , 59 , 60 , 61 , 62 , 63 , 64 ].
Key differences in risk of bias were seen in domain 2 (risk of bias due to exposure measurement) and domain 5 (risk of bias due to missing data). In domain 2, some studies were ‘high risk’ as they used a narrow or atypical measure of ACEs ( n = 8) [ 40 , 42 , 44 , 46 , 55 , 56 , 60 , 64 ]; others were graded as having ‘some concerns’ as they used a broader but still incomplete measure of ACEs ( n = 8) [ 43 , 45 , 48 , 49 , 50 , 52 , 54 , 62 ]; the remainder were ‘low risk’ as they used an established or comprehensive list of ACE questions [ 41 , 47 , 51 , 53 , 57 , 58 , 59 , 61 , 63 ]. In domain 5, some studies were ‘high risk’ as they failed to acknowledge or appropriately address missing data ( n = 7) [ 40 , 42 , 43 , 45 , 51 , 53 , 60 ]; others were graded as having ‘some concerns’ as they had a significant amount of missing data (> 10% for exposure, outcome, or confounders) but mitigated for this with appropriate strategies ( n = 6) [ 41 , 50 , 56 , 57 , 62 , 64 ]; the remainder were ‘low risk’ as they reported low levels of missing data ( n = 12) [ 44 , 46 , 47 , 48 , 49 , 52 , 54 , 55 , 58 , 59 , 61 , 63 ].
Most studies assessed an exposure that was ‘adverse childhood experiences’ ( n = 10) [ 41 , 42 , 50 , 51 , 53 , 57 , 58 , 61 , 63 , 64 ], ‘childhood maltreatment’ ( n = 6) [ 44 , 45 , 46 , 48 , 49 , 59 ], or ‘childhood adversity’ ( n = 3) [ 47 , 54 , 62 ]. The other exposures studied were ‘birth phase relative to World War Two’ [ 40 ], ‘childhood abuse’ [ 43 ], ‘childhood disadvantage’ [ 56 ], ‘childhood racial discrimination’ [ 55 ], ‘childhood trauma’ [ 52 ], and ‘quality of childhood’ (all n = 1) [ 60 ]. More than half of studies ( n = 13) did not provide a formal definition of their exposure of choice [ 42 , 43 , 44 , 45 , 49 , 52 , 53 , 54 , 57 , 58 , 60 , 61 , 64 ]. The upper age limit for childhood ranged from < 15 to < 18 years with the most common cut-off being < 18 years ( n = 9). The median number of ACEs measured in each study was 7 (IQR 4–10). In total, 58 different ACEs were reported; 17 ACEs were reported by at least three studies, whilst 33 ACEs were reported by only one study. The most frequently reported ACEs were physical abuse ( n = 19) and sexual abuse ( n = 16) (Table 1 ). The exposure details for each study can be found in Additional File 1: Table S4.
Thirteen studies provided sufficient data to allow for a meta-analysis of the prevalence of exposure to ≥ 1 ACE; the pooled prevalence was 48.1% (95% CI 33.4 to 63.1%, I 2 = 99.9%, Cochran Q = 18,092, p < 0.001) (Fig. 2 ) [ 41 , 43 , 44 , 46 , 47 , 49 , 50 , 52 , 53 , 57 , 59 , 61 , 63 ]. Six studies provided sufficient data to allow for a meta-analysis of the prevalence of exposure to ≥ 4 ACEs; the pooled prevalence was 12.3% (95% CI 3.5 to 35.4%, I 2 = 99.9%, Cochran Q = 9071, p < 0.001) (Additional File 1: Fig. S1) [ 46 , 50 , 51 , 53 , 59 , 63 ].

Meta-analysis of prevalence of exposure to ≥ 1 adverse childhood experiences. Meta-analysis of prevalence of exposure to ≥ 1 adverse childhood experience. ACE, adverse childhood experience; CI, confidence interval
Thirteen studies explicitly assessed multimorbidity as an outcome, and all of these defined the threshold for multimorbidity as the presence of two or more LTCs [ 40 , 41 , 42 , 44 , 46 , 47 , 50 , 55 , 57 , 60 , 61 , 62 , 64 ]. The remaining studies assessed comorbidities, morbidity, or disease counts [ 43 , 45 , 48 , 49 , 51 , 52 , 53 , 54 , 56 , 58 , 59 , 63 ]. The median number of LTCs measured in each study was 14 (IQR 12–21). In total, 115 different LTCs were reported; 36 LTCs were reported by at least three studies, whilst 63 LTCs were reported by only one study. Two studies did not report the specific LTCs that they measured [ 51 , 53 ]. The most frequently reported LTCs were hypertension ( n = 22) and diabetes ( n = 19) (Table 2 ). Fourteen studies included at least one mental health LTC. The outcome details for each study can be found in Additional File 1: Table S5.
Fifteen studies provided sufficient data to allow for a meta-analysis of the prevalence of multimorbidity; the pooled prevalence was 34.5% (95% CI 23.4 to 47.5%, I 2 = 99.9%, Cochran Q = 24,072, p < 0.001) (Fig. 3 ) [ 40 , 41 , 44 , 46 , 47 , 49 , 50 , 51 , 52 , 55 , 57 , 58 , 59 , 60 , 63 ].

Meta-analysis of prevalence of multimorbidity. Meta-analysis of prevalence of multimorbidity. CI, confidence interval; LTC, long-term condition; MM, multimorbidity
All studies reported significant positive associations between measures of ACE and multimorbidity, though they varied in their means of analysis and reporting of the relationship. Nine studies reported an association between the number of ACEs (variably considered as a continuous or categorical parameter) and multimorbidity [ 41 , 43 , 46 , 47 , 50 , 56 , 57 , 61 , 64 ]. Eight studies reported an association between the number of ACEs and comorbidity counts in specific patient populations [ 45 , 48 , 49 , 51 , 53 , 58 , 59 , 63 ]. Six studies reported an association between individual ACEs or ACE subgroups and multimorbidity [ 42 , 43 , 44 , 47 , 55 , 62 ]. Two studies incorporated a measure of frequency within their ACE measurement tool and reported an association between this ACE score and multimorbidity [ 52 , 54 ]. Two studies reported an association between proxy measures for ACEs and multimorbidity; one reported ‘birth phase relative to World War Two’, and the other reported a self-report on the overall quality of childhood [ 40 , 60 ].
Eight studies, involving a total of 197,981 participants, provided sufficient data (either in the primary text, or following author correspondence) for quantitative synthesis [ 41 , 46 , 47 , 49 , 50 , 51 , 57 , 58 ]. Log-linear (Fig. 4 ) and non-linear (Additional File 1: Fig. S2) random effects models were compared for goodness of fit: the Wald-type test for linearity was non-significant ( χ 2 = 3.7, p = 0.16) and the AIC was lower for the linear model (− 7.82 vs 15.86) indicating that the log-linear assumption was valid. There was a significant dose-dependent relationship between ACE exposure and multimorbidity ( p < 0.001), with every additional ACE exposure contributing to a 12.9% (95% CI 7.9 to 17.9%) increase in the odds for multimorbidity ( I 2 = 76.9%, Cochran Q = 102, p < 0.001).

Dose–response meta-analysis of the relationship between adverse childhood experiences and multimorbidity. Dose–response meta-analysis of the relationship between adverse childhood experiences and multimorbidity. Solid black line represents the estimated relationship; dotted black lines represent the 95% confidence intervals for this estimate. ACE, adverse childhood experience
This systematic review and meta-analysis synthesised the literature on ACEs and multimorbidity and showed a dose-dependent relationship across a large number of participants. Each additional ACE exposure contributed to a 12.9% (95% CI 7.9 to 17.9%) increase in the odds for multimorbidity. This adds to previous meta-analyses that have shown an association between ACEs and individual LTCs, health behaviours, and other health outcomes [ 1 , 28 , 31 , 65 , 66 ]. However, we also identified substantial inter-study heterogeneity that is likely to have arisen due to variation in the definitions, methodology, and analysis of the included studies, and so our results should be interpreted with these limitations in mind.
Although 25 years have passed since the landmark Adverse Childhood Experiences Study by Felitti et al. [ 3 ], there is still no consistent approach to determining what constitutes an ACE. This is reflected in this review, where fewer than half of the 58 different ACEs ( n = 25, 43.1%) were reported by more than one study and no study reported more than 15 ACEs. Even ACE types that are commonly included are not always assessed in the same way [ 67 ], and furthermore, the same question can be interpreted differently in different contexts (e.g. physical punishment for bad behaviour was socially acceptable 50 years ago but is now considered physical abuse in the UK). Although a few validated questionnaires exist, they often focus on a narrow range of ACEs; for example, the childhood trauma questionnaire demonstrates good reliability and validity but focuses on interpersonal ACEs, missing out on household factors (e.g. parental separation), and community factors (e.g. bullying) [ 68 ]. Many studies were performed on pre-existing research cohorts or historic healthcare data, where the study authors had limited or no influence on the data collected. As a result, very few individual studies reported on the full breadth of potential ACEs.
ACE research is often based on ACE counts, where the types of ACEs experienced are summed into a single score that is taken as a proxy measure of the burden of childhood stress. The original Adverse Childhood Experiences Study by Felitti et al. took this approach [ 3 ], as did 17 of the studies included in this review and our own quantitative synthesis. At the population level, there are benefits to this: ACE counts provide quantifiable and comparable metrics, they are easy to collect and analyse, and in many datasets, they are the only means by which an assessment of childhood stress can be derived. However, there are clear limitations to this method when considering experiences at the individual level, not least the inherent assumptions that different ACEs in the same person are of equal weight or that the same ACE in different people carries the same burden of childhood stress. This limitation was strongly reinforced by our patient and public involvement group (CPAG). Two studies in this review incorporated frequency within their ACE scoring system [ 52 , 54 ], which adds another dimension to the assessment, but this is insufficient to understand and quantify the ‘impact’ of an ACE within an epidemiological framework.
The definitions of multimorbidity were consistent across the relevant studies but the contributory long-term conditions varied. Fewer than half of the 115 different LTCs ( n = 52, 45.2%) were reported by more than one study. Part of the challenge is the classification of healthcare conditions. For example, myocardial infarction is commonly caused by coronary heart disease, and both are a form of heart disease. All three were reported as LTCs in the included studies, but which level of pathology should be reported? Mental health LTCs were under-represented within the condition list, with just over half of the included studies assessing at least one ( n = 14, 56.0%). Given the strong links between ACEs and mental health, and the impact of mental health on quality of life, this is an area for improvement in future research [ 31 , 32 ]. A recent Delphi consensus study by Ho et al. may help to address these issues: following input from professionals and members of the public they identified 24 LTCs to ‘always include’ and 35 LTCs to ‘usually include’ in multimorbidity research, including nine mental health conditions [ 9 ].
As outlined in the introduction, there is a strong evidence base supporting the link between ACEs and long-term health outcomes, including specific LTCs. It is not unreasonable to extrapolate this association to ACEs and multimorbidity, though to our knowledge, the pathophysiological processes that link the two have not been precisely identified. However, similar lines of research are being independently followed in both fields and these areas of overlap may suggest possible mechanisms for a relationship. For example, both ACEs and multimorbidity have been associated with markers of accelerated epigenetic ageing [ 69 , 70 ], mitochondrial dysfunction [ 71 , 72 ], and inflammation [ 22 , 73 ]. More work is required to better understand how these concepts might be linked.
This review used data from a large participant base, with information from 372,162 people contributing to the systematic review and information from 197,981 people contributing to the dose–response meta-analysis. Data from the included studies originated from a range of sources, including healthcare settings and dedicated research cohorts. We believe this is of a sufficient scale and variety to demonstrate the nature and magnitude of the association between ACEs and multimorbidity in these populations.
However, there are some limitations. Firstly, although data came from 11 different countries, only two of those were from outside Europe and North America, and all were from either high- or middle-income countries. Data on ACEs from low-income countries have indicated a higher prevalence of any ACE exposure (consistently > 70%) [ 74 , 75 ], though how well this predicts health outcomes in these populations is unknown.
Secondly, studies in this review utilised retrospective participant-reported ACE data and so are at risk of recall and reporting bias. Studies utilising prospective assessments are rare and much of the wider ACE literature is open to a similar risk of bias. To date, two studies have compared prospective and retrospective ACE measurements, demonstrating inconsistent results [ 76 , 77 ]. However, these studies were performed in New Zealand and South Africa, two countries not represented by studies in our review, and had relatively small sample sizes (1037 and 1595 respectively). It is unclear whether these are generalisable to other population groups.
Thirdly, previous research has indicated a close relationship between ACEs and childhood socio-economic status (SES) [ 78 ] and between SES and multimorbidity [ 10 , 79 ]. However, the limitations of the included studies meant we were unable to separate the effect of ACEs from the effect of childhood SES on multimorbidity in this review. Whilst two studies included childhood SES as covariates in their models, others used measures from adulthood (such as adulthood SES, income level, and education level) that are potentially influenced by ACEs and therefore increase the risk of bias due to confounding (Additional File 1: Table S3). Furthermore, as for ACEs and multimorbidity, there is no consistently applied definition of SES and different measures of SES may produce different apparent effects [ 80 ]. The complex relationships between ACEs, childhood SES, and multimorbidity remain a challenge for research in this field.
Fourthly, there was a high degree of heterogeneity within included studies, especially relating to the definition and measurement of ACEs and multimorbidity. Whilst this suggests that our results should be interpreted with caution, it is reassuring to see that our meta-analysis of prevalence estimates for exposure to any ACE (48.1%) and multimorbidity (34.5%) are in line with previous estimates in similar populations [ 2 , 11 ]. Furthermore, we believe that the quantitative synthesis of these relatively heterogenous studies provides important benefit by demonstrating a strong dose–response relationship across a range of contexts.
Our results strengthen the evidence supporting the lasting influence of childhood conditions on adult health and wellbeing. How this understanding is best incorporated into routine practice is still not clear. Currently, the lack of consistency in assessing ACEs limits our ability to understand their impact at both the individual and population level and poses challenges for those looking to incorporate a formalised assessment. Whilst most risk factors for disease (e.g. blood pressure) are usually only relevant within healthcare settings, ACEs are relevant to many other sectors (e.g. social care, education, policing) [ 81 , 82 , 83 , 84 ], and so consistency of assessment across society is both more important and more challenging to achieve.
Some have suggested that the evidence for the impact of ACEs is strong enough to warrant screening, which would allow early identification of potential harms to children and interventions to prevent them. This approach has been implemented in California, USA [ 85 , 86 , 87 ]. However, this is controversial, and others argue that screening is premature with the current evidence base [ 88 , 89 , 90 ]. Firstly, not everyone who is exposed to ACEs develops poor health outcomes, and it is not clear how to identify those who are at highest risk. Many people appear to be vulnerable, with more adverse health outcomes following ACE exposure than those who are not exposed, whilst others appear to be more resilient, with good health in later life despite multiple ACE exposures [ 91 ] It may be that supportive environments can mitigate the long-term effects of ACE exposure and promote resilience [ 92 , 93 ]. Secondly, there are no accepted interventions for managing the impact of an identified ACE. As identified above, different ACEs may require input from different sectors (e.g. healthcare, social care, education, police), and so collating this evidence may be challenging. At present, ACEs screening does not meet the Wilson-Jungner criteria for a screening programme [ 94 ].
Existing healthcare systems are poorly designed to deal with the complexities of addressing ACEs and multimorbidity. Possibly, ways to improve this might be allocating more time per patient, prioritising continuity of care to foster long-term relationships, and greater integration between different healthcare providers (most notably primary vs secondary care teams, or physical vs mental health teams). However, such changes often demand additional resources (e.g. staff, infrastructure, processes), which are challenging to source when existing healthcare systems are already stretched [ 95 , 96 ]. Nevertheless, increasing the spotlight on ACEs and multimorbidity may help to focus attention and ultimately bring improvements to patient care and experience.
ACEs are associated with a range of poor long-term health outcomes, including harmful health behaviours and individual long-term conditions. Multimorbidity is becoming more common as global populations age, and it increases the complexity and cost of healthcare provision. This is the first systematic review and meta-analysis to synthesise the literature on ACEs and multimorbidity, showing a statistically significant dose-dependent relationship across a large number of participants, albeit with a high degree of inter-study heterogeneity. This consolidates and enhances an increasing body of data supporting the role of ACEs in determining long-term health outcomes. Whilst these observational studies do not confirm causality, the weight and consistency of evidence is such that we can be confident in the link. The challenge for healthcare practitioners, managers, policymakers, and governments is incorporating this body of evidence into routine practice to improve the health and wellbeing of our societies.
Availability of data and materials
No additional data was generated for this review. The data used were found in the referenced papers or provided through correspondence with the study authors.
Abbreviations
Adverse childhood experience
Akaike information criterion
CONSORTIUM Against pain inequality
Confidence interval
Chronic pain advisory group
Interquartile range
Long-term condition
International prospective register of systematic reviews
Preferred reporting items for systematic reviews and meta-analyses
Risk of bias in non-randomised studies of exposures
Socio-economic status
Hughes K, Bellis MA, Hardcastle KA, Sethi D, Butchart A, Mikton C, et al. The effect of multiple adverse childhood experiences on health: a systematic review and meta-analysis. Lancet Public Health. 2017;2:e356–66.
Article PubMed Google Scholar
Bellis MA, Lowey H, Leckenby N, Hughes K, Harrison D. Adverse childhood experiences: retrospective study to determine their impact on adult health behaviours and health outcomes in a UK population. J Public Health Oxf Engl. 2014;36:81–91.
Article Google Scholar
Felitti VJ, Anda RF, Nordenberg D, Williamson DF, Spitz AM, Edwards V, et al. Relationship of childhood abuse and household dysfunction to many of the leading causes of death in adults. The Adverse Childhood Experiences (ACE) Study. Am J Prev Med. 1998;14:245–58.
Article CAS PubMed Google Scholar
Maniglio R. The impact of child sexual abuse on health: a systematic review of reviews. Clin Psychol Rev. 2009;29:647–57.
Yu J, Patel RA, Haynie DL, Vidal-Ribas P, Govender T, Sundaram R, et al. Adverse childhood experiences and premature mortality through mid-adulthood: a five-decade prospective study. Lancet Reg Health - Am. 2022;15:100349.
Wang Y-X, Sun Y, Missmer SA, Rexrode KM, Roberts AL, Chavarro JE, et al. Association of early life physical and sexual abuse with premature mortality among female nurses: prospective cohort study. BMJ. 2023;381: e073613.
Article PubMed PubMed Central Google Scholar
Rogers NT, Power C, Pereira SMP. Child maltreatment, early life socioeconomic disadvantage and all-cause mortality in mid-adulthood: findings from a prospective British birth cohort. BMJ Open. 2021;11: e050914.
Hardcastle K, Bellis MA, Sharp CA, Hughes K. Exploring the health and service utilisation of general practice patients with a history of adverse childhood experiences (ACEs): an observational study using electronic health records. BMJ Open. 2020;10: e036239.
Ho ISS, Azcoaga-Lorenzo A, Akbari A, Davies J, Khunti K, Kadam UT, et al. Measuring multimorbidity in research: Delphi consensus study. BMJ Med. 2022;1:e000247.
Barnett K, Mercer SW, Norbury M, Watt G, Wyke S, Guthrie B. Epidemiology of multimorbidity and implications for health care, research, and medical education: a cross-sectional study. Lancet Lond Engl. 2012;380:37–43.
Chowdhury SR, Das DC, Sunna TC, Beyene J, Hossain A. Global and regional prevalence of multimorbidity in the adult population in community settings: a systematic review and meta-analysis. eClinicalMedicine. 2023;57:101860.
Noël PH, Chris Frueh B, Larme AC, Pugh JA. Collaborative care needs and preferences of primary care patients with multimorbidity. Health Expect. 2005;8:54–63.
Chau E, Rosella LC, Mondor L, Wodchis WP. Association between continuity of care and subsequent diagnosis of multimorbidity in Ontario, Canada from 2001–2015: a retrospective cohort study. PLoS ONE. 2021;16: e0245193.
Article CAS PubMed PubMed Central Google Scholar
Nicholson K, Liu W, Fitzpatrick D, Hardacre KA, Roberts S, Salerno J, et al. Prevalence of multimorbidity and polypharmacy among adults and older adults: a systematic review. Lancet Healthy Longev. 2024;5:e287–96.
Albreht T, Dyakova M, Schellevis FG, Van den Broucke S. Many diseases, one model of care? J Comorbidity. 2016;6:12–20.
Soley-Bori M, Ashworth M, Bisquera A, Dodhia H, Lynch R, Wang Y, et al. Impact of multimorbidity on healthcare costs and utilisation: a systematic review of the UK literature. Br J Gen Pract. 2020;71:e39-46.
World Health Organization (WHO). Ageing and health. 2022. https://www.who.int/news-room/fact-sheets/detail/ageing-and-health . Accessed 23 Apr 2024.
Franke HA. Toxic stress: effects, prevention and treatment. Children. 2014;1:390–402.
Parade SH, Huffhines L, Daniels TE, Stroud LR, Nugent NR, Tyrka AR. A systematic review of childhood maltreatment and DNA methylation: candidate gene and epigenome-wide approaches. Transl Psychiatry. 2021;11:1–33.
Ridout KK, Levandowski M, Ridout SJ, Gantz L, Goonan K, Palermo D, et al. Early life adversity and telomere length: a meta-analysis. Mol Psychiatry. 2018;23:858–71.
Elwenspoek MMC, Kuehn A, Muller CP, Turner JD. The effects of early life adversity on the immune system. Psychoneuroendocrinology. 2017;82:140–54.
Danese A, Baldwin JR. Hidden wounds? Inflammatory links between childhood trauma and psychopathology. Annu Rev Psychol. 2017;68:517–44.
Brindle RC, Pearson A, Ginty AT. Adverse childhood experiences (ACEs) relate to blunted cardiovascular and cortisol reactivity to acute laboratory stress: a systematic review and meta-analysis. Neurosci Biobehav Rev. 2022;134: 104530.
Teicher MH, Samson JA, Anderson CM, Ohashi K. The effects of childhood maltreatment on brain structure, function and connectivity. Nat Rev Neurosci. 2016;17:652–66.
McLaughlin KA, Weissman D, Bitrán D. Childhood adversity and neural development: a systematic review. Annu Rev Dev Psychol. 2019;1:277–312.
Koyama Y, Fujiwara T, Murayama H, Machida M, Inoue S, Shobugawa Y. Association between adverse childhood experiences and brain volumes among Japanese community-dwelling older people: findings from the NEIGE study. Child Abuse Negl. 2022;124: 105456.
Antoniou G, Lambourg E, Steele JD, Colvin LA. The effect of adverse childhood experiences on chronic pain and major depression in adulthood: a systematic review and meta-analysis. Br J Anaesth. 2023;130:729–46.
Huang H, Yan P, Shan Z, Chen S, Li M, Luo C, et al. Adverse childhood experiences and risk of type 2 diabetes: a systematic review and meta-analysis. Metabolism. 2015;64:1408–18.
Lopes S, Hallak JEC, de Machado Sousa JP, de Osório F L. Adverse childhood experiences and chronic lung diseases in adulthood: a systematic review and meta-analysis. Eur J Psychotraumatology. 2020;11:1720336.
Hu Z, Kaminga AC, Yang J, Liu J, Xu H. Adverse childhood experiences and risk of cancer during adulthood: a systematic review and meta-analysis. Child Abuse Negl. 2021;117: 105088.
Tan M, Mao P. Type and dose-response effect of adverse childhood experiences in predicting depression: a systematic review and meta-analysis. Child Abuse Negl. 2023;139: 106091.
Zhang L, Zhao N, Zhu M, Tang M, Liu W, Hong W. Adverse childhood experiences in patients with schizophrenia: related factors and clinical implications. Front Psychiatry. 2023;14:1247063.
Emsley E, Smith J, Martin D, Lewis NV. Trauma-informed care in the UK: where are we? A qualitative study of health policies and professional perspectives. BMC Health Serv Res. 2022;22:1164.
ROBINS-E Development Group (Higgins J, Morgan R, Rooney A, Taylor K, Thayer K, Silva R, Lemeris C, Akl A, Arroyave W, Bateson T, Berkman N, Demers P, Forastiere F, Glenn B, Hróbjartsson A, Kirrane E, LaKind J, Luben T, Lunn R, McAleenan A, McGuinness L, Meerpohl J, Mehta S, Nachman R, Obbagy J, O’Connor A, Radke E, Savović J, Schubauer-Berigan M, Schwingl P, Schunemann H, Shea B, Steenland K, Stewart T, Straif K, Tilling K, Verbeek V, Vermeulen R, Viswanathan M, Zahm S, Sterne J). Risk Of Bias In Non-randomized Studies - of Exposure (ROBINS-E). Launch version, 20 June 2023. https://www.riskofbias.info/welcome/robins-e-tool . Accessed 20 Jul 2023.
Balduzzi S, Rücker G, Schwarzer G. How to perform a meta-analysis with R: a practical tutorial. Evid Based Ment Health. 2019;22:153–60.
Schwarzer G, Chemaitelly H, Abu-Raddad LJ, Rücker G. Seriously misleading results using inverse of Freeman-Tukey double arcsine transformation in meta-analysis of single proportions. Res Synth Methods. 2019;10:476–83.
Crippa A, Orsini N. Multivariate dose-response meta-analysis: the dosresmeta R Package. J Stat Softw. 2016;72:1–15.
Greenland S, Longnecker MP. Methods for trend estimation from summarized dose-response data, with applications to meta-analysis. Am J Epidemiol. 1992;135:1301–9.
Shim SR, Lee J. Dose-response meta-analysis: application and practice using the R software. Epidemiol Health. 2019;41: e2019006.
Arshadipour A, Thorand B, Linkohr B, Rospleszcz S, Ladwig K-H, Heier M, et al. Impact of prenatal and childhood adversity effects around World War II on multimorbidity: results from the KORA-Age study. BMC Geriatr. 2022;22:115.
Atkinson L, Joshi D, Raina P, Griffith LE, MacMillan H, Gonzalez A. Social engagement and allostatic load mediate between adverse childhood experiences and multimorbidity in mid to late adulthood: the Canadian Longitudinal Study on Aging. Psychol Med. 2021;53(4):1–11.
Chandrasekar R, Lacey RE, Chaturvedi N, Hughes AD, Patalay P, Khanolkar AR. Adverse childhood experiences and the development of multimorbidity across adulthood—a national 70-year cohort study. Age Ageing. 2023;52:afad062.
Cromer KR, Sachs-Ericsson N. The association between childhood abuse, PTSD, and the occurrence of adult health problems: moderation via current life stress. J Trauma Stress. 2006;19:967–71.
England-Mason G, Casey R, Ferro M, MacMillan HL, Tonmyr L, Gonzalez A. Child maltreatment and adult multimorbidity: results from the Canadian Community Health Survey. Can J Public Health. 2018;109:561–72.
Godin O, Leboyer M, Laroche DG, Aubin V, Belzeaux R, Courtet P, et al. Childhood maltreatment contributes to the medical morbidity of individuals with bipolar disorders. Psychol Med. 2023;53(15):1–9.
Hanlon P, McCallum M, Jani BD, McQueenie R, Lee D, Mair FS. Association between childhood maltreatment and the prevalence and complexity of multimorbidity: a cross-sectional analysis of 157,357 UK Biobank participants. J Comorbidity. 2020;10:2235042X1094434.
Henchoz Y, Seematter-Bagnoud L, Nanchen D, Büla C, von Gunten A, Démonet J-F, et al. Childhood adversity: a gateway to multimorbidity in older age? Arch Gerontol Geriatr. 2019;80:31–7.
Hosang GM, Fisher HL, Uher R, Cohen-Woods S, Maughan B, McGuffin P, et al. Childhood maltreatment and the medical morbidity in bipolar disorder: a case–control study. Int J Bipolar Disord. 2017;5:30.
Hosang GM, Fisher HL, Hodgson K, Maughan B, Farmer AE. Childhood maltreatment and adult medical morbidity in mood disorders: comparison of unipolar depression with bipolar disorder. Br J Psychiatry. 2018;213:645–53.
Lin L, Wang HH, Lu C, Chen W, Guo VY. Adverse childhood experiences and subsequent chronic diseases among middle-aged or older adults in China and associations with demographic and socioeconomic characteristics. JAMA Netw Open. 2021;4: e2130143.
Mendizabal A, Nathan CL, Khankhanian P, Anto M, Clyburn C, Acaba-Berrocal A, et al. Adverse childhood experiences in patients with neurologic disease. Neurol Clin Pract. 2022. https://doi.org/10.1212/CPJ.0000000000001134 .
Noteboom A, Have MT, De Graaf R, Beekman ATF, Penninx BWJH, Lamers F. The long-lasting impact of childhood trauma on adult chronic physical disorders. J Psychiatr Res. 2021;136:87–94.
Patterson ML, Moniruzzaman A, Somers JM. Setting the stage for chronic health problems: cumulative childhood adversity among homeless adults with mental illness in Vancouver. British Columbia BMC Public Health. 2014;14:350.
Post RM, Altshuler LL, Leverich GS, Frye MA, Suppes T, McElroy SL, et al. Role of childhood adversity in the development of medical co-morbidities associated with bipolar disorder. J Affect Disord. 2013;147:288–94.
Reyes-Ortiz CA. Racial discrimination and multimorbidity among older adults in Colombia: a national data analysis. Prev Chronic Dis. 2023;20:220360.
Sheikh MA. Coloring of the past via respondent’s current psychological state, mediation, and the association between childhood disadvantage and morbidity in adulthood. J Psychiatr Res. 2018;103:173–81.
Sinnott C, Mc Hugh S, Fitzgerald AP, Bradley CP, Kearney PM. Psychosocial complexity in multimorbidity: the legacy of adverse childhood experiences. Fam Pract. 2015;32:269–75.
Sosnowski DW, Feder KA, Astemborski J, Genberg BL, Letourneau EJ, Musci RJ, et al. Adverse childhood experiences and comorbidity in a cohort of people who have injected drugs. BMC Public Health. 2022;22:986.
Stapp EK, Williams SC, Kalb LG, Holingue CB, Van Eck K, Ballard ED, et al. Mood disorders, childhood maltreatment, and medical morbidity in US adults: an observational study. J Psychosom Res. 2020;137: 110207.
Tomasdottir MO, Sigurdsson JA, Petursson H, Kirkengen AL, Krokstad S, McEwen B, et al. Self reported childhood difficulties, adult multimorbidity and allostatic load. A cross-sectional analysis of the Norwegian HUNT study. PloS One. 2015;10:e0130591.
Vásquez E, Quiñones A, Ramirez S, Udo T. Association between adverse childhood events and multimorbidity in a racial and ethnic diverse sample of middle-aged and older adults. Innov Aging. 2019;3:igz016.
Yang L, Hu Y, Silventoinen K, Martikainen P. Childhood adversity and trajectories of multimorbidity in mid-late life: China health and longitudinal retirement study. J Epidemiol Community Health. 2021;75:593–600.
Zak-Hunter L, Carr CP, Tate A, Brustad A, Mulhern K, Berge JM. Associations between adverse childhood experiences and stressful life events and health outcomes in pregnant and breastfeeding women from diverse racial and ethnic groups. J Womens Health. 2023;32:702–14.
Zheng X, Cui Y, Xue Y, Shi L, Guo Y, Dong F, et al. Adverse childhood experiences in depression and the mediating role of multimorbidity in mid-late life: A nationwide longitudinal study. J Affect Disord. 2022;301:217–24.
Liu M, Luong L, Lachaud J, Edalati H, Reeves A, Hwang SW. Adverse childhood experiences and related outcomes among adults experiencing homelessness: a systematic review and meta-analysis. Lancet Public Health. 2021;6:e836–47.
Petruccelli K, Davis J, Berman T. Adverse childhood experiences and associated health outcomes: a systematic review and meta-analysis. Child Abuse Negl. 2019;97: 104127.
Bethell CD, Carle A, Hudziak J, Gombojav N, Powers K, Wade R, et al. Methods to assess adverse childhood experiences of children and families: toward approaches to promote child well-being in policy and practice. Acad Pediatr. 2017;17(7 Suppl):S51-69.
Bernstein DP, Stein JA, Newcomb MD, Walker E, Pogge D, Ahluvalia T, et al. Development and validation of a brief screening version of the Childhood Trauma Questionnaire. Child Abuse Negl. 2003;27:169–90.
Kim K, Yaffe K, Rehkopf DH, Zheng Y, Nannini DR, Perak AM, et al. Association of adverse childhood experiences with accelerated epigenetic aging in midlife. JAMA Network Open. 2023;6:e2317987.
Jain P, Binder A, Chen B, Parada H, Gallo LC, Alcaraz J, et al. The association of epigenetic age acceleration and multimorbidity at age 90 in the Women’s Health Initiative. J Gerontol A Biol Sci Med Sci. 2023;78:2274–81.
Zang JCS, May C, Hellwig B, Moser D, Hengstler JG, Cole S, et al. Proteome analysis of monocytes implicates altered mitochondrial biology in adults reporting adverse childhood experiences. Transl Psychiatry. 2023;13:31.
Mau T, Blackwell TL, Cawthon PM, Molina AJA, Coen PM, Distefano G, et al. Muscle mitochondrial bioenergetic capacities are associated with multimorbidity burden in older adults: the Study of Muscle, Mobility and Aging (SOMMA). J Gerontol A Biol Sci Med Sci. 2024;79(7):glae101.
Friedman E, Shorey C. Inflammation in multimorbidity and disability: an integrative review. Health Psychol Off J Div Health Psychol Am Psychol Assoc. 2019;38:791–801.
Google Scholar
Satinsky EN, Kakuhikire B, Baguma C, Rasmussen JD, Ashaba S, Cooper-Vince CE, et al. Adverse childhood experiences, adult depression, and suicidal ideation in rural Uganda: a cross-sectional, population-based study. PLoS Med. 2021;18: e1003642.
Amene EW, Annor FB, Gilbert LK, McOwen J, Augusto A, Manuel P, et al. Prevalence of adverse childhood experiences in sub-Saharan Africa: a multicounty analysis of the Violence Against Children and Youth Surveys (VACS). Child Abuse Negl. 2023;150:106353.
Reuben A, Moffitt TE, Caspi A, Belsky DW, Harrington H, Schroeder F, et al. Lest we forget: comparing retrospective and prospective assessments of adverse childhood experiences in the prediction of adult health. J Child Psychol Psychiatry. 2016;57:1103–12.
Naicker SN, Norris SA, Mabaso M, Richter LM. An analysis of retrospective and repeat prospective reports of adverse childhood experiences from the South African Birth to Twenty Plus cohort. PLoS ONE. 2017;12: e0181522.
Walsh D, McCartney G, Smith M, Armour G. Relationship between childhood socioeconomic position and adverse childhood experiences (ACEs): a systematic review. J Epidemiol Community Health. 2019;73:1087–93.
Ingram E, Ledden S, Beardon S, Gomes M, Hogarth S, McDonald H, et al. Household and area-level social determinants of multimorbidity: a systematic review. J Epidemiol Community Health. 2021;75:232–41.
Darin-Mattsson A, Fors S, Kåreholt I. Different indicators of socioeconomic status and their relative importance as determinants of health in old age. Int J Equity Health. 2017;16:173.
Bateson K, McManus M, Johnson G. Understanding the use, and misuse, of Adverse Childhood Experiences (ACEs) in trauma-informed policing. Police J. 2020;93:131–45.
Webb NJ, Miller TL, Stockbridge EL. Potential effects of adverse childhood experiences on school engagement in youth: a dominance analysis. BMC Public Health. 2022;22:2096.
Stewart-Tufescu A, Struck S, Taillieu T, Salmon S, Fortier J, Brownell M, et al. Adverse childhood experiences and education outcomes among adolescents: linking survey and administrative data. Int J Environ Res Public Health. 2022;19:11564.
Frederick J, Spratt T, Devaney J. Adverse childhood experiences and social work: relationship-based practice responses. Br J Soc Work. 2021;51:3018–34.
University of California ACEs Aware Family Resilience Network (UCAAN). acesaware.org. ACEs Aware. https://www.acesaware.org/about/ . Accessed 6 Oct 2023.
Watson CR, Young-Wolff KC, Negriff S, Dumke K, DiGangi M. Implementation and evaluation of adverse childhood experiences screening in pediatrics and obstetrics settings. Perm J. 2024;28:180–7.
Gordon JB, Felitti VJ. The importance of screening for adverse childhood experiences (ACE) in all medical encounters. AJPM Focus. 2023;2: 100131.
Finkelhor D. Screening for adverse childhood experiences (ACEs): Cautions and suggestions. Child Abuse Negl. 2018;85:174–9.
Cibralic S, Alam M, Mendoza Diaz A, Woolfenden S, Katz I, Tzioumi D, et al. Utility of screening for adverse childhood experiences (ACE) in children and young people attending clinical and healthcare settings: a systematic review. BMJ Open. 2022;12: e060395.
Gentry SV, Paterson BA. Does screening or routine enquiry for adverse childhood experiences (ACEs) meet criteria for a screening programme? A rapid evidence summary. J Public Health Oxf Engl. 2022;44:810–22.
Article CAS Google Scholar
Morgan CA, Chang Y-H, Choy O, Tsai M-C, Hsieh S. Adverse childhood experiences are associated with reduced psychological resilience in youth: a systematic review and meta-analysis. Child Basel Switz. 2021;9:27.
Narayan AJ, Lieberman AF, Masten AS. Intergenerational transmission and prevention of adverse childhood experiences (ACEs). Clin Psychol Rev. 2021;85: 101997.
VanBronkhorst SB, Abraham E, Dambreville R, Ramos-Olazagasti MA, Wall M, Saunders DC, et al. Sociocultural risk and resilience in the context of adverse childhood experiences. JAMA Psychiat. 2024;81:406–13.
Wilson JM, Jungner G. Principles and practice of screening for disease. World Health Organisation; 1968.
Huo Y, Couzner L, Windsor T, Laver K, Dissanayaka NN, Cations M. Barriers and enablers for the implementation of trauma-informed care in healthcare settings: a systematic review. Implement Sci Commun. 2023;4:49.
Foo KM, Sundram M, Legido-Quigley H. Facilitators and barriers of managing patients with multiple chronic conditions in the community: a qualitative study. BMC Public Health. 2020;20:273.
Download references
Acknowledgements
The authors thank the members of the CAPE CPAG patient and public involvement group for providing insights gained from relevant lived experiences.
The authors are members of the Advanced Pain Discovery Platform (APDP) supported by UK Research & Innovation (UKRI), Versus Arthritis, and Eli Lilly. DS is a fellow on the Multimorbidity Doctoral Training Programme for Health Professionals, which is supported by the Wellcome Trust [223499/Z/21/Z]. BT, BS, and LC are supported by an APDP grant as part of the Partnership for Assessment and Investigation of Neuropathic Pain: Studies Tracking Outcomes, Risks and Mechanisms (PAINSTORM) consortium [MR/W002388/1]. TH and LC are supported by an APDP grant as part of the Consortium Against Pain Inequality [MR/W002566/1]. The funding bodies had no role in study design, data collection/analysis/interpretation, report writing, or the decision to submit the manuscript for publication.
Author information
Authors and affiliations.
Chronic Pain Research Group, Division of Population Health & Genomics, School of Medicine, University of Dundee, Ninewells Hospital, Dundee, DD1 9SY, UK
Dhaneesha N. S. Senaratne, Bhushan Thakkar, Blair H. Smith & Lesley A. Colvin
Institute of Academic Anaesthesia, Division of Systems Medicine, School of Medicine, University of Dundee, Dundee, UK
Tim G. Hales
School of Health Sciences, University of Dundee, Dundee, UK
Louise Marryat
You can also search for this author in PubMed Google Scholar
Contributions
DS and LC contributed to review conception and design. DC, BT, BS, TH, LM, and LC contributed to search strategy design. DS and BT contributed to study selection and data extraction, with input from LC. DS and BT accessed and verified the underlying data. DS conducted the meta-analyses, with input from BT, BS, TH, LM, and LC. DS drafted the manuscript, with input from DC, BT, BS, TH, LM, and LC. DC, BT, BS, TH, LM, and LC read and approved the final manuscript.
Corresponding author
Correspondence to Dhaneesha N. S. Senaratne .
Ethics declarations
Ethics approval and consent to participate.
Not applicable.
Consent for publication
Competing interests.
The authors declare that they have no competing interests.
Additional information
Publisher’s note.
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
12916_2024_3505_moesm1_esm.docx.
Additional File 1: Tables S1-S5 and Figures S1-S2. Table S1: Search strategy, Table S2: Characteristics of studies included in the systematic review, Table S3: Risk of bias assessment (ROBINS-E), Table S4: Exposure details (adverse childhood experiences), Table S5: Outcome details (multimorbidity), Figure S1: Meta-analysis of prevalence of exposure to ≥4 adverse childhood experiences, Figure S2: Dose-response meta-analysis of the relationship between adverse childhood experiences and multimorbidity (using a non-linear/restricted cubic spline model).
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/ . The Creative Commons Public Domain Dedication waiver ( http://creativecommons.org/publicdomain/zero/1.0/ ) applies to the data made available in this article, unless otherwise stated in a credit line to the data.
Reprints and permissions
About this article
Cite this article.
Senaratne, D.N.S., Thakkar, B., Smith, B.H. et al. The impact of adverse childhood experiences on multimorbidity: a systematic review and meta-analysis. BMC Med 22 , 315 (2024). https://doi.org/10.1186/s12916-024-03505-w
Download citation
Received : 01 December 2023
Accepted : 14 June 2024
Published : 15 August 2024
DOI : https://doi.org/10.1186/s12916-024-03505-w
Share this article
Anyone you share the following link with will be able to read this content:
Sorry, a shareable link is not currently available for this article.
Provided by the Springer Nature SharedIt content-sharing initiative
- Adverse childhood experiences
- Childhood adversity
- Chronic disease
- Long-term conditions
- Multimorbidity
BMC Medicine
ISSN: 1741-7015
- Submission enquiries: [email protected]
- General enquiries: [email protected]

IMAGES
COMMENTS
We're going to go ahead to patient case No. 1. This is a 27-year-old woman who's presented for evaluation of a complex depressive syndrome. She has not benefitted from 2 recent trials of antidepressants—sertraline and escitalopram. This is her third lifetime depressive episode.
The Role of Case Studies in Advancing Knowledge about Bipolar Disorder. Case studies, like Susan's, play a significant role in advancing knowledge about bipolar disorder. They provide unique insights into individual experiences, treatment outcomes, and the wider impact of the condition. Researchers and healthcare providers can learn from ...
This is an interesting case, as we take a look at this 23-year-old female who first comes in to see her psychiatrist with moderate depressive symptoms. At the time of the interview, her chief complaint included feeling like she's lacking energy, she's feeling depressed. She's also reporting difficulty in paying attention, organizing her ...
CASE STUDY Gary (bipolar disorder) Case Study Details. Gary is a 19-year-old who withdrew from college after experiencing a manic episode during which he was brought to the attention of the Campus Police ("I took the responsibility to pull multiple fire alarms in my dorm to ensure that they worked, given the life or death nature of fires"). ...
Case Study Details. Sarah is a 42-year-old married woman who has a long history of both depressive and hypomanic episodes. Across the years she has been variable diagnoses as having major depression, borderline personality disorder, and most recently, bipolar disorder.
A 30-year-old man has taken short-term disability leave from work due to the progression of a depressive episode. He received a diagnosis of bipolar I disorder about 10 years ago. He had his first episode of mania at the age of 20 and 2 subsequent episodes of mania between the ages of 21 and 29. He was treated with lithium, which was highly ...
The terms "soft bipolar" or "bipolar spectrum" were first proposed by Akiskal and Mallya ( 4) to describe psychopathological states that could not be easily diagnosed. It has been reported that soft bipolar cases may be prevalent up to 5.1%-23.7% ( 5 ). Cyclothymia and unspecified type of bipolar disorder are suggested to be present ...
Presents a case report of a 30-year-old married Caucasian woman, presented to our university clinic seeking a new psychiatrist to manage her bipolar illness. She had moved to the Southeast due to her husband's job relocation three months ago, and had few social contacts in her new city. She reported emerging from the depths of a severe major depressive episode one year ago and since then had ...
Bipolar disorder (BD) is a severe mental disorder with a lifelong prevalence rate of 1 %-1.5 % ... Data from this single case study suggest that MCT might be a feasible and potentially effective approach to BD. However, more studies are needed to investigate this effect further.
Chapter 5 covers the psychiatric treatment of bipolar disorder, including a case history, key principles, assessment strategy, differential diagnosis, case formulation, treatment planning, nonspecific factors in treatment, potential treatment obstacles, ethical considerations, common mistakes to avoid in treatment, and relapse prevention.
Bipolar disorder (BD) is a severe mental disorder with a lifelong prevalence rate of 1 %-1.5 %. Until now medication and self-management and cognitive behaviour therapy (CBT) has been the most documented treatment approaches to (BD).. Metacognitive therapy (MCT) is a transdiagnostic approach not yet tested on BD • In an AB single case design three patients diagnosed with BD 2 received 7-12 ...
Study replication6 found similar lifetime prevalence rates for BD-I (1.0%) and BD-II (1.1%) among men and women. Subthreshold symptoms of hypomania (bipolar spectrum disorder) were more common, with prevalence rate estimates of 2.4%.6 Incidence rates, which largely focus on BD-I, have been estimated at approximately 6.1 per 100 000 person years ...
According to the diagnostic criteria of DSM -5, symptoms. of a depressive episode include depressed mood, sign ificant. changes in sleep patterns and appetite, psychomoto r agitation or ...
This study used a single case study approach and was qualitative in nature. A patient with bipolar affective disorder without psychotic symptoms participated in the trial. A case history form and a mental state assessment instrument were used to gather the data, which was then analysed using the content analysis approach. ... The article, A ...
Case Study Details Richard is a 62-year-old single man who says that his substance dependence and his bipolar disorder both emerged in his late teens. He says that he started to drink to "feel better" when his episodes of depression made it hard for him to interact with his peers.
se Report on Bipolar Afective Disorder: Mania with Psychotic SymptomsInvestIgAtIonsBlood investigation findings showed: serum creatinine—0.75 mg/dL, serum urea—15 mg/dL, seru. sodium—142 mEq/dL, serum potassium—5.1 mEq/dL, and serum chloride—101 mEq/dL. She underwent special investigation such as psychometric assessment—young mania ...
Early-onset Bipolar Disorder. Studies have shown that bipolar disorder usually begins with an index episode of depression: positive family history (Pavuluri, Birmaher, & Naylor, 2005), clinical severity, psychotic symptoms, and psychomotor retardation are well documented predictors of bipolarity.Approximately 20% of youths with a first major depressive episode will develop a manic episode.
In a longitudinal study of 32 patients at early symptomatic stages of AD, the baseline topography of tau PET signal predicted subsequent atrophy on MRI at the single patient level, independent of baseline cortical thickness . This correlation was strongest in early-onset AD patients, who also tended to have higher tau signal and more rapid ...
The study, led by the Psychiatric Genomics Consortium bipolar disorder working group, is published in Nature Genetics . The Psychiatric Genomics Consortium is a global collaborative effort consisting of more than 800 investigators, including researchers in the National Institute of Mental Health (NIMH) Intramural Research Program and extramural ...
Bipolar disorder is a complex and heritable psychiatric condition that affects millions of people worldwide. This article reviews the current knowledge on the genetic factors that contribute to bipolar disorder, and discusses the challenges and opportunities for future research. The article also explores the implications of genetic findings for the diagnosis, treatment and prevention of ...
sing mania since medical management is different based on its etiology. Herein, we report a case of a manic episode in a middle-aged female with a prolonged history of BD who received a recent diagnosis of MS 1 year ago. Patient Concerns: A 56-year-old female presented with an episode of mania and psychosis while receiving a phenobarbital taper for chronic lorazepam use. She had a prolonged ...
Abstract. When a patient suffering from bipolar II disorder is misdiagnosed as experiencing unipolar depression, the recommended treatment of the latter may precipitate a hypomanic or manic episode. Unchecked hypomanic symptoms may include risky behaviors, through which a patient could sustain irreparable damage to relationships, careers, and ...
1 Introduction. Bipolar II disorder is marked by significant emotional and psychological distress, characterized by periods of depressive episodes and hypomania ().This condition not only affects an individual's psychological well-being but also has profound implications on their social and occupational functioning ().The complexity of Bipolar II disorder, especially with treatment-resistant ...
Patients 49 with depression, DBDs, and EDs had higher initiation rates (76.5%, 82.1%, and 100%, 50 respectively) for psychiatric treatment compared to those with GAD and SA (55.3% and 61.5%). 51 Nearly 15% of patients did not initiate treatment for SI. 52 Limitations: This retrospective study has a limited sample size in a single institution ...
problems. This study used a single case study approach and was qualitative in nature. A patient with bipolar affective disorder without psychotic symptoms participated in the trial. A case history form and a mental state assessment instrument were used to gather the data, which was then analysed using the content analysis approach.
Background Adverse childhood experiences (ACEs) have been implicated in the aetiology of a range of health outcomes, including multimorbidity. In this systematic review and meta-analysis, we aimed to identify, synthesise, and quantify the current evidence linking ACEs and multimorbidity. Methods We searched seven databases from inception to 20 July 2023: APA PsycNET, CINAHL Plus, Cochrane ...