
Presentations made painless
- Get Premium

100 Cosmetology Essay Topic Ideas & Examples
Inside This Article
Cosmetology is a diverse and exciting field that offers many different career paths and opportunities for individuals who are passionate about beauty, fashion, and personal care. If you are studying cosmetology or considering a career in the industry, you may be required to write essays on various topics related to this field. To help you get started, here are 100 cosmetology essay topic ideas and examples to inspire your writing:
- The history and evolution of cosmetology.
- The role of a cosmetologist in society.
- The importance of professionalism in the cosmetology industry.
- The impact of social media on beauty standards.
- The benefits of pursuing a career in cosmetology.
- The challenges faced by cosmetologists and how to overcome them.
- The influence of cultural diversity on beauty trends.
- The ethical considerations in the cosmetology industry.
- The impact of technology on modern cosmetology practices.
- The psychological effects of beauty treatments on individuals.
- The role of cosmetology in enhancing self-esteem and confidence.
- The relationship between fashion and cosmetology.
- The significance of continuing education in the cosmetology field.
- The potential health risks associated with certain beauty treatments.
- The impact of natural and organic products on the cosmetology industry.
- The role of cosmetology in the entertainment industry.
- The challenges faced by male cosmetologists in a female-dominated industry.
- The importance of sanitation and hygiene in cosmetology practices.
- The impact of beauty pageants on societal beauty standards.
- The rise of eco-friendly and sustainable practices in cosmetology.
- The influence of celebrities on beauty trends and consumer behavior.
- The role of cosmetology in promoting inclusivity and diversity.
- The cultural significance of different hairstyles and haircare practices.
- The impact of cosmetology on the environment and ways to reduce it.
- The benefits of natural skincare products in cosmetology.
- The role of cosmetology in therapeutic and wellness treatments.
- The importance of customer service skills in the cosmetology industry.
- The impact of social media influencers on cosmetology trends.
- The role of cosmetology in preparing individuals for special events.
- The challenges faced by independent cosmetologists and salon owners.
- The influence of different cultures on makeup trends.
- The significance of hair color and its impact on personal style.
- The impact of mass production on the cosmetology industry.
- The role of cosmetology in empowering individuals to express themselves.
- The challenges faced by cosmetologists working with diverse hair types.
- The role of cosmetology in promoting mental and emotional well-being.
- The impact of beauty standards on body image and self-esteem.
- The importance of proper skincare routines and products.
- The influence of the fashion industry on cosmetology trends.
- The significance of haircuts and hairstyles in personal identity.
- The challenges faced by cosmetologists working with different skin types.
- The role of cosmetology in preparing individuals for job interviews.
- The impact of beauty advertisements on consumer behavior.
- The importance of teamwork in a salon or spa environment.
- The influence of different eras and time periods on cosmetology trends.
- The significance of nail care and the art of nail design.
- The challenges faced by cosmetologists working with aging clients.
- The role of cosmetology in preparing individuals for weddings and special occasions.
- The impact of social media filters and editing apps on beauty standards.
- The importance of haircare routines and products for maintaining healthy hair.
- The influence of different cultures on skincare rituals and practices.
- The significance of eyebrow shaping and its impact on facial features.
- The challenges faced by cosmetologists working with clients with special needs.
- The role of cosmetology in promoting gender equality and inclusivity.
- The impact of beauty treatments on the environment and ways to minimize it.
- The importance of proper makeup application techniques.
- The influence of different art forms on cosmetology trends.
- The significance of hair extensions and their impact on personal style.
- The challenges faced by cosmetologists working in high-stress environments.
- The role of cosmetology in preparing individuals for fashion shows and photo shoots.
- The impact of cosmetic surgery on the cosmetology industry.
- The importance of haircare routines and products for different hair textures.
- The influence of different cultures on fragrance preferences and trends.
- The significance of makeup contouring and its impact on facial structure.
- The challenges faced by cosmetologists working with clients with skin conditions.
- The role of cosmetology in promoting body positivity and self-acceptance.
- The impact of beauty salons and spas on local economies.
- The importance of proper nail care and hygiene.
- The influence of different music genres on cosmetology trends.
- The significance of hairstyling tools and their impact on hair health.
- The challenges faced by cosmetologists working with clients with allergies.
- The role of cosmetology in preparing individuals for television and film productions.
- The impact of cosmetic brands and their marketing strategies on consumer choices.
- The importance of facial skincare routines and products.
- The influence of different architectural styles on cosmetology trends.
- The significance of hair accessories and their impact on personal style.
- The challenges faced by cosmetologists working with clients with disabilities.
- The role of cosmetology in promoting environmentally friendly practices.
- The impact of beauty competitions on the cosmetology industry.
- The importance of proper body skincare routines and products.
- The influence of different dance styles on cosmetology trends.
- The significance of makeup palettes and their impact on artistic expression.
- The challenges faced by cosmetologists working with clients with hair loss.
- The role of cosmetology in preparing individuals for red carpet events.
- The impact of beauty bloggers and vloggers on consumer preferences.
- The importance of hand and foot care in overall well-being.
- The influence of different literary genres on cosmetology trends.
- The significance of hair accessories and their impact on bridal looks.
- The challenges faced by cosmetologists working with clients with skin sensitivities.
- The role of cosmetology in promoting eco-friendly packaging and product options.
- The impact of beauty trends and fads on the cosmetology industry.
- The importance of proper sun protection and skincare.
- The influence of different painting techniques on cosmetology trends.
- The significance of makeup brushes and tools in achieving desired looks.
- The challenges faced by cosmetologists working with clients with medical conditions.
- The role of cosmetology in preparing individuals for theater and stage performances.
- The impact of beauty subscription boxes on consumer shopping habits.
- The importance of proper lip care and the art of lipstick application.
- The influence of different photography styles on cosmetology trends.
- The significance of skincare rituals and practices in different cultures.
These 100 cosmetology essay topic ideas and examples should give you a starting point for your writing. Remember to choose a topic that interests you and allows you to showcase your knowledge and enthusiasm for the field of cosmetology. Good luck with your essays!
Want to research companies faster?
Instantly access industry insights
Let PitchGrade do this for me
Leverage powerful AI research capabilities
We will create your text and designs for you. Sit back and relax while we do the work.
Explore More Content
- Privacy Policy
- Terms of Service
© 2024 Pitchgrade
Thank you for visiting nature.com. You are using a browser version with limited support for CSS. To obtain the best experience, we recommend you use a more up to date browser (or turn off compatibility mode in Internet Explorer). In the meantime, to ensure continued support, we are displaying the site without styles and JavaScript.
- View all journals
- ADVERTISEMENT FEATURE Advertiser retains sole responsibility for the content of this article
Recognizing the beauty of science, and the science behind beauty
Produced by

Lisa Napolione, Senior Vice President, Global Research & Development at The Estée Lauder Companies
The Estée Lauder Companies has a long history of science and innovation. Fifty years ago, the prestige beauty company created the world’s first allergy-tested, fragrance free skincare line — and it has continued to roll out transformative beauty products ever since. The Company recently partnered with Nature Research to create two new prizes designed to inspire women in science, technology, engineering or mathematics (STEM), one to honour early career female scientists making pioneering discoveries, and the other to recognize leaders — women and men — behind initiatives supporting greater equality in STEM. Biochemical engineer Lisa Napolione, who leads the Company’s R&D efforts, explains the impetus for the awards and how The Estée Lauder Companies takes a science-driven approach to skincare and beauty.
The new prizes are designed to inspire women in STEM, and focus on different things. Why put the spotlight on educators and young researchers?
Both of these areas are critical in their own right and integral to everything we do at The Estée Lauder Companies. We are a company that was founded by a pioneering woman who supported other women and who remains an inspiration to all of us — and so honouring exceptional female researchers through the Inspiring Science Award really spoke to us. We hope it not only shines a much-deserved light on the achievements of exceptional women in STEM, but also helps to establish a new generation of role models. The second award — the Innovating Science Award — recognizes a person or an organization that promotes STEM to girls and young women. I really feel strongly that young girls need role models and mentors in STEM, because without these influences we wouldn’t have the deep bench of research expertise among the next generation of scientists.
This all sounds very personal to you.
It is! I was so fortunate that early in my education, I had a mentoring role-model, Nora Kyser, who was one of the first female chemical and ceramic engineers in all of the United States. She was my high school chemistry teacher in my little hometown in western New York, and she arranged with the school district that, if she paid for her own research, she could work after hours in the school’s laboratory. She saw something in me, and hired me as her lab assistant. Her hands-on personal attention affected me so much. It was an amazing experience for which I will be forever grateful — and it inspired me to do for others what she did for me. I do what I do today because of her.
How does science inform how products are developed at The Estée Lauder Companies?
Many people in the scientific community don’t appreciate the breadth and depth of the serious science that happens in beauty. The Estée Lauder Companies’ R&D teams are constantly looking at breakthroughs in other fields of science and technology to inspire our skincare research. We conduct epigenetic research, including into sirtuins, nrf2 activation and cellular repair. Notably, we have conducted research on Nobel Prize-winning topics: Estée Lauder was the first cosmetic brand to research skin cells’ circadian rhythm and the role of ‘clock genes’ n cellular repair, and the first to research autophagy and its role in helping repair skin-cell damage. Last year, Estée Lauder R&D started mechanobiology research to help understand why and how blinking ages the look of the eyes, and now how light pollution, specifically blue light at night, desynchronizes skin cells’ natural repair.
Much of our research uses nature as the ultimate inspiration with more than 12,000 bioactive molecules in our database supported by hundreds of technical measures and assays. Plus, we have fermentation and biotechnology capabilities that allow us to produce high-performance ingredients in a multitude of organisms, including bacteria, yeast and mould. The power of fermentation can be seen in our La Mer brand, which is built around the Miracle Broth™ created from a unique three to four-month fermentation with a specific sea kelp. In all, this research has resulted in new products that can help protect against the visible effects of pollution, improve skin tone, reduce undesired pigmentation, and counteract visible skin and hair ageing. We are always looking at relevant applications in emerging scientific areas that have yet to fully materialize — because if it’s hot in science, it will be hot in beauty.
What’s an example of this process in action?
We took a multi-pronged approach for the Clinique Even Better Clinical product for improving skin tone and visibly correcting dark spots. It’s such an interesting area of science, because there are multiple pathways behind the abnormal accumulation of melanin, the pigment responsible for these discolouration issues. We explored several approaches: new antioxidant ingredients that protect against free radicals; biofermentation technologies that target melanin clusters; and exfoliating agents to get rid of the dead skin cells that contain melanin dust. This all required a very detailed understanding of the underlying biology and a degree of scientific rigour that’s absolutely necessary for us to deliver products that really work.
Quick links
- Explore articles by subject
- Guide to authors
- Editorial policies
- Cosmetics Paper Topics Topics: 58
- Chlamydia Research Topics Topics: 52
- Hepatitis Essay Topics Topics: 57
- Asthma Topics Topics: 155
- Dorothea Orem’s Theory Research Topics Topics: 85
- Patient Safety Topics Topics: 148
- Heart Attack Topics Topics: 54
- Communicable Disease Research Topics Topics: 58
- Arthritis Paper Topics Topics: 58
- Hypertension Essay Topics Topics: 155
- Breast Cancer Paper Topics Topics: 145
- Heart Disease Topics Topics: 150
- Heart Failure Essay Topics Topics: 83
- Nursing Theory Research Topics Topics: 207
- Tuberculosis Paper Topics Topics: 133
61 Cosmetology Essay Topics
🏆 best essay topics on cosmetology, 🎓 most interesting cosmetology research paper topics, 📌 easy cosmetology essay topics.
- Cosmetic Surgery: Modern Trends
- Cosmetic Surgery Advertising: Multimodal Analysis
- Negative Customer Experience in Cosmetics Store
- Use of Botulinum Toxin for Cosmetic Purposes
- Beauty, Skincare, and Cosmetics Challenges
- Cosmetic Surgery History and Effects
- The Regulation of Chemicals in Cosmetics
- Chemical Peel and Microneedling in Cosmetology
- Correcting the Form of Eyebrows: Cosmetic Eyebrow Tattooing
- Cosmetic Surgery Popularity in the Middle East
- The Evolution of Cosmetology: From Ancient Practices to Modern Techniques
- Cosmetology Education: Training the Next Generation of Beauty Professionals
- The Importance of Ergonomics in Cosmetology
- Cosmetology as an Art Form: Creativity and Expression in Beauty
- The Impact of Technology on the Future of Cosmetology
- Cosmetology and a Career as a Cosmetologist: Pros and Cons
- Major Problems of the Cosmetology Industry
- Cosmetology Careers: Exploring Diverse Paths in the Beauty Industry
- The Importance of Cosmetology in Modern Society
- Cosmetology and Cultural Influences on Beauty Standards
- The Science Behind Beauty: Understanding Cosmetology Chemistry
- Cosmetology Ethics: Professional Standards in the Beauty Industry
- An Observation of Health Hazards in Cosmetology
- The Future of Cosmetology: Emerging Technologies and Innovations
- Cosmetology Licensing and Regulations: Ensuring Quality and Safety
- Bridging the Gap: Integrating Technology into Cosmetology Education
- Cosmetology and Mental Health: The Therapeutic Role of Beauty Services
- Trends in Sustainable and Cruelty-Free Cosmetology Products
- How Cosmetology Has Changed Over Time
- Cosmetology Trends: Navigating the Dynamic World of Beauty Fashion
- The Business of Beauty: Entrepreneurship in the Cosmetology Industry
- Cosmetology and Social Media: Influencer Culture and Beauty Trends
- Barber License vs. Cosmetology License: New Opportunities and Legal Limitations
- Cosmetology and Wellness: Integrating Health and Beauty Practices
- Diversity in Cosmetology: Celebrating a Range of Beauty Representations
- Cosmetology Tools and Technology Advancements: Revolutionizing Beauty Services
- How to Start a Successful Cosmetology Business
- Cosmetology and Sustainable Practices: Eco-Friendly Approaches to Beauty
- The Intersection of Cosmetology and Fashion: Influences and Collaborations
- Cosmetology Specializations: Niche Careers in the Beauty Industry
- The Role of Cosmetology in the 21st Century
- Cosmetology and Client Communication: Building Trust and Relationships
- Black Women Who Changed the Cosmetology World Forever
- Cosmetology and the Psychology of Beauty: Understanding Client Needs
- Innovations in Cosmetology Products: Enhancing Beauty Care Solutions
- Beyond the Salon: The Business Side of Cosmetology Entrepreneurship
- Cosmetology vs. Cosmetic Science: What’s the Difference?
- Top Cosmetology Skills to Master and How to Highlight Them
- Cosmetology and Aging: Beauty Practices for Different Life Stages
- Exploring the Pros and Cons: Is a Career in Cosmetology Worth It?
- Cosmetology and Global Beauty Traditions: Cultural Exchange in Aesthetics
- Beauty, Confidence, and the Significance of Cosmetology
- Cosmetology and Body Positivity: Redefining Beauty Standards
- The History of Cosmetology Schools: Shaping Beauty Education
- Cosmetology and Dermatology: Intersections in Skin and Hair Care
- The Influence of Cosmetology on Beauty Pageant Culture
- Cosmetology and Marketing: Promoting Beauty Services in the Digital Age
- What Skills Are Needed for Cosmetology?
- Cosmetology Career Goals: Pursuing Beauty and Creativity
- The Art of Cosmetology: Exploring Techniques and Inspirations
Cite this post
- Chicago (N-B)
- Chicago (A-D)
StudyCorgi. (2024, February 17). 61 Cosmetology Essay Topics. https://studycorgi.com/ideas/cosmetology-essay-topics/
"61 Cosmetology Essay Topics." StudyCorgi , 17 Feb. 2024, studycorgi.com/ideas/cosmetology-essay-topics/.
StudyCorgi . (2024) '61 Cosmetology Essay Topics'. 17 February.
1. StudyCorgi . "61 Cosmetology Essay Topics." February 17, 2024. https://studycorgi.com/ideas/cosmetology-essay-topics/.
Bibliography
StudyCorgi . "61 Cosmetology Essay Topics." February 17, 2024. https://studycorgi.com/ideas/cosmetology-essay-topics/.
StudyCorgi . 2024. "61 Cosmetology Essay Topics." February 17, 2024. https://studycorgi.com/ideas/cosmetology-essay-topics/.
These essay examples and topics on Cosmetology were carefully selected by the StudyCorgi editorial team. They meet our highest standards in terms of grammar, punctuation, style, and fact accuracy. Please ensure you properly reference the materials if you’re using them to write your assignment.
This essay topic collection was updated on February 24, 2024 .
- News & Analysis on Cosmetics Innovation
Skin science: Top 10 most-read stories on cosmetics science and research of 2021
17-Dec-2021 - Last updated on 17-Dec-2021 at 02:30 GMT
- Email to a friend
![research topics in cosmetology Featuring the biggest cosmetic science stories of the year. [Getty Images]](https://www.cosmeticsdesign-asia.com/var/wrbm_gb_food_pharma/storage/images/_aliases/wrbm_large/publications/cosmetics/cosmeticsdesign-asia.com/article/2021/12/17/skin-science-top-10-most-read-stories-on-cosmetics-science-and-research-of-2021/13064170-1-eng-GB/Skin-science-Top-10-most-read-stories-on-cosmetics-science-and-research-of-2021.jpg)
1 – Sunscreen still needed: Protective surgical face masks ineffective against UV protection, says Kao
People still need to protect their skin from ultraviolet sun rays with sunscreen even when wearing protective surgical face masks, according to new research from Kao.
For the first experiment, researchers from the firm’s Beauty Research and Creation Center (BRCC) conducted the experiment on a model with ‘skin’ that turns purple when it comes into contact with ultraviolet (UV) rays.
A cream sunscreen with SPF 50+ and a triple plus protection grade (PA) was applied to half of the model’s face and protected with a standard non-woven surgical face mask.
As hypothesised, the skin underneath the mask that was not protected by sunscreen turned purple, indicating exposure to UV rays.
2 – Power of sandalwood: Quintis to double down on cosmetics market on the back of antioxidant effectiveness
Australian sandalwood supplier Quintis is eyeing new opportunities in the cosmetic space after a peer-reviewed study showed that it is a more potent antioxidant than vitamin E.
Quintis Sandalwood is a supplier of Indian and Australian sandalwood raw materials, including oil, powder, logs and chips.
It supplies sandalwood materials to multiple industries for use in fragrance, cosmetics, as well as incense and religious carvings.
The firm owns and manages an Indian sandalwood plantation that spans over 12,000 hectares across northern Australia and is home to more than 5.5 million trees.
The fourth iteration of Shiseido’s best-selling Ultimune serum showcases the company’s research into the co-relation between blood circulation and skin health.
The latest serum feature’s Shiseido’s The Lifeblood technology as well as new ingredients Houttuynia cordata and fermented hibiscus Extract.
Shiseido’s latest dermatological discovery, dubbed the Lifeblood Research, dove into the significance of blood circulation and its relation to skin health and its appearance.
“The latest ground-breaking Lifeblood Research involves fundamentally improving the skin by constantly enhancing blood flow. It is different from conventional skincare technology, which only deals with individual skin concerns temporarily and only on the surface level,” said Ryota Yukisada, chief brand officer of Shiseido.
4 – Smart skin: Amorepacific to use wearable device to develop cosmetics for specific environmental needs
South Korean cosmetics firm Amorepacific said it intends to use the sweat-proof wearable skin measurement device it developed with MIT to produce cosmetic products according to environmental needs.
Amorepacific announced in June that it had collaborated with Massachusetts Institute of Technology (MIT) to develop a wearable skin measuring device.
The film-like and stretchable patch is embedded with a flexible sensor. It can be stuck on the skin and used to measure the skin condition for a long period of time, withstanding sweat and remaining comfortable for the user.
“Efforts to measure the condition of human skin more precisely and stably have been carried out in various fields. However, the skin is naturally affected by various external environmental changes such as sweat, and thus, it was difficult to maintain measurement and observe changes without interruption,” said Han Jiyeon, a scientist from the Amorepacific R&D centre clinical research lab.
5 – Personalisation and skin microbiome: S. Korea’s Cosmax to develop AI-powered platform
South Korean ODM giant Cosmax is set to develop an AI-powered platform that will help both its customers and end-consumers get personalised information about the skin microbiome.
CosmeticsDesign-Asia has learnt that the commercial platform would map out its specialised technology by factors such as efficacy to aid developers in producing skin microbiome products.
To build up the system, the company has collected skin microbiome information from over 1,000 people to date and analysed the statistical significance between them.
Furthermore, the company is planning to develop a consumer-based personal skin microbiome platform as well to provide “high-quality value” to consumers.
6 – Mask-proof lip gloss? Maquillage launches new transfer-resistant product featuring Shiseido’s latest tech
Shiseido-owned make-up brand Maquillage has launched a new lip product featuring new technology that makes it transfer resistant despite its glossy finish.
Maquillage Dramatic Lip Tint debuted on October 21 with five shades that retail for JPY2,300 (U$21).
The newest product was developed in response to the increased use of protective face masks, which caused a devastating blow to lipstick sales in the past 18 months or so.
“Due to COVID-19, consumers' values and behaviours around the world are changing dramatically. Reflecting such changes there is increased awareness of health and skincare, while at the same time frequencies to use make-up like lipstick are decreasing as people go out less and wear masks,” remarked Masahiko Uotani, CEO of Shiseido, last year.
7 – A good alternate? Aussie firm eyeing opportunities for Indian sandalwood as a CBD alternative following China ban
Australian sandalwood supplier Quintis is eyeing new opportunities in China’s cosmetic space after a new scientific review revealed Indian sandalwood oil has more scientifically proven benefits than CBD oil.
Quintis Sandalwood is a supplier of Indian and Australian sandalwood raw materials, including oil, powder, logs and chips for multiple industries for use in fragrance, cosmetics, as well as incense and religious carvings.
Previously, the company told CosmeticsDesign-Asia that it has been placing more emphasis on the cosmetics side of the business, believing it could tap into the demand for natural products in the market.
Now, Quintis is looking to target the Chinese market on the back of a new review revealing that Indian sandalwood oil, or Santalum album, has more substantiated benefits than cannabidiol (CBD).
8 – Seaweed saviour: Marinova highlights ‘unmet needs’ in the market for skin microbiome-friendly atopic dermatitis treatment
There is a gap in the market for skin microbiome solutions to help treat atopic dermatitis , claims biotechnology company Marinova, for which it believes its brown seaweed extract could play a major role.
Atopic dermatitis is the most common type of eczema, a widespread condition characterised by dry and itchy skin.
While there are treatments for atopic dermatitis available in the market, Tasmania-based biotech firm Marinova believes there is a gap in the market targeting the skin microbiome.
“Broadly, atopic dermatitis is quite a complex illness where there's a multitude of factors that contribute to it… and there's an unmet need, particularly in the skin microbiome space,” said Dr Damien Stringer, operations manager, Marinova.
9 – Too complex: Chinese team questions ‘quality and value’ of Asian herb research on skin whitening
A team of researchers from two Chinese institutes have questioned the validity of the existing research undertaken on Asian herbs for skin whitening applications, concluding that the ingredients were “too complex to obtain reliable results”.
Despite being fraught with potential hazards, skin care products with whitening claims continue to thrive in the Asian beauty market because fair skin is still considered the ideal of beauty.
According to a 2019 report by Grand View Research, the global market size of skin whitening in 2018 was $8.3bn. In the largest product segment – the cream category at 53% -- China, Japan, India, Indonesia and South Korea, emerged as the top five-ranked countries in terms of sales.
The high demand for skin whitening solutions coupled with the increasing concern for product safety, has led to a raft of research into traditional Asian herbs and potential skin whitening properties.
10 – Clean sweep: Clé De Peau’s new micellar cleansing water to feature Shiseido’s new make-up removing tech
Japanese beauty giant Shiseido has developed new technology to enhance the effectiveness of micellar water that it will launch with Clé De Peau Beauté starting from June 2021.
The latest tech will be applied to Clé De Peau Beauté Micellar Cleansing Water will be rolled out in Japan in June and will be launched subsequently overseas from July.
Clé De Peau Beauté Micellar Cleansing Water will be the first product to be enhanced with the newly developed tech and Shiseido intends to develop more cleansing products with it in the future.
The impetus of the new launch was Shiseido’s development of a unique state of surfactant, the sponge phase.
Related news
![research topics in cosmetology Carbonwave uses sargassum seaweed to develop different materials for different sectors, one of which is a broad-spectrum natural emulsifier. [Getty Images]](https://www.cosmeticsdesign-asia.com/var/wrbm_gb_food_pharma/storage/images/_aliases/wrbm_medium/publications/cosmetics/cosmeticsdesign-asia.com/article/2022/08/16/carbonwave-targets-asia-s-clean-beauty-market-with-upcycled-seaweed-emulsifier/15660738-1-eng-GB/Carbonwave-targets-Asia-s-clean-beauty-market-with-upcycled-seaweed-emulsifier.jpg)
Related products

Want to bottle youth with senolytics science?
dsm-firmenich | Recorded the 21-Aug-2024 | Webinar
Results from a clinical study will be presented, revealing novel insights into the relationship between senescent cells and facial aging skin.
Register for free

Calendula Cellular Elixir: Alchemy in a cell
Content provided by Naolys | 11-Jun-2024 | Product Brochure
Get ready to experience a cellular evolution in skincare with InnerLift Calendula's advanced Plant Cell biotechnology.

Go Green, Glow Beautiful with SYMPARE MES
Content provided by KLK OLEO | 22-Mar-2024 | Product Brochure
Derived from vegetable oil, SYMPARE MES champions sustainability with a natural origin index of 0.84.
Exosomes: Passing Trend or Transformative Reality?
Content provided by Naolys | 23-Feb-2024 | Sponsored Link
Exosomes, microscopic vesicles naturally present in abundance within Plant Cells, have garnered significant attention within the scientific and cosmetic...
- Calendula Cellular Elixir: Alchemy in a cell Naolys | Download Product Brochure
On-demand webinars
- Want to bottle youth with senolytics science? dsm-firmenich
- New frontiers: Niche opportunities in beauty Webinar
![research topics in cosmetology The business developments from L'Oréal, Shiseido, Nykaa and more. [Getty Images]](https://www.cosmeticsdesign-asia.com/var/wrbm_gb_food_pharma/storage/images/_aliases/wrbm_tiny/publications/cosmetics/cosmeticsdesign-asia.com/headlines/business-financial/financial-focus-l-oreal-shiseido-nykaa-in-our-beauty-business-update/17681905-1-eng-GB/Financial-focus-L-Oreal-Shiseido-Nykaa-in-our-beauty-business-update.jpg)
Promotional features

CosmeticDesign-Asia
- Advertise with us
- Apply to reuse our content
- Press Releases – Guidelines
- Contact the Editor
- Report a technical problem
- Why Register?
- Whitelist our newsletters
- Editorial calendar
- Expert advisory panel
An official website of the United States government
The .gov means it’s official. Federal government websites often end in .gov or .mil. Before sharing sensitive information, make sure you’re on a federal government site.
The site is secure. The https:// ensures that you are connecting to the official website and that any information you provide is encrypted and transmitted securely.
- Publications
- Account settings
Preview improvements coming to the PMC website in October 2024. Learn More or Try it out now .
- Advanced Search
- Journal List

Nanotechnology in Cosmetics and Cosmeceuticals—A Review of Latest Advancements
Vaibhav gupta.
1 Nanotechnology Lab, School of Pharmaceutics Education and Research (SPER), Jamia Hamdard University, New Delhi 110062, Delhi, India; moc.oohay@nggvahbiav (V.G.); moc.liamg@eenabis (S.M.); moc.liamg@114amzu (U.F.); moc.liamg@5229vahsek (K.K.)
Sradhanjali Mohapatra
Harshita mishra.
2 Smart Society Research Team, Faculty of Business and Economics, Mendel University, 61300 Brno, Czech Republic; moc.liamg@8801arhsimatihsrah
Uzma Farooq
Keshav kumar, mohammad javed ansari.
3 Department of Pharmaceutics, College of Pharmacy, Prince Sattam Bin Abdulaziz University, Alkharj 16278, Saudi Arabia or [email protected] (M.J.A.); [email protected] (M.F.A.); [email protected] (A.S.A.)
Mohammed F. Aldawsari
Ahmed s. alalaiwe, mohd aamir mirza, zeenat iqbal, associated data.
Not applicable.
Nanotechnology has the potential to generate advancements and innovations in formulations and delivery systems. This fast-developing technology has been widely exploited for diagnostic and therapeutic purposes. Today, cosmetic formulations incorporating nanotechnology are a relatively new yet very promising and highly researched area. The application of nanotechnology in cosmetics has been shown to overcome the drawbacks associated with traditional cosmetics and also to add more useful features to a formulation. Nanocosmetics and nanocosmeceuticals have been extensively explored for skin, hair, nails, lips, and teeth, and the inclusion of nanomaterials has been found to improve product efficacy and consumer satisfaction. This is leading to the replacement of many traditional cosmeceuticals with nanocosmeceuticals. However, nanotoxicological studies on nanocosmeceuticals have raised concerns in terms of health hazards due to their potential skin penetration, resulting in toxic effects. This review summarizes various nanotechnology-based approaches being utilized in the delivery of cosmetics as well as cosmeceutical products, along with relevant patents. It outlines their benefits, as well as potential health and environmental risks. Further, it highlights the regulatory status of cosmeceuticals and analyzes the different regulatory guidelines in India, Europe, and the USA and discusses the different guidelines and recommendations issued by various regulatory authorities. Finally, this article seeks to provide an overview of nanocosmetics and nanocosmeceuticals and their applications in cosmetic industries, which may help consumers and regulators to gain awareness about the benefits as well as the toxicity related to the continuous and long-term uses of these products, thus encouraging their judicious use.
1. Introduction
Nanotechnology and nanodelivery systems are innovative areas of science that comprise the design, characterization, manufacturing, and application of materials, devices, and systems at the nanoscale level (1–100 nm). Nanotechnology, being recognized as one of the revolutionizing technologies, is extensively studied in the area of cosmetics and cosmeceuticals [ 1 , 2 ]. The incorporation of nanotechnology has led to advancements in cosmetic science, resulting in increased consumer demand throughout the world [ 3 ]. Presently, nanomaterials are attracting attention in this area, as they offer greater advantages over traditionally used cosmetic products. Further, the amalgamation of nanomaterials has greatly contributed to the global increase in the market share of pharmaceuticals and cosmetics. In the year 2019, the international market size of nanomaterials was estimated to be USD 8.5 billion and is expected to increase with up to a 13.1% compound annual growth rate from the years 2020 to 2027 [ 4 ]. Although the concept of nanomaterials (gold and silver nanoparticles) has been used in cosmetics for several years, the extensivity of applications has intensified in recent years.
Cosmetics are preparations that have been used by humans for a long time, primarily for regenerative purposes, and are appreciated by both genders. They can be defined as preparations that are typically used externally and can be formulated from a single or combination of substances obtained from either natural or artificial sources [ 5 ]. The US Food and Drug Administration (USFDA) defines cosmetics as a formulation “intended to be applied to the human body for cleansing, beautifying, promoting attractiveness, or altering the appearance without affecting the body’s structure or functions”. This expansive definition encompasses any material proposed for use as a component of a cosmetic item, although soap is explicitly excluded from this class [ 6 ]. However, under this act, the word “cosmeceutical” has no definition. As per the Federal Food Drug and Cosmetic Act (FD&C Act), there is no such word as “cosmeceutical”. This word is only used for industrial purposes to refer to cosmetic products with therapeutic actions. The European Union Cosmetics Directive (EUCD) defines cosmetics as “any substance or preparation intended to be placed in contact with the various external parts of the human body (epidermis, hair system, nails, lips and external genital organs) or with the teeth and the mucous membranes of the oral cavity with a view exclusively or mainly of cleaning them, perfuming them, changing their appearance and/or correcting body odours and/or protecting them or keeping them in good condition” [ 7 ]. The Drugs and Cosmetics Act 1940 and Rules 1945 defines a cosmetic as “any article intended to be rubbed, poured, sprinkled or sprayed on, or introduced into, or otherwise applied to the human body or any part thereof for cleansing, beautifying, promoting attractiveness, or altering the appearance, and includes any article intended for use as a component of cosmetic” [ 8 ]. Despite these definitions, the legal meaning of cosmetics in many nations is more extensive. In some Western nations, cosmetics are normally interpreted as just beautifying products, such as lipstick, mascara, eyeliners, highlighter, and a few other items of this kind [ 9 ].
Cosmeceuticals can be described as preparations that contain therapeutically active ingredients that specifically possess remedial effects upon surface application with traditionally used cosmetics. These products have quantifiable restorative effects on the skin and hair and are utilized for the treatment of different conditions, such as damaged hair, wrinkles, photoaging, skin dryness, light spots, hyperpigmentation, etc. Acting as a bridge between drugs and beauty care products, they promise an improvement in appearance [ 10 , 11 ]. Presently, cosmeceuticals are considered one of the fastest-growing segments of the personal care industry, and the market for individual consideration is massively expanding [ 2 ]. It is one of the most rapidly developing ventures, demanding an expansion in nanocosmeceuticals research, exploration, and applications.
The manipulation of materials at the atomic level by utilizing nanotechnology has great potential in the area of cosmeceuticals, opening up new avenues for the cosmetics industry. The incorporation of various nanomaterials during the development of cosmetic/cosmeceutical products results in nanocosmetics/nanocosmeceuticals, respectively. Prolongation of action, augmented bioavailability, and improved aesthetic appeal of products are a few of the advantages associated with nanotechnology-based cosmeceuticals. These products offer several other benefits over traditionally used cosmeceuticals, such as small size and huge surface-to-volume ratio, which makes them effective adjuvants in cosmeceuticals. Further, the inclusion of nanoparticles in cosmetic formulations does not change the properties of cosmeceuticals but improves their appearance, coverage, and adherence to the skin. Cosmetic manufacturers employ nanosized ingredients to improve UV protection, skin penetration, color, the release of fragrance, finish quality, anti-aging effect, and a variety of other properties. They prolong the duration of action by either controlling the delivery of active ingredients, causing site-specificity, improving biocompatibility, or enhancing the drug-loading capacity. All of these factors make them more popular among consumers, necessitating clinical trials in this area to address their safety concerns. Nanocosmeceuticals have also been highly exploited for formulating various anti-aging formulations. They are successfully marketed as skincare, hair care, and nail care products, among others, claiming to stimulate their growth, protect their structure, and increase hydration power, thus improving their effectiveness as cosmetic products [ 12 , 13 ]. Although they have several benefits, at the same time, they possess limitations related to stability, scalability, toxicity, cost, etc. Moreover, the safety and toxicity profiles of nanomaterials are still debatable. The small size, increased surface area, and positive surface charge of nanoparticles improve their ability to interact with the microenvironment biologically. On the other hand, they have dose-dependent toxicity through different routes of administration. It is well known that the bioavailability of an active ingredient is better influenced by the dosage rather than the physicochemical properties of the active moiety [ 14 ]. Hence, in the case of cosmetic products, a major concern in the advancement of nanoformulations is that they may enhance the concentration of active ingredients reaching the blood and impact the toxicity [ 15 ]. Figure 1 depicts the overall action of nanoparticles in cosmetics and cosmeceuticals.

Advantages of nanocosmeceuticals.
Based on these facts, a narrative review of all of the relevant articles and reports was conducted by searching related keywords across different sources. Google Patents was used to collect data regarding related patents. Selected studies were compared and condensed to obtain a qualitative output based on existing theories and principles. The present review outlines various nanoparticles and nanodelivery systems used for cosmetic and cosmeceutical products, highlighting their positive and negative characteristics along with related patents. It also discusses the health and environmental risks linked with nanocosmeceuticals with suggested solutions. Further, the present review highlights the regulatory scenarios and compares the various regulations related to cosmetics. Additionally, it is intended to assist the industry and other stakeholders in identifying potential safety issues associated with nanomaterials in cosmetics. It also discusses various guidelines and recommendations prescribed by different regulatory agencies. Finally, this article aims to provide an overview of nanocosmetics and nanocosmeceuticals and their applications in cosmetic industries and suggest future directions, which may help consumers and regulators to gain awareness about the benefits as well as the toxicity related to the continuous and long-term uses of these products, thus encouraging their judicious use.
2. Nanomaterials Used in Cosmetic Products
Nanomaterials are materials having at least one dimension in the nano range and significantly distinct physicochemical properties. These materials have been commonly used in the cosmetic industry for many years. Cosmetics incorporating nanomaterials show more advantages as compared to microscale cosmetics. The large surface area of these particles is responsible for their efficient transportation, absorption, bioavailability, and transparency and the sustained effect of the product. However, consideration should be given to the concentration to circumvent the associated toxicity. The following Table 1 describes different nanomaterials used in the cosmetic industry.
Different nanomaterials used for preparing cosmetics and cosmeceuticals.
| S. No. | Nanomaterial | Advantage | Disadvantage | Uniqueness | Type of Cosmeceutical | Commercially Available Product | Reference |
|---|---|---|---|---|---|---|---|
| Inorganic particles (TiO , ZnO) | Hydrophilic, biocompatible, safe, and stable | Pulmonary toxicity | Absorb/reflect UV light | Sunscreen | Phytorx UV Defense Sun Block SPF 100—Lotus Professionals | [ , ] | |
| Silica (SiO ) | Hydrophilic, ↓ manufacturing cost | Pulmonary toxicity | Used as filler to ↑ the bulk of the cosmetic formulation | Lipstick | Face FWD >> Blush Stick—Sugar Cosmetics | [ ] | |
| Carbon black | Light weight, ↑ chemical and thermal stability, and ↓ cost | Cytotoxicity; alters the phagocytic property of macrophages | Color pigment | Facemask | Face Masque—Carbon BAE | [ , ] | |
| Mascara | Mascara Black—Lakme | ||||||
| Nano-organic (tris-biphenyl triazine) | Powerful and photostable filter | Hazardous to the aquatic environment | Most efficient UVB and UVA 2 filter | Sunscreen | Extra UV Gel—Allie | [ , ] | |
| Nano-hydroxyapatite | Dental desensitizer and polish remineralization of teeth | Very brittle nature | Safe in pediatric toothpaste | Toothpaste | Kinder Karex Hydroxyapatite | [ , , ] | |
| APAGARD M plus—Sangi | |||||||
| Gold and silver nanoparticles | Uniform shape, size, and branch length; tuned pharmacokinetics and biodistribution; antibacterial and antifungal activity; and chemical stability | Damages human cells and DNA at high doses; pulmonary toxicity | Surface-enhanced Raman scattering | Facemask | Gold Radiance Peel Off Mask–VLCC | [ , , , ] | |
| Anti-aging cream | Nano Gold Firming Treatment—Chantecaille | ||||||
| Buckyballs (buckminsterfullerene/C60) | Exhibits antioxidant activity, thermostability, and photostability; prevents many skin problems related to oxidative stress | Pulmonary toxicity; damages brain tissues; highly hydrophobic | Potent scavenger of free radicals | Face cream | Brightening Essence—Juva Skincare | [ , , ] |
2.1. Inorganic Particles
These are more hydrophilic, more biocompatible, safer, and exceptionally more stable particles as compared to natural nanoparticles. They can be significantly distinct, as these nanoparticles are derived from inorganic components (Ag, Au, Ti, etc.), while the natural ones are manufactured from polymers. Figure 2 shows the percentages of different inorganic nanoparticles in cosmetic and cosmeceutical formulations.

Proportions of different inorganic nanoparticles in cosmetics formulation.
There are many inorganic nanoparticles used in cosmeceuticals. A few important particles are described below.
2.1.1. Titanium Dioxide and Zinc Oxide
Sunscreens are useful for shielding the skin from the hazardous impacts of solar radiation, including UVB, UVA-2, and UVA-1 [ 27 , 28 ]. They usually consist of zinc oxide (ZnO) and titanium dioxide (TiO 2 ) as inorganic UV radiation filters, which prevent the harmful radiation of sunlight from reaching the skin. It has been established that ZnO is more effective for obstructing UVA, and TiO 2 is better for the UVB range. Hence, the appropriate proportion of the mixture of these particles guarantees wide-range UV protection [ 29 ]. TiO 2 is possibly the most broadly utilized and efficient inorganic nanoparticle for sunscreens and has a higher sun protection factor (SPF) at the nanoscale, which makes it more effective and results in a superior restorative effect due to its transparency, in contrast with its original color. These properties of TiO 2 are attributable to its large surface-area-to-volume ratio in the nano range [ 30 ], as it makes it be highly capable of carrying molecules when their sizes are reduced to 10–20 nm. Further, it has been reported that nanoscale TiO 2 and ZnO show incredible benefits over numerous materials that are larger than the nano range [ 31 ]. TiO 2 and ZnO nanoparticles used as UV filters in sunscreens [ 32 ] start at a size of 20 nm. They show better scattering and produce a superior restorative or protective effect. On the other hand, inhalation of a large amount of these nanoparticles has been shown to be harmful [ 33 ]. Thus, an alternate route of administration (i.e., dermal application) focuses on normal sunscreen ingredients, as these are safer, and there is no evidence of their infiltration into the epidermis or significant toxicity issues [ 16 , 34 ]. The International Agency for Research on Cancer (IARC) classifies TiO 2 as an IARC Group 2B carcinogen [ 32 ]. An investigation was carried out on rodents exposed to large quantities of TiO 2 nanoparticles and pigments, which resulted in cellular breakdown in the lungs of the rodents; this situation is comparable to working in dusty environments, causing serious harmful effects in individuals exposed to them. However, ZnO is considered a safe entity by the USFDA for use as a UV filter in cosmetics or cosmeceuticals. As an alternative, naturally occurring nanoparticles, such as ivy nanoparticles, which are secreted from the roots of English ivy (Hedera helix), are generally safer and employed for their UV-protective effects [ 35 ]. The enhanced visual transparency and safety of Ivy nanoparticles make them an attractive alternative to replace other toxic nanoparticles, reducing the impact on the health and environment.
2.1.2. Gold and Silver Nanoparticles
Gold and silver nanoparticles display antibacterial as well as antifungal properties [ 36 ] and are widely utilized in cosmetic formulations such as antiperspirants, anti-aging creams, and face masks. Gold has a long history of usage for skin health management and beauty care products in Egypt, where gold was used to maintain skin complexion. Egyptians believed that gold improved their skin composition and flexibility. Currently, gold is incorporated into different skincare items, such as salves, creams, and skincare treatments. Generally, gold in skincare products is called colloidal gold or, more precisely, nanogold if it is in the size range of 5 nm to 400 nm. Its color ranges from red to purple, depending upon the size and total surface area [ 2 , 37 ]. Gold nanoparticles have diverse shapes, such as nanospheres, nanorods, nanoclusters, nanostars, nanoshells, nanocubes, and nanotriangles, and the state of these particles determines their cell uptake and optical behavior. Properties such as stability and biocompatibility make them more appropriate for skincare and cosmetics [ 2 ]. Furthermore, their antifungal, antibacterial, and anti-aging benefits are well established, which are highly significant in cosmeceutical industries and in wound healing applications [ 38 ]. Gold nanoparticles play a substantial role in fixing skin damage and improving skin surface, grace, and flexibility. The soothing properties of gold make it an exceptional agent for treating skin inflammation, sunburn, and hypersensitivity. Hence, it can be successfully used in face masks and other cosmetics.
Silver nanoparticles can be utilized as successful inhibitors of various microorganisms. Silver and silver-based mixtures can be utilized to control bacterial development in different formulations [ 39 ]. The utilization of silver in cosmetics can be problematic, as silver readily precipitates in silver-based mixtures, which can be overcome by the utilization of silver nanoparticles. In Europe, the safety of colloidal silver in nanostructures concerning its use in oral and dermal cosmetic items is ambiguous [ 40 ]. In the USA, due to the lack of FDA regulations, cosmetic items are thought to lack promising antibacterial properties [ 41 ]. According to research, the use of silver nanoparticles as an additive in cosmetics makes the formulation stable, without showing sedimentation, for more than 1 year. Furthermore, silver nanoparticles showed adequate protection against microbes and their growth and did not enter human skin [ 42 ].
2.1.3. Silica (SiO 2 )
Because silica nanoparticles have hydrophilic surfaces favoring extended distribution and low manufacturing costs, interest towards these materials has increased, particularly in the cosmetic sector. Nanosilica is utilized to improve the adequacy, surface, and period of actual usability of cosmetic items [ 43 ]. It has been shown that silica nanoparticles may help to improve the appearance and appropriation of shades in lipsticks and keep colors in place [ 44 ]. Silica nanoparticles are present as nanodispersions with a size range of 5 to 100 nm and can deliver both hydrophilic and lipophilic entities to their respective targets by encapsulation [ 12 ]. These nanoparticles are generally found in leave-on and wash-off cosmetic items for hair, skin, lips, face, and nails, and the further expansion of silica nanoparticles in cosmetic items is expected [ 45 ]. However, the practical uses of silica-based nanoparticles are questionable and raise concerns about their safety, but factors such as size and surface changes ought to be considered while surveying its toxicity [ 45 , 46 ]. However, the commercial use of silica nanoparticles in beautifying agents is still ambiguous, requiring long-term trials [ 16 ].
2.1.4. Carbon Black
Carbon black, CI 77266, is known to be a significant ingredient in cosmetic formulations and is frequently utilized as a colorant in eye and skin cosmetic products. The EU has approved it for use in its nanostructure form and as a colorant at a maximum percentage of 10%. An evaluation of carbon black nanoparticles showed that they displayed a higher propensity for causing cytotoxicity, aggravation, and changes in phagocytosis in human monocytes as compared to micron-sized nanoparticles [ 47 ]. As per the EU, it can be utilized in cosmetic items when there is no danger of being breathed in [ 16 ].
2.1.5. Nano-Hydroxyapatite
Nano-hydroxyapatite is utilized in cosmetic items specifically meant for oral preparations that are used for treating extreme dental sensitivity and polish remineralization of the teeth [ 48 ]. It is regarded as a promising and safe option for these purposes by the US Food and Drug Administration (USFDA) [ 49 ]. These particles have been incorporated into oral formulations, such as dentifrices and mouthwashes, owing to their remineralization and desensitization properties. Such preparations could provide an alternative to fluoride toothpaste [ 16 ].
2.2. Nano-Organic (Tris-Biphenyl Triazine)
Tris-biphenyl triazine is a novel, powerful, and photostable filter specifically used in sunscreen formulations [ 50 ]. In its nano form, it functions as a broad-spectrum UV protectant and is thus frequently used in sunscreen preparations. It offers significant photostability and is an approved UV protectant in Europe. It is used under the name TINOSORB ® A2B by BASF SE. Methylene bis-benzo triazolyl tetramethyl butylphenol (nano), or MBBT, is another approved UV protectant in the EU market and can be utilized at percentages of up to 10% w/w in dermally applied cosmetic preparations. According to the Scientific Committee on Consumer Safety (SCCS) assessment, MBBT does not represent a danger to people if applied to solid, unbroken skin. Nonetheless, it has raised concerns related to possible harmful impacts and has the potential to bioaccumulate in selected tissues [ 16 ].
2.3. Bucky Balls (Buckminsterfullerene/C60)
Carbon fullerene has been extensively used in cosmetics and cosmeceuticals due to its antioxidative properties. Fullerenes are widely used in skin-rejuvenating cosmeceutical formulations because of their potent scavenging ability of free radical oxygen species, thus helping to reduce the effects of UV damage, such as hyperpigmentation and wrinkles [ 51 ]. Fullerene is a three-dimensional spherical compound that comprises a carbon ring with an odd number of carbon atoms [ 52 ] and is hence called “buckyballs” or buckminsterfullerene. Fullerenes alone have limited applications because of their hydrophobic nature, but the use of surface-active agents in a suitable concentration has improved their aqueous solubility and hence has successfully increased their utilization in pharmaceutical applications [ 36 , 53 ].
2.4. Miscellaneous
Nanoparticles utilized in cosmetics or cosmeceuticals can be comprehensively classified into two categories: biodegradable nanoparticles (made up of lipids, chitosan, etc.) and non-biodegradable nanoparticles (ZnO, silica-based nanoparticles, etc.) [ 54 ]. Chitin and its deacetylated derivative chitosan are another class that is of extraordinary interest to the cosmeceutical industry owing to their special organic and mechanical properties [ 55 ]. Nanofibrils of chitin are obtained from the shellfish exoskeleton with the removal of protein fractions and carbonate [ 54 ]. Chitin nanofibrils in emulsions can organize into a hygroscopic subatomic film that hinders water dissipation and adds to skin hydration [ 16 , 56 ].
3. Nano-Drug Delivery Systems Used in Cosmetics
Over the past few decades, nanotechnology has been providing novel solutions to several problems in the medical and pharmaceutical arenas. This same concept has been applied in cosmetics, resulting in novel formulations termed nanocosmeceuticals and providing customized remedies for cosmeceutical problems. The novel benefits may be attributed to a smaller size that helps to acquire new properties, such as better solubility, transparency, chemical reactivity, and stability. Several nanomaterials, such as liposomes, ethosomes, solid lipid nanoparticles, nanocapsules, dendrimers, nanocrystals, cubosomes, and nanoemulsions, are used in the cosmetic industry. Currently, cosmetic formulations incorporating nanoscience are extensively marketed. The following sections, including Figure 3 and Table 2 , describe various submicron-sized novel drug delivery systems used in cosmetic industries to deliver active ingredients.

Various nanoformulations used in the cosmetic industry.
Nanoformulations used for preparing various cosmeceuticals.
| S. No. | Nanoformulation | Advantage | Disadvantage | Uniqueness | Type of Cosmeceutical | Commercially Available Product | Reference |
|---|---|---|---|---|---|---|---|
| 1. | Nanoliposomes | Biodegradable, biocompatible, amphiphilic, and ↑ skin penetration | May trigger an immune response, ↓ medication stacking, ↓ reproducibility, and physicochemical flimsiness | Ability to compartmentalize and solubilize both hydrophilic and lipophilic materials | Moisturizer | Dermosome—Microfluidics | [ , , ] |
| Anti-wrinkle cream | Capture Totale—Dior | ||||||
| 2. | Niosomes | ↑ Efficiency, penetration, bioavailability, and stability of drugs | ↑ Cost of production, physical and chemical instability, leakage of the drug, time-consuming production | Surface development and alteration are extremely simple due to presence of useful functional groups on the hydrophilic head | Anti-aging cream | Lancome —Loreal, Paris | [ ] |
| 3. | Ethosomes | ↑ Efficiency and penetration of cosmetic delivery into the skin | Poor yield problems, ↓ stability, and possibility of coalescence | Consist of a relatively high percentage of ethanol | Moisturizer | Supravir Cream—Trima, Israel | [ , ] |
| 4. | Sphingosomes | Reestablishment of barrier function of skin and repair of dehydrated and damaged skin | Poor entrapment efficiency and expensive | Consist of sphingolipid, which makes them more stable than phospholipid liposomes | Anti-cellulite cream | Noicellex—NTT, Israel | [ , ] |
| 5. | Solid lipid nanoparticles (SLNs) | ↑ Duration of action, ease of large-scale production, ↑ bioavailability and biodegradability | ↓ Shelf life, decreased drug encapsulation | Crystalline in nature, ↑ drug loading matrix; consists of solid lipid | Perfume and cream | Chanel Allur | [ , , ] |
| 6. | Nanostructured lipid carriers (NLCs) | ↑ Shelf life, ease of large-scale production | ↓ Duration of action, higher drug encapsulation | The matrix consists of a blend of solid and liquid lipids | Face spa cream | Dr. Rimpler—Cutanova | [ , ] |
| 7. | Nanocapsules | Protection of ingredients, masking of undesirable odors, resolution of incompatibility issues between formulation components, sustained release formulation | Additional purification step is required after nanocapsule formulation | Formation of micelles and amphiphilic in nature | Anti-wrinkle cream | Primordiale Intense—L’Or’ea | [ , , ] |
| Hair care | Nano Collagen—Braziliss | ||||||
| 8. | Dendrimers | ↑ Solubility of the lipophilic drug, controlled-release drug formulation, and maintenance of the stability of the drug in cosmetic formulations | Not good materials for hydrophilic drugs, cellular toxicity, ↑ manufacturing cost | ↑ Shelf life of the cosmetic formulation | Sunscreen | Topical Resveratrol Formulation | [ ] |
| 9. | Nanoemulsions | Transparent, stable, and amphiphilic | Preparation is difficult in cosmetic formulations, acid-sensitive, and ↓ duration of action | Creams containing nanoemulsions do not show problems of inherent creaming, flocculation, coalescence, or sedimentation | Body lotions, skin creams, balsams, salves, and gels | Cosmeceutical Vitamin A, D, E, K—Vitalipid | [ , ] |
| Moisturizer | Nano Emulsion Multi-PeptideMoisturizer—Hanacure | ||||||
| 10. | Nanocrystal | ↑ Drug solubility, particle distribution, adhesiveness, dissolution rate, skin penetration of poorly water-soluble drugs | Possibility of aggregation, not appropriate for aqueous APIs, only stable to a certain extent | 100% drug loading ability | Moisturizer | Nano-In Hand and Nail Moisturizing Serum and Foot Moisturizing Serum—Nano-Infinity Nanotech | [ , , , , ] |
| Toothpaste | Nano WhiteningToothpaste—Whitewash |
3.1. Nanoliposomes
These are a nanometric form of liposomes that can be described as vesicles with concentric bilayers, where the fluid volume is encapsulated by bilayers of phospholipids [ 1 ], and are widely used as controlled release systems. Conventional liposomes are large and are actually liposomes inside another liposome; hence, they have a limited ability to enter narrow blood vessels or the skin, whereas nanoliposomes have better penetration ability. Being biodegradable and biocompatible, they behave as an exceptionally versatile nanomaterial in the field of cosmetics [ 68 ]. Cosmetic formulations incorporating liposomes have greater stability on the skin, as they are not easily washed off. These are ideal carriers of cells and biomembranes and can be successfully applied to the skin because they resemble the biological composition of the skin. They can also be utilized for fixing and transporting nutrients and for imparting pleasant scents to body wash, lipsticks, and antiperspirants [ 12 ]. Nanoliposomes in cosmetics enhance the hydration of the skin due to the smaller size of the particles, making the skin smooth and elastic. These are able to transport active moieties into the deeper layers of the skin, even to the systemic circulation, and can act as a transdermal drug delivery system (TDDS) in cosmeceutical applications. However, despite their promising features, low medication stacking, low reproducibility, and physicochemical fragility issues have restricted their commercial applications in beauty care products [ 16 , 69 , 70 ]. In the cosmetic industry, they are primarily employed for moisturizing and anti-aging purposes.
Recently, Han et al. demonstrated a novel approach to improve the absorption profile of collagen peptides obtained from Asterias pectinifera by using elastic nanoliposomes. This combination led to a promising formulation that not only resulted in a reduction in the expression of MMP-1 (produced upon exposure to UV radiation), thus preventing light-induced aging, but also may be used as an eco-friendly source of materials for anti-aging cosmetics [ 71 ]. Further, Kocic et al. performed an experiment that compared the moisturizing effect of marketed creams and nanoliposome creams incorporating skimmed donkey milk. They concluded that the nanoliposome encapsulated cream was able to penetrate deeper layers, resulting in reasonable moisturizing capacity with a rapid rate of hydration, and it therefore may contribute to anti-aging activity [ 72 ].
3.2. Ethosomes
The largest organ of the human body is the skin, which is known to restrict the movement of substances into the systemic circulation owing to the presence of a thick stratum corneum, which acts as a key physiological barrier. Ethosomes containing a very high concentration of ethanol and lipids are soft and flexible vesicles used as carriers to enhance transdermal delivery of a variety of cosmetic agents [ 73 ]. They can be customized for the safe and effective skin permeation of cosmeceutical products incorporating antioxidants, anti-wrinkle agents, salicylic acid, and many others. These systems are much more efficient than conventional liposomes in delivering topically applied cosmetics to the skin [ 74 ].
Research has found that the ethosomal formulation of niacinamide and melatonin can improve their ability to penetrate the skin with increased efficacy [ 75 ]. Another study claimed that ethosomes incorporating phenylethyl resorcinol delivered the active agent successfully into the skin for its skin-lightening activity [ 76 , 77 ]. One of the studies carried out by Yücel et al. claimed that the transdermal application of ethosomes loaded with rosmarinic acid (having anti-aging properties) exhibited better efficiency than that of the liposomal formulation. The skin permeation profile for the ethosomal formulation was found to be high with increased transdermal flux as compared to that of the rosmarinic acid solution and liposomes [ 78 ]. Another investigation conducted by Pravalika et al. with ethosomal vesicles incorporating minoxidil (a drug for the treatment of baldness) showed that the ethosomal gel had improved penetration as compared to other marketed formulations, which was concluded from both ex vivo permeability and hair growth experiments [ 79 ].
3.3. Solid Lipid Nanoparticles (SLNs) and Nanostructured Lipid Carriers (NLCs)
These are two novel delivery systems made up of a single layer of shells having a lipoidal center [ 2 , 63 ] and are used for formulating pharmaceutical as well as cosmeceutical products [ 80 ]. These formulations are characterized by a solid-state lipid matrix having a size in the nano range. The small size of the formulations permits direct entry into the corneum layer, which enhances the infiltration of active ingredients into the skin [ 81 , 82 ]. They show improved biocompatibility and safety and act as a successful transporter delivery system in cosmeceutical applications [ 12 ]. Since 2005, SLNs have been used in several dermal cosmetic products to achieve good outcomes [ 83 , 84 ]. SLNs are widely used in formulating sunscreens, where they act as active carriers for molecular sunscreen agents. They reduce the necessary amount of the sunscreen agent while offering the same protection as compared to conventionally used formulations. SLNs formulated with tocopherol acetate prevented chemical degradation, and improvement in the UV-blocking capacity was reported [ 85 ]. Another investigation incorporating a combination of chemical UV absorbers, chitin, and tocopherol in SLNs showed enhanced UVB protection action [ 86 ]. They are also used for film formation, which helps in the re-enforcement and repair of the skin barrier, making them perfect for cosmeceuticals that are used to treat irritated and itchy skin and dermatitis. Comparing the two, the crystalline nature of SLNs leads to less drug encapsulation efficiency as compared to NLCs, which have comparatively better encapsulation. Additionally, SLNs have a short shelf life along with slower drug release rates compared to NLCs [ 83 ].
A recent experimental study carried out on an SLN formulation integrating fucoxanthin (protects against UVB light) concluded that the presence of the SLN carrier improved the bioavailability of fucoxanthin and can be a promising carrier for sunscreen cosmetics, showing greater stability and good sunscreen-boosting action [ 84 , 87 ]. Another study combined the capability of the flavonoid as a natural antioxidant with NLCs to form an effective system for delivery into the cells. Further, the produced NLCs were incorporated into the skin with good stability and no significant cytotoxicity, suggesting that they can be used as anti-aging and moisturizing cosmetics in the future [ 88 ].
3.4. Nanocapsules
These are polymeric nanomaterials that encapsulate an oily or water phase within them. They are employed in beauty care products for protecting ingredients, masking undesirable odors, and mitigating incompatibility issues between various components in the formulations. Polymeric nanocapsule suspensions can be applied on the skin directly or can be fused into semisolid systems and used as carriers. The level of skin penetration can be regulated by the use of polymers and surfactants in the formulation [ 89 ]. In one study, nanoprecipitation was used to fabricate stable poly-l-lactic acid nanocapsules with a size of around 115 nm, and the continual release of fragrance was successfully established by encapsulating odorous atoms in a polymeric nano-transporter [ 90 ]. This sort of encapsulation of atoms in biocompatible nanocapsules can assume a critical role in antiperspirant formulations to enhance their effectiveness [ 16 ]. Recently, researchers developed novel stimuli-responsive nanocapsules that were developed to carry vitamins and extracts and finally were incorporated into semisolid formulations such as creams. When these formulations were applied on the skin, stimuli induced by damaged skin, such as a pH change and the presence of enzymes, forced the nanocapsules to release their active ingredients at the particular location of the skin [ 91 ].
Recently, one study demonstrated the successful incorporation of perfluorodecalin (oxygen carrier) into a silica nanocapsule core as a new tactic for topical therapy of aging skin due to the inherent instability of perfluorocarbon emulsions. Furthermore, this combination displayed better delivery and stability compared to emulsions [ 92 ]. Barbosa et al. developed nanocapsules composed of poly(ε-caprolactone) carrot oil and Pluronic containing benzophenone-3 in a sunscreen formulation. These nanocapsules improved the stability of the benzophenone in the topical formulation and also showed synergistic SPF activity with a non-irritant profile [ 93 ].
3.5. Dendrimers
Dendrimers are three-dimensional nanostructured macromolecules that are extensively branched, and this assembly accounts for their great adaptability [ 12 ]. They are generally polymers, and because of their stability, they are helpful in delivering active ingredients through the skin [ 82 ]. These molecules can be used in formulating shampoos and antiperspirants with increased efficiency. The surface movement and branches of dendrimers are due to the hydrophobic properties of their peripheral regions combined with the hydrophilic attributes of their central regions [ 68 ]. Moreover, properties such as monodispersion, polyvalence, and dependability make them ideal transporters for drug and cosmetic delivery [ 2 ]. Dendrimers of resveratrol (having antioxidant and anti-aging activities) have been developed and have assisted in improving the general solubility and skin infiltration [ 94 ], which later encouraged the scale-up and commercialization of this dendrimer structure-based formulation [ 16 ].
3.6. Nanocrystals
These are clusters made up of thousands of molecules joined together in a fixed pattern to form a group with sizes ranging from 10 to 400 nm and are usually utilized for the administration of poorly soluble drugs [ 95 ]. Nanocrystals mainly incorporate bioactive compounds and help to improve their dissolution rate. “Juvedical”, developed by Juvena in the year 2000, was the first marketed formulation containing nanocrystals with rutin as a key ingredient [ 96 ].
A study claimed that nanocrystals of rutin showed higher bioactivity as compared to the normal rutin glycoside [ 97 ]. In one of the latest studies carried out by Köpke et al. on the anti-pollution agent SymUrban, the solubility and the penetration profiles were observed to remarkably increase in its nanocrystal form. These nanocrystals increased the dermal bioavailability of the poorly soluble active ingredient in SymUrban and appeared to be a favorable delivery system for this material [ 98 ].
3.7. Cubosomes
Cubosomes are nanoparticles, particularly fluid crystalline particles, of a specific surfactant with an appropriate proportion of water combined in a nanostructure. Monoglyceride glycerol monoolein is the most common surfactant used to make cubosomes. These are distinct nanostructured particles that are used as cosmeceuticals for skincare formulations and also used in antiperspirant preparations. A number of investigations in collaboration with cosmetic organizations are attempting to utilize cubosomes for absorbing pollutants from cosmeceutical formulations and also employ them as a stabilizer for the oil-in-water type of emulsions [ 36 , 99 , 100 ].
Khan et al. reported a cubosome formulation containing erythromycin and concluded that the said non-invasive formulation exhibited better activity and effectiveness in preventing and treating acne and worked in a prolonged-release manner [ 101 ]. Further, one of the clinical studies conducted by El-Komy et al. claimed that the prepared cubosomal topical gel formulation incorporating alpha-lipoic acid is a safe and efficacious alternative for improving skin aging problems [ 102 ].
3.8. Nanoemulsions
Nanoemulsions are normally water-in-oil (w/o) or oil-in-water (o/w) colloidal solutions that range from a couple of nanometers to 200 nm [ 103 ]. The small size of the droplets is responsible for their alluring optical, rheological, and improved drug delivery properties, as compared to traditional formulations. Further, low viscosity, high solubilization ability, and increased kinetic stability due to sedimentation and flocculation make it more popular. Generally, these are transparent and stable and are employed for cleansing purposes, specifically in the cosmetic industry. These materials are used as powerful vehicles in the cosmetic industry for formulating body lotions, skin creams, sunscreens, etc. Nanoemulsions are also used in designing novel delivery systems for drugs and fatty materials such as essential oils, fatty acids, flavors, and colors. These systems are most appropriate for delivering lipophilic compounds, thus increasing their concentration in the skin; hence, they play a significant role in cosmetic formulations. An increase in patent-filing activity identified for nanoemulsions shows the emerging interest of industries in nanoemulsions [ 104 ]. An O/W nanoemulsion incorporating hydroglycolic extract of Opuntia ficus-indica (L.) Mill was formulated and characterized by high strength and saturating capacity [ 105 ]. Further studies revealed that nanoemulsions could generally impact the distribution profiles of atoms, specifically O/W nanoemulsions, which significantly improved the penetration profiles of polar ingredients relative to traditional emulsions [ 16 , 106 ].
Antioxidants play a significant role in the cosmetic and pharmaceutical industries but suffer from various limitations, such as insolubility and instability. To overcome these problems, researchers have fabricated antioxidants as nanoemulsions with enhanced efficacy [ 107 ]. In one of the investigations, to overcome the poor aqueous solubility problems of ellagic acid, Zhang et al. fabricated an oil-in-water nanoemulsion. They concluded that the developed nanoformulation had increased aqueous solubility and permeability through the skin, thus strengthening its whitening effect [ 108 ].
3.9. Micellar Nanoparticles
These are recognized as one of the most effective nanotechnology-based particles and have been widely used in the cosmetic industry. They offer a robust and versatile platform to encompass wide-ranging lipophilic active ingredients possessing diverse physicochemical properties in cosmetic formulations. Smaller particle size, better encapsulation efficiency, and reasonable manufacturing cost are the key features of these particles that make them more efficient than other nanocarriers [ 109 , 110 ]. Usually, they are employed in skin cleansing products for effective removal of oil and dirt from the skin without affecting barrier integrity and are used as an alternative to conventional cleansers [ 36 , 111 ]. These nanoparticles have led to a revolution in transdermal drug delivery (TDD). Micellar nanoparticle-based emulsions are attractive candidates for systemic drug delivery through topical application. The technology permits a high concentration of the drug to permeate the skin, creating a drug formulation attaining the same benefits as those of TDD, making the formulation more acceptable. Facial cleansing formulations incorporating micellar nanotechnology are claimed to be the most effective products by different cosmetic brands.
In one of the investigations by Zięba et al., the authors formulated a micellar shampoo and concluded that it had a higher viscosity and increased ability to emulsify fatty deposits as compared to traditional shampoos.
4. Health Risks Associated with Nanocosmeceuticals
It has been well established that nanoparticles pose serious health risks to humans due to their potential toxicity, which may further depend upon the quantity, route, and time of exposure of the nanoparticles. Other factors may include shape, surface structure, surface charge, chemical composition, and solubility [ 112 ]. Due to their small size and shape, nanoparticles can move easily inside the human body and are able to cross membranes and gain access to cells, tissues, and organs that are not accessible to larger-sized particles [ 113 ]. They can even enter cells, causing more damage or cell death [ 114 ].
At the nanoscale, the fundamental properties of substances become altered. For the same substance, the physicochemical properties differ between the nanoparticulate and larger particulate states. At the nano level, the chemical reactivity and biological activity are often higher as compared to those of larger-sized particles, which is attributed to their higher surface-area-to-volume ratios. Further, nanoparticles experience higher chemical reactivity, resulting in increased production of reactive oxygen species (ROS), including free radicals [ 115 ]. This is one of the key mechanisms of toxicity initiation that may lead to inflammation, oxidative stress, and consequent damage to membranes, proteins, and DNA. Nanomaterials may induce toxicity in various human systems, such as pulmonary, reticuloendothelial, neurological, and cardiovascular systems, and also have endocrine-disrupting or immunological effects.
The entry of nanoparticles into the body primarily occurs by three different routes, namely, inhalation, ingestion, and through the skin.
Inhalation is the most widely recognized route of exposure to airborne nanoparticles, as per the National Institute of Occupational Health and Safety (NIOHS). For instance, during the manufacturing of nanocosmeceutical products, workers may be exposed and may breathe in nanomaterials. Further, customers may also breathe in nanomaterials when applying products that contain them. For example, sunscreen sprays consisting of nanoscale TiO 2 may cause the inhalation of nanomaterials, which may travel through the nasal nerves to reach the cerebrum (brain) and sensory system and enter the blood and various organs, causing life-threatening adverse effects [ 116 , 117 ].
Further, the ingestion of nanomaterials may take place accidentally via transfer from the hand to the mouth or may be ingested intentionally. After ingestion, a moderate amount of the nanoparticles may be taken up by the body and move into vital organs and tissues, causing side effects [ 116 , 117 ].
Topical use of nanomaterials may also cause harmful effects. Various experimental data have shown that certain nanomaterials gain entry into the deeper layers of pig skin within 24 h of exposure [ 118 ]. As per investigations by the US Government Accountability Office (GAO), nanomaterials present in sunscreens can enter through damaged skin, causing serious side effects [ 116 , 117 ]. Figure 4 illustrates different diseases related to nanoparticle exposure.

Diseases associated with nanoparticle exposure [ 119 ].
As per the Yearly Meeting of the American Association for Disease Research, 2007, nanoparticles can damage DNA and lead to malignancy. Nanoparticles are small enough to infiltrate cell layers, yet they are large enough to alter normal cell functions, according to researchers at the University of Massachusetts. Because of their tiny size, they can be hard to separate by traditional separation procedures, and they can come across malignant cells and can cause significant damage, as per Sara Pacheco, a scientist at the College of Massachusetts. However, due to their small size, it is difficult to confirm exactly how they interact with the environment and how they affect human health [ 120 ].
Neonatal toxicity is another area of toxicity where exposure to these particles may cause severe harm in pregnant women. Nanoparticles used for various purposes may enter the placenta, endometrium, yolk sac, or fetus, resulting in oxidative stress and irritation. These problems may lead to placental damage, delayed neonatal development, fetal deformities, neurotoxicity, and reproductive dysfunction in infants. Further, nanoparticles may induce cytokine production in pregnant women, which may enter the fetus and result in poor development of the fetal brain [ 121 ].
5. Environmental Risks of Nanoparticles
In the modern era, nanotechnology is exploited as a useful tool to improve the quality of the environment, such as water and air, by cutting down waste production, reducing the emission of greenhouse gases, and decreasing the discharge of hazardous chemicals in the environment. However, they also have a negative impact on the environment. Owing to the novel and unique physical and chemical properties of nanoparticles, they can easily enter very small spaces [ 122 , 123 ]. They can cause biochemical interference by participating in reactions in biological systems. The mechanisms by which a nanomaterial can cause cell impairments can include bioaccumulation, ROS formation, oxidative stress, autophagy or lysosomal dysfunctions, etc. [ 124 ]. The influences of nanoparticles on the environment depend on the way that they are used in the workplace, their segregation mechanism in diverse media (such as air, water, and soil), their mobility, and their stability. Further, nanotechnology exposure in the environment and transportation are the fundamental factors that determine the overall influence on the environment. Nanoparticles are essential elements in various biogeochemical processes, and hence, any global-scale impact of specific nanoparticles on elemental cycles should be considered. Additionally, the environmental impact of these particles depends on factors such as solution chemistry, biochemical reactions, redox potential, temperature, pressure, presence of coating, etc., which should also be taken into consideration.
During the manufacturing process, nanomaterials may discharge into the water, air, and soil, causing serious environmental risks. As indicated by an investigation by the US Government Accountability Office (GAO), nanomaterials having antibacterial properties, whenever delivered in sufficient quantities, might interfere with the useful activity of microbes in water treatment plants and result in failure of the water proposed for reuse [ 1 ]. Nanoparticulate TiO 2 , used to decompose pollutants and for disinfection purposes, may have the ability to stimulate other organic transformations and have an influence on photochemical reactions in the atmosphere. Likewise, in an investigation coordinated by the University of Toledo, it was found that nano-TiO 2 , which is considered a close-to-home element, reduced the organic functions of microbes after an hour of exposure. Hence, it has been suggested that these particles, which end up at civil sewage treatment plants, could affect organisms that provide indispensable functions in the environment and thus should be used judiciously [ 1 ].
It has been evident that nanoparticulate TiO 2 , which is a key constituent of sunscreen, is released in significant quantities, causing potential damage to marine life with chronic exposure. In many sunscreen products, TiO 2 acts as a protective chemical coating, but when exposed to water, this defensive coating tends to be lost due to the influence of either UV light or seawater composition, releasing the toxic TiO 2 into the aquatic environment, specifically to algae and daphnids. This ultimately affects aquatic ecological balances [ 125 , 126 ]. Carbon-based nanomaterials show severe cytotoxic effects not only in human beings but also in other mammals by accumulating in different organs, such as lungs and kidney tissues [ 127 ]. At higher concentrations, they disturb the metabolic activity of microbes by interrupting the biogeochemical cycle of nutrients and also upsetting the nutrient balance. Further, metal nanoparticles have a high affinity for proteins [ 128 ] and can induce harmful cellular reactions and produce toxic effects on cells. An investigation carried out on the effects of titanium, polystyrene, and fullerene nanoparticles indicated that they are more toxic under biotic conditions than abiotic conditions because they induce oxidative stress [ 129 ].
In one of the studies carried out on carbon fullerenes, it was concluded that they can cause mild harm in largemouth bass (fish) [ 130 ], an animal used as a model for characterizing ecotoxicological impacts. Fullerenes have similarly been found to destroy water fleas and have bactericidal properties [ 131 ]. The Center for Biological and Environmental Nanotechnology of Rice University, Houston (USA), has called attention to the binding of nanoparticles to chemicals in the climate, such as cadmium and petrochemicals. This tendency would make nanoparticles a likely component of long-term and long-range vehicles of contaminants in groundwater [ 132 , 133 ].
These nanomaterials also affect plants after their absorption and translocation, producing severe effects. Nanoparticles of Ag, ZnO, fullerenes, silica, etc., are taken up by plants and algae, thereby producing toxicity and also hindering seed germination, as confirmed by researchers. They also impede the metabolic pathways affecting the growth and functioning of cells. In this way, these particles may enter the food chain and become biomagnified.
A workshop conducted by the National Science Foundation and the US Environmental Protection Agency was launched to identify the critical risk issues relating to nanomaterials. The workshop’s specific goals were to determine the exposure and toxicity of manufactured nanoparticles, the ability to deduce manufactured nanoparticle toxicity using existing particle and fiber toxicology archives, and the recyclability and total sustainability of manufactured nanomaterials.
At present, there is very little information available relating to the environmental risk of manufactured nanomaterials. Only a few findings have been published that describe the direct and indirect impacts of nanomaterials. To assess their risks, it is essential to have basic information about the behaviors and risks of nanoparticles. However, to date, there are no accurate data or guidelines available to quantify such effects. Further, it is essential to identify the sources, environmental paths, and uses of nanoparticles, along with plants and animals that are sensitive to nanoparticles, for the environmental risk assessment.
Currently, several researchers are determining the concentrations of a variety of nanoparticles in indoor and outdoor working areas. This helps to determine the levels of exposure of employees for that particular purpose and also for precautionary measures. Few studies have been published on the impacts of this type of exposure to nanomaterials. However, extensive research is still needed to evaluate the interactions of nanoparticles with biological systems as well as the ecosystem. Additionally, research should be conducted to establish the absorption, interaction, biodistribution, and excretion pathways of nanoparticles in living systems, along with nanotoxicological studies [ 134 ].
The biosynthesis of new nanomaterials from existing ones, polymeric coatings of metal nanoparticles to prevent leaching, combustion of carbon-based nanomaterials, recycling of metal-based nanoparticles, and bioaccumulation by means of plants or fungi can be used as effective methods for removing nanoparticles from different media. Green technology or green manufacturing can be used as a remedial solution for ecological protection. This is an environmentally friendly technology that intends to produce nanoparticles with reduced raw materials, minimum energy utilization, and less waste production, aiming to conserve natural resources. It employs green chemicals that are less toxic to the environment and is an energy-efficient procedure [ 135 , 136 ]. Lastly, awareness of individuals about the toxicity of nanomaterials, as well as attention to the use of safe and eco-friendly alternatives, may help resolve the problem [ 137 ].
6. Regulatory Guidelines of Cosmetics and Cosmeceuticals
The worldwide cosmetic market, valued at USD 532.43 billion in the year 2017, is predicted to reach USD 805.61 billion by the year 2023, with an annual growth rate of 7.14% [ 111 ]. Table 3 lists various patents linked with cosmeceuticals that may highlight the current global trend of cosmeceuticals. Looking at this increasing market value, it is necessary to regulate the cosmetics market with appropriate legal guidelines that will help to ensure the well-being of consumers. Although it is challenging to fulfill legal obligations for personalized products, it can be achieved with the correct approach and sustained compliance.
List of patents related to cosmeceuticals.
| S. No. | Patent No. | Country | Title | Application | Proof of Concept |
|---|---|---|---|---|---|
| 1. | CN100386064C | China | Biological wave nano-bioactive skin protection product | ↑ Microcirculation of the skin, hence ↑ metabolism and activation of cells, thereby improving the quality and activity of the skin, wherein vitamin E and ginsenosides can prevent skin aging and nourish the skin with good freckle removal effects. Prevents skin aging, nourishes the skin, and has ultraviolet resistance capacity. | A biological wave nano-bioactive skin protectant product comprising nanoparticles of ZrO and ZnO, vitamin E, and biological wave functional materials, such as ginsenosides and bioactive materials |
| 2. | KR101224378B1 | South Korea | Composite Pigment for cosmetic compositions and manufacturing method and manufacturing apparatus thereof | Complexing nanosized pigment particles (shell particles) to the surface of the extender pigment (core particles) to prevent reaggregation of nanosized pigment particles as shell particles and absorption into the human body | A composite pigment for cosmetics and a method for its manufacturing, wherein the composite pigment for cosmetics is coated with shell particles by physical pressure on the surface of the core particles |
| 3. | CN106660812A | China | Porous silica particles, a method for producing same, and cosmetic compounded with same | Porous silica particles in a cosmetic formulation act as a texture enhancer | This provides porous silica particles with a small specific surface area and a large pore volume, provides a method for producing the particles, and provides a cosmetic in which porous silica particles are present |
| 4. | BR102015012999A2 | Brazil | Composition, the process of preparation and use of nanocosmetic based on arnauba wax and quercetin with moisturizing, antioxidant and photoprotective action | A nanoparticle of carnauba wax lipid incorporating quercetin with 3-fold higher effectiveness as moisturizer, photoprotector, and antioxidant | The present invention describes a composition and process for the preparation and use of nanocosmetics consisting of lipid nanoparticles formulated with carnauba wax and quercetin incorporated into cosmetic formulations in gel, cream, lotion, or gel–cream forms |
| 5. | KR101578466B1 | South Korea | Porous sphere type zinc oxide powder of nanosize, manufacturing method thereof and color cosmetic composition using the same | Provides a spherical porous zinc oxide powder having a uniform particle size which ↑ the use by ↓ the opacity of the powder | The present invention relates to a spherical porous zinc oxide powder at the nano-scale, a process for its production, and a color cosmetic composition containing the same |
| 6. | KR20120091509A | South Korea | Nano-emulsion containing niacinamide and cosmetic composition comprising the same | A cosmetic composition containing niacinamide-containing nanoemulsions is provided to ↑ the transdermal absorption of niacinamide and to effectively and safely treat dry skin | Associated with niacinamide-containing nanoemulsion and a cosmetic formulation comprising the same |
| 7. | KR101528741B1 | South Korea | Silica-containing complex nanoparticles and hydrogel moisturizing patches comprising the same | Silica/zwitterionic polymer complex nanoparticles are able to strongly bind to moisture and accordingly ↓ vaporization speed, thereby having the effects of maintaining moisture and reinforcing the skin barrier | A silica/zwitterionic polymer complex nanoparticle, a manufacturing method thereof, and a hydrogel moisturizing patch |
| 8. | US9700042B2 | USA | Nanoformulation of musk-derived bioactive ingredients for nanocosmetic applications | Nanoformulation is applicable to cosmetic and textile manufacturing for providing fragrance and antimicrobial properties in cosmetic and textile products | Nanocarrier composition consists of hyaluronic acid (15–25%) and fatty acids (50–70%) cross-linked with ultra-low-molecular-weight chitosan (15–25%) incorporating isolated compounds from musk and their combinations |
| 9. | CN102274129A | China | Nano-sized core-shell composite material used for cosmetics and preparation method thereof | The composite material has sun-screening and moisturizing functions and dispersibility | The invention comprises a nanosized core–shell composite material composed of titanium oxide and zinc oxide based on the integration of characteristics of the 2 compounds, belonging to the field of skincare cosmetic chemicals |
| 10. | BR102015021346B1 | Brazil | Anti-inflammatory, healing and moisturizing tropic cosmeceutical formulation with active ingredients from Atallea Speciosa mart. Ex spreng (Babacu) | Provides anti-inflammatory, healing, and moisturizing activity and may be an alternative and/or therapeutic complement in the treatment of inflammation, tissue healing, and skin hydration processes | Topical anti-inflammatory, healing, and moisturizing cosmetic formulation with active ingredients of Mart. ex Spreng (babaçu) as plant bioactive compounds containing standardized mesocarp extract and almond oil capable of providing anti-inflammatory, healing, and moisturizing activity |
| 11. | TW201143840A | Taiwan | Compositions and methods for providing ultraviolet radiation protection | Provides excellent UV protection | Sunscreen compositions and related methods that can include a cosmetically acceptable carrier and a multitude of nanoparticles dispersed in the carrier |
| 12. | KR20120058795A | South Korea | Cosmetic composition containing carbon dioxide with nanopore | Provides excellent UV protection | Contains titanium dioxide to ensure high adhesion to the skin and excellent UV protection ability |
| 13. | KR101191268B1 | South Korea | Capsule composition contained nano inorganic particles for sunscreen product by hydrogel-forming polymers and manufacturing method thereof | Used to prevent skin penetration of nano-inorganic particles, as deep tissue penetration of nanoparticles results in various types of toxicity | Contains nano-inorganic particles providing UV protection and a method for manufacturing to form a thin hydrogel film |
| 14. | CN108401417A | China | Including improving the cosmetics of nano-particles and preparation method thereof of active principle containing whitening | Excellent nanoparticle for ↑ active principle containing whitening with long-term stability and cutaneous permeation of active principle | A cosmetic incorporating nanoparticles for ↑ active principle with skin-whitening effects; in more detail, the composition functions by ↑ the nanoparticle solubility to hydrophobic whitening active principle via micellization |
| 15. | CN104958189B | China | Light-sensitive color-matching makeup-removal-preventing nanopowder composition and application thereof in cosmetics | The novel skincare product can selectively reflect or scatter external light, only allows skin color light to enter skin, modifies dark skin and uneven surface shadows, and enables the skin to be bright and glossy | A light-sensitive color-matching makeup-removal-preventing nanopowder composition and a preparation method of the composition in cosmetics and application in cosmetics |
| 16. | KR20190085395A | South Korea | Patch composition comprising dog bone gold nano rod, graphene oxide or charcoal | The patch composition can be used as a patch, gel mask, and mask pack with excellent drug delivery into the skin and serves the function of causing an exothermic reaction when activated by LED light source having a wavelength of 700–1200 nm in the near-infrared region | A patch composition containing dog bone gold nanorod or charcoal or graphene oxide having ↑ visible light absorption; it can be used as a mask pack, having the effect of ↑ the drug delivery efficiency into the skin by implementing the target photothermal effect temperature of 41–45 °C by using an exothermic reaction |
| 17. | CN107001774B | China | Positive spherical monodisperse nanoparticle polyester resin water system dispersion and manufacturing method, positive spherical monodisperse nanoparticle polyester resin particle and cosmetics | Provides a kind of cosmetic that has good ductility in which there is good water resistance, softening skin | A novel technical method that is simple and inexpensive, steadily obtains useful positive spherical monodispersed nano-particle polyester resin particles and and water system dispersion |
| 18. | CN102958505B | China | Nanofiber laminate sheet | Appropriately used as a sheet-shaped make-up cosmetic | A nanofiber laminate sheet consisting of a layer of nanofibers composed of a water-insoluble polymeric compound, and a layer of a water-soluble polymeric compound includes a cosmetic component/a medicinal component |
| 19. | KR20130134580A | South Korea | Cosmetic compositions and layer comprising ultra-thin carbon | The thin-layer-laminated structure forms a coating film of the cosmetic composition on the skin, thereby having structural effects of making the active ingredients of the cosmetic composition useful for a long time and exhibiting excellent physical properties through a synergy effect | A cosmetic composition containing an ultra-thin carbon material having a surface diameter of 5–50 μm, which is a plate-type material made from graphite and has 1–10 molecular layers. |
| 20. | KR20140030395A | South Korea | The sunblock through hybrid of nanoparticle of a metal compound, the process for producing thereof, and the cosmetic utilizing thereof | Excellent UV protection | A sunblock agent formed by hybridization of nanoparticles of a metal compound, a process for producing the same, and cosmetic products utilizing the sunblock. More specifically, an organic and inorganic hybrid sunblock causes organic and inorganic hybridization by binding metal oxide nanoparticles of TiO or ZnO with one or more types of silane |
| 21. | TW200846027A | Taiwan | Nanocomposite pigments in a topical cosmetic application | ↑ Aesthetics and skin appearance | Introduction and the preparation of nano-pigments, with their role in ↑ aesthetic properties and skin appearance |
| 22. | US20100003291A1 | USA | Nano-particles for cosmetic applications | ↑ Characteristics of nanocosmetics and nanocosmeceuticals | Nanocosmetic and nanocosmeceutical preparations and their role in ↑ characteristics by improving the shortcomings of the traditional cosmetic preparation |
| 23. | CN101909580A | China | The Nanoparticulate compositions of enhanced color are provided to cosmetic formulations | ↑ Aesthetic properties, specifically color and skin appearance | The pharmaceutical composition comprises 1 or more pigments and the method for ↑ the external appearance of the biological surface by the optical characteristics |
| 24. | TW200533379A | Taiwan | Healthcare and cosmetic compositions containing nanodiamond | ↑ Mechanical strength of the cosmetic formulation | Shows ↑ binding capacity with the biological system and thus ↑ its mechanical strength; used in a variety of cosmetic formulations such as shampoo, nail polish, deodorants, eyeliners, etc. |
| 25. | KR20120058795A | South Korea | Cosmetic composition containing carbon dioxide with nanopore | Outstanding UV skin protectant due to good adhesive properties | Formulation containing 0.1–10 wt % TiO having a size in the range of 200–500 nm, providing protection against UV radiation and making the formulation softer |
| 27. | CA3124455A1 | Canada | Microparticles of cellulose nanocrystals with pigment nanoparticles bound thereto and method of production thereof | Preparation of several nanocosmeceuticals and nanocosmetics | Microparticles are formed by clustering nanocrystals and nanoparticle pigments, which are absorbed on the surface of nanocrystals |
| 28. | CN102112100B | China | Preparation of cationic nanoparticles and personal care compositions comprising said nanoparticles | Used as an antimicrobial agent in the cosmetic preparation | Utilization of cationic nanoparticles in the cosmetic formulation and their method of preparation and applications |
| 29. | KR100740275B1 | South Korea | Method for preparing zinc oxide powder with nanosize | UV protectant with ↑ transparency and adhesiveness | Method of preparation of nano-range ZnO particles by one-step wet preparation and their application in cosmetics |
| 30. | KR100785484B1 | South Korea | Base composition encapsulating high concentration of idebenone with nano sizes, its manufacturing method thereof, and cosmetic compositions containing it | Easily alter the viscosity of the cosmetic and hence ↑ the efficiency of production with ↓ cost | Method of preparation and application of the nanoencapsulation of ↑ concentration of idebenone in a bioactive base for cosmetics production |
| 31. | WO2021144889A1 | WIPO (PCT) | Nanobubble-containing cosmetic | Preparation of stable nanobubble solution | Introduction of nanobubble solution in the cosmetic formulation as an active ingredient |
| 32. | KR101436540B1 | South Korea | UV protection cosmetic composition comprising titania nanorod | UV protectant and transparency in cosmetics | Synthesis of a cosmetic formulation comprising titania nanorods as a potential sun protectant |
| 33. | CN102397168B | China | Flexible nanoliposomes with charges for cosmetics and preparation method thereof | ↑ Stability, permeability, efficiency, retention time, and action of the active ingredients | Introduction to flexible nanoliposomes and their utilization in the cosmetic formulations |
There are many regulatory documents that are intended to assist the cosmetics industry and other stakeholders (academicians, researchers, etc.) in identifying and investigating the safety aspects of nanomaterials in cosmetics. There are compilations in the literature that summarize the legal aspects of nanomaterials [ 138 ] or the use of nanomaterials specifically in cosmetics [ 139 ]. Here, we briefly summarize the main recommendations of a few important guidance documents.
6.1. Food and Drug Administration (FDA): Guidance for Industry Safety of Nanomaterials in Cosmetic Products
This document provides guidance to industry and other stakeholders on the FDA’s current thinking on the safety assessment of nanomaterials in cosmetic products. The FDA’s guidance documents, including this guidance, do not establish legally enforceable responsibilities. Instead, guidance should be viewed only as recommendations unless specific regulatory or statutory requirements are cited.
This guidance also refers to other relevant reports, such as the Organization for Economic Co-operation and Development (OECD) Working Party on Manufactured Nanomaterials “Preliminary Review of OECD Test Guidelines for their Applicability to Manufactured Nanomaterials”, the Scientific Committee on Consumer Safety (SCCS) “Guidance on the Safety Assessment of Nanomaterials in Cosmetics”, and relevant ICCR reports, such as on the “Currently Available Methods for Characterization of Nanomaterials,” and “Principles of Cosmetic Product Safety Assessment.”
This guidance presents recommendations very comprehensively. However, in summary, for any cosmetic product that has new or altered properties, data needs and testing methods should be evaluated to address any unique properties and functions of the nanomaterials used in the cosmetic products. The FDA recommends that the safety assessment of cosmetic products using nanomaterials address several important factors, including:
- The physicochemical characteristics,
- Agglomeration and size distribution of nanomaterials under the conditions of toxicity testing and as expected in the final product,
- Impurities,
- Potential routes of exposure to the nanomaterials,
- Potential for aggregation and agglomeration of nanoparticles in the final product,
- Dosimetry for in vitro and in vivo toxicology studies, and
- In vitro and in vivo toxicological data on nanomaterial ingredients and their impurities, dermal penetration, potential inhalation, irritation (skin and eye), sensitization studies, and mutagenicity/genotoxicity studies.
The safety of a cosmetic product should be evaluated by analyzing the physicochemical properties and the relevant toxicological endpoints of each ingredient in relation to the expected exposure resulting from the intended use of the finished product. If the manufacturer wishes to use a nanomaterial in a cosmetic product, either new material or an altered version of an already marketed ingredient, this guidance recommends that it meets with the FDA to discuss the test methods and data needed to substantiate the product’s safety, including short-term toxicity and other long-term toxicity data, as appropriate [ 140 ].
6.2. International Cooperation on Cosmetics Regulation (ICCR): Report of the ICCR Working Group—Safety Approaches to Nanomaterials in Cosmetics
Discussions at the 4th annual meeting of International Cooperation on Cosmetics Regulation (ICCR-4) on cosmetics and cosmetic-like drugs in Canada in July 2010 led to the formation of a Joint Industry/Regulator Working Group (WG) for nanomaterial safety. The purpose of this Joint WG was to examine existing safety approaches for their applicability to nanomaterials relevant to activities within the cosmetic industry. The main task was to carry out a review of existing safety approaches, identify any specific aspects relevant to consumer safety that should be taken into consideration when assessing nanomaterials in cosmetics, and produce a draft report for discussion by the ICCR members.
The members of the Joint WG discussed the main issues and prepared a report after considering several key reports, opinions, guidance documents, and relevant publications. The report’s aim is to provide information to those intending to use or assess the safety of nanomaterials in a cosmetic product. This report expresses the views of the experts on the key safety aspects that need to be assessed when using nanomaterials in cosmetic products.
The main highlights of the report are:
- The existing risk assessment pattern (exposure assessment, hazard identification, hazard characterization, and risk characterization) used for conventional chemicals is also applicable to nanomaterials.
- The WG identified physicochemical parameters that should be measured for nanomaterials at the raw material stage.
- The assessment should include the investigation of systemic exposure, local effects, possible routes of exposure (dermal, respiratory, or oral), and foreseeable uses of the cosmetic product.
- If systemic absorption is seen, it should be further investigated to confirm whether the absorbed material was in particle form or in a solubilized/metabolized form. The absorption, distribution, metabolism, and excretion (ADME) profile should be investigated to assess the fate and behavior of the nanoparticles in the body and identify the plausible target organs.
- For nanomaterials having very low absorption, processes such as accumulation should also be considered.
- The effects of the formulation should also be considered, as certain formulations may alter the bioavailability and toxicological profile of active ingredients.
- The EU Cosmetics Regulation bans the testing of cosmetics on animals. This makes the safety evaluation of new nanomaterial cosmetic ingredients difficult. Though there are several validated alternative methods that can be used in place of animal tests for conventional substances, none of those methods is yet validated for nanomaterials. However, they may still be useful for hazard identification and provide additional supporting evidence to the results of in vivo studies [ 141 ].
6.3. Scientific Committee on Consumer Safety (SCCS): Guidance on the Safety Assessment of Nanomaterials in Cosmetics
This document is an up-to-date guidance on the safety assessment of nanomaterials in cosmetic products, covering the main elements of safety assessment, i.e., general considerations, material characterization, exposure assessment, hazard identification/dose–response characterization, and risk assessment.
The main points of this guidance may be summarized as follows.
6.3.1. Definition of Nanomaterial
Material specifications such as particle size distribution, solubility, and persistence should be considered to decide whether a cosmetic ingredient is a nanomaterial or not. Imaging by electron microscopy may be used for further clarification if needed. Where a cosmetic ingredient qualifies to be defined as a nanomaterial, it will be subjected to safety assessment based on the data relevant to nanoscale properties.
6.3.2. Material Characterization
Considering the special behavior of the nanomaterials, their clear identification and detailed characterization are essential requirements for safety assessment. The characterization of the nanomaterial needs to be carried out at the raw material stage, in the cosmetic formulation, and during exposure for toxicological evaluations. The characterization data must identify the materials in accordance with Cosmetics Regulation (EC) No 1223/2009. Measurements must be carried out using generally accepted techniques, and detailed documentation must be provided. Particle size, being the most important factor, must be measured by more than one method, one of which should be electron microscopy.
6.3.3. Exposure Assessment
Safety assessment of nanomaterials is performed using the same procedure as for non-nano-ingredients, but with special attention to the nano aspects. Firstly, the likelihood and extent of local and systemic exposure need to be determined in relation to dermal, oral, and inhalation exposure routes. The potential translocation of nanoparticles across skin, lung, or gastrointestinal barriers should be determined. The methods used for this purpose should be mainstream and state-of-the-art with a low limit of detection. ADME parameters should be studied to determine the extent of systemic exposure, fate, and behavior of the nanomaterial and to identify the target organs. If systemic exposure is indicated, further investigations should be carried out to confirm whether the absorbed material was in particle form or in a solubilized/metabolized form. In cases where systemic exposure is not indicated, local exposure and local adverse effects should be investigated.
6.3.4. Hazard Identification/Dose–Response Characterization
Data from toxicological studies on local/systemic effects are required as per SCCS Notes of Guidance. Hazard identification/dose–response characterization includes consideration of insoluble or partially soluble particulate forms, aggregation and agglomeration behavior of the particles, potential penetration of nanoparticles through biological membranes, possible interaction with biological entities at local and systemic levels, surface adsorption/binding of other substances, surface-catalyzed reactions, persistence, etc. The prohibition of animal testing as per the Cosmetics Regulation must be observed in any toxicological testing. The SCCS can accept results from methods that may not have been formally validated for nanomaterials but can be demonstrated to be scientifically valid for hazard identification, provided that they are carried out with due consideration of the nano-related aspects and appropriate controls.
6.3.5. Safety Assessment
With the EU ban on animal testing of cosmetic ingredients/products, the applicant needs to collect relevant data from different alternative methods and provide evidence to support the safety of the cosmetic ingredient. Where safety assessment is based on in vitro test results, extrapolation of in vitro to in vivo (IVIVE) data will be required [ 142 ].
6.4. Comparison of Regulation of Cosmetics/Cosmeceuticals across Different Countries
The safety and efficacy of cosmetic products are governed by different regulatory bodies of countries all around the world according to their own guidelines. In a few countries, the final product’s safety is assured before marketing approval by the manufacturers. The label should contain all constituents of the formulation along with the limits that are identified for the cosmetic and cosmeceutical ingredients and products, and the mentioned limits should comply with the established limits. Simultaneously, many countries lack these regulations. The following section lists a few of the current regulatory scenarios of cosmetic products in the United States of America (USA), the European Union (EU), and India [ 7 ], and Table 4 compares the regulation of cosmetics/cosmeceuticals in these regions. This may help the reader to understand various regulatory procedures in different countries.
Comparison of regulation of cosmetics/cosmeceuticals in the USA, the European Union, and India [ 7 ].
| Country | Regulatory Authority | Rules | Approval (Premarket) | Labeling | Labeling Declarations | Language of Label | Expiry Date | Safety | Warning |
|---|---|---|---|---|---|---|---|---|---|
| USA | USFDA | Food, Drug, and Cosmetic Act | No specific requirement | Must conform with the FP&L and FD&C | 21 CFR 701 and 740 of USFDA | English | Not required | Manufacturer responsibility | On the primary display panel |
| EU | EMEA | Council Directive 76/768/EEC | No specific requirement | Based on Council Directive | Cosmetic Directive, Article 6 | National or member state | If the stability is <30 months → Date of minimum stability is mentioned; If stability is >30 months → days/months/years after opening is mentioned | Information file of the product is being maintained by the manufacturer | On both outer and inner label |
| India | CDSCO | Drugs and Cosmetics Act, 1940 | Required under the state government licensing | Comply with D&C rules 1945—Part XV | BIS and PCRO | English | It should have “Use before date” | The records of the product’s safety must be maintained by the manufacturer | On inner label |
6.4.1. United States of America
In the USA, the regulation of cosmetics is approved by the United States Food and Drug Administration (USFDA) and controlled by the Food Drug and Cosmetic Act (FDCA). It is well known that drugs are regulated by the FDA. They must either have FDA’s premarket approval or follow the final regulations specifying conditions to be recognized as safe and effective, but cosmetics lack this premarket approval procedure, except for color additives [ 7 ].
The most commonly used cosmetics include toothpaste; nail polish, skin, eye, and facial creams, lotions, lipsticks, perfumes, antiperspirants, shampoos, hair products, etc. Soaps mainly comprising a soluble base salt of unsaturated fat used for cleaning the human body are not regarded as cosmetics as per the law (USFDA, 2014). Cosmetic products do not have comprehensive rules for approval before their marketing, in contrast to drugs. In the United States, at a minimum, the manufacturers, distributors, and packagers of the cosmetic item are expected to use the Voluntary Cosmetic Registration Program (VCRP), which offers benefits for participation. VCRP provides information to the FDA about beauty care products and their manufacturing, distribution, and recurrence activity. The producer or wholesaler must prepare a documentary report, which is known as a Cosmetic Product Ingredient Statement, for each item that the firm has brought to the market. According to the law of the organization, the USFDA might carry out an examination, inspect items and the organization wherein items are manufactured or stored, and identify misbranded or tainted cosmetic or cosmeceutical products. The Personal Product Act 2013 was established to enable the USFDA to guarantee that cosmetic items are completely safe and contain no harmful entities [ 7 ].
The cosmetic regulatory guidelines that apply to contaminated and misbranded products are provided in the Federal Food, Drug, and Cosmetic Act, which requires a one-year-long registration of an operation involving advertisement, manufacturing, or dispensing cosmetics. It also requires the disclosure of data and the labeling of active ingredients as well as excipients and also should reveal the related adverse effects, if any [ 7 ].
The act requires the organization to maintain standard records of restricted constituents and the constituents that are completely safe and unregulated for the purpose of utilization in cosmetic formulations. Along with this, the manufacturers are required to conduct certain basic important tests of the constituents to ensure their safety. Additionally, this act puts forth prerequisites identified for nanotechnology in the production of cosmetics, compulsory and voluntary review of beauty care products, and alternatives to testing on animals. To share the information, the act establishes the Interagency Council on Cosmetic Safety (ICCS) and, just as importantly, helps the organization that looks after the safety of cosmetics with government analysts. As per the Federal Food, Drug, and Cosmetic Act, 2013, cosmetic products that fail to justify their labels are considered misbranded. The label and the packaging of the product should provide buyers with exact information on how to use or apply it, and the packaging should have information on the comparable quantities of ingredients in the particular product as per the Fair Packaging and Labeling Act. Additionally, the above-mentioned label should be wrapped around the product or placed within it. The principal display panel (part of the label most plainly visible when displayed under the standard environmental factors in settings where it is available for purchase) should show the item name and provide an accurate report of the net quantity of ingredients in the formulation as a measure, weight, count, or a combination thereof. The declaration should be noticeable, situated on the front of the packaging, and at a size proportionate to the size of the package. The packaging must incorporate inserts, booklets, risers, or some other printed or data associated with the product (Cosmetic Labeling Guide, 2015). All of the essential guidelines should be written in the English language and should be placed within the label in such a way that they can be easily noticed and observed by the consumer [ 7 ].
In 2006, the FDA established an interior nanotechnology team to manage nanoparticle-based items. This step was taken to improve the safety and effectiveness of nanomaterials. Later, in 2007, alterations were recommended by the FDA; many have been executed, and a few are under investigation. Further, in 2014, the FDA identified three rules concerning the safety of nanoparticles; two of them are associated with cosmetic products. The first rule elucidated the assurance of FDA-managed formulations incorporating nanoparticles. The second one is focused on the safety of nanomaterials in cosmetic items. Additionally, the FDA has not been required to disclose the list of nanomaterials incorporated in formulations on labels [ 143 ] and regularly updates manufacturers about nanomaterial-linked risks for continual improvement in the safety of the cosmetics. By implementing this process, formulations are continuously adjusted so that the utilization of hazardous substances is limited [ 111 ].
6.4.2. European Union
The European Medicines Evaluation Agency (EMEA) is the regulatory agency for cosmetics in Europe, which is under the direct control of Council Directive 76/768/EEC. It covers the safety associated with the use of cosmetic items and the record of permitted colorants. The safety of cosmetics and smooth operation for all administrators in this area are governed by European Regulation 1223/2009. The above-mentioned regulation provides a strong, universally recognized system that establishes product safety considering the most recent scientific data, including the feasible utilization of nanomaterials [ 7 ].
According to the EU, a product safety report should be made prior to entering the market. Only cosmetic products for which a natural or legal individual is delegated can be sold on the cosmetics market; genuine undesirable effects should be communicated to public government agencies, which will later accumulate related data from health experts and clients and will inform the other EU Member States. Preservatives, colorants, and UV protectants with materials in the nano range should be approved. Formulations containing nanomaterials are not under the control of the EU cosmetic regulation and have to go through a complete examination under the supervision of EU experts. The manufacturer will provide information about its formulation with the help of an EU notification portal known as the EU cosmetic products notification portal (CPNP); it should mention the market name and the registered address of the manufacturer [ 7 ].
European Commission Regulation No. 1907/2006 regulates nanomaterials in the EU. The nanomaterial ingredients should be suffixed with the term “nano”, e.g., “zinc oxide (nano)”, as per European Commission, 2015 [ 144 ]. According to EU guidelines, nanomaterials are characterized as insoluble material and deliberately fabricated with at least one external measurement or an inner dimension in the size of 1–100 nm. Data on the item detailing undesirable effects, the safety profile, and toxicity should be provided half a year before the market approval of nanocosmeceutical/nanoparticle-based items. It requires premarket approval for nano-based cosmeceuticals, anti-aging creams, colorants, and sunscreen items [ 111 ].
6.4.3. India
In India, the cosmetic market is known to be the quickest rising retail segment, and the active Indian market offers freedom for foreign brands. It permits access to imported beautifying agents with no restrictions. In the last 20 years, many different participants have entered the Indian cosmetic market, hence demanding strict regulations to preserve the safety of consumers. The Central Drug Standard Control Organization (CDSCO) regulates all activities related to cosmetics or cosmeceuticals in India and is controlled by the Drug and Cosmetics Act and Rules. Further, the Bureau of Indian Standards (BIS) regulates the labeling contents of cosmetics or cosmeceuticals. It sets the minimum quality of cosmetic products for the recorded items and provides details about hair care products and creams. Under CDSCO, the Drug Controller General of India (DCGI) regulates all related activities [ 7 ].
In India, as indicated by the Drug and Cosmetics Act, labels, whether external or internal, should contain the name of the cosmetics and the address of the manufacturer. If the size of the package is small, then the name of the manufacturer’s address with a pin code is sufficient. The external label should contain the name of the ingredients along with their quantities in the formulation. The internal label should include directions for the use of the product and the name and quantity of poisonous or hazardous substances that are used, along with warnings, if any. The particular batch number, which is indicated by the letter “B”, should be included in all cosmetic or cosmeceutical formulations, but in the case of soap, the manufacturing month and year must be present, omitting the letter “B” in the label. However, this is not the case for solid or semisolid cosmetic formulations having a weight equal to or less than 10 g and for liquid cosmetic formulations having a volume equal to or less than 25 mL. The manufacturing license number must be present on the label, which is indicated by the letter “M”, as per Drugs and Cosmetics Act and Rules, 2013 [ 7 ].
The Government of India invested in the nanoscience and technology initiative and provided efficient resources to different colleges, scholar societies, public research facilities, and new companies with R&D units. In India, the important bodies involved in the public health research frameworks are the Indian Council of Medical Research, the Department of Science and Technology, the Council of Scientific and Industrial Exploration, and the Department of Biotechnology (all in New Delhi, India). The Ministry of Health and Family Welfare (New Delhi, India) plays an essential role in the prevention and control of health-related issues in India. Furthermore, the Nanotechnology Sectional Committee, which includes specialists subsidiary to different research foundations and associations, is effectively responsible for the standardization of nano-based items and their safety [ 111 ]. Currently, nanomaterial concerns are continuously evolving in India and demand special attention for the improved safety of the public.
7. Conclusions and Future Direction
Currently, nanotechnology is regarded as a promising and revolutionizing field and is being utilized and appreciated in the areas of cosmetics, cosmeceuticals, dermatology, biomedical applications, etc. The introduction of newer advancements and novel drug delivery systems make cosmetics and cosmeceuticals more popular with increased market share. Today, these cosmetics are an indispensable part of the daily routine; further, the introduction of nanotechnology to cosmetics has enhanced its acceptance among users all around the world. However, its associated toxicity owing to its penetrability is a major concern that is often overlooked, leading to adverse health issues. Presently, novel nanocarriers such as liposomes, ethosomes, cubosomes, NLC, SLNs, nanoemulsions, niosomes, etc., are exploited to formulate various cosmetics and cosmeceuticals with enhanced outcomes. Nanosystems carry and deliver these formulations across the skin by diverse mechanisms and impart several functions, such as sun protection, moisturization, wrinkle reduction, etc. Even though these nanomaterial products are gaining impressive market value, there is tremendous debate concerning their safety and toxicity in humans, demanding more careful investigations. Hence, the cosmetic legislation should provide a specific list of references as well as the ingredients that produce unintended environmental effects for all users of cosmetic products, such as consumers and professional users, thus ensuring the safety of the usage of cosmetic products. Long-term toxic or carcinogenicity studies of cosmetics, including nanocosmetics and nanocosmeceuticals (and their ingredients), should be conducted before the commercialization of these products. Nanocosmeceuticals should be manufactured in such a way that they add value to the health of consumers. Moreover, careful clinical trials of cosmeceuticals should be conducted, such as those performed for drugs, to assure the safety of the formulations in humans. Additionally, stringent regulations should be imposed on the manufacturing, storage, import, and marketing of cosmeceuticals and nanoparticles incorporated therein. Universal collaborative efforts among researchers as well as global regulatory agencies are required to develop standard rules and regulations for using nanosystems in cosmetics and help address the existing gaps in the related data. Non-governmental organizations and government bodies should work in a coordinated manner to develop and propagate effective education materials for consumers. They should establish special programs, such as written and video materials, through multimedia or seminars with the aim to provide education for the wise use of cosmetics containing nanocosmetics and nanocosmeceuticals. Finally, there is a need to harmonize regulations internationally to establish a better regulatory framework for safety, efficacy, and marketing, which ultimately helps the cosmetic industries and also protects consumers from potential hazards. Moreover, awareness among consumers can also help to improve this situation by enabling informed choices of products.
Acknowledgments
The authors are highly grateful to Jamia Hamdard and DST for providing support in the form of DST PURSE.
Author Contributions
Conceptualization, V.G. and S.M.; methodology, S.M.; software, M.A.M., M.J.A. and H.M.; data curation, U.F. and K.K.; writing—original draft preparation, V.G. and S.M.; writing—review and editing, S.M. and H.M.; visualization, U.F. and K.K.; supervision, M.A.M. and Z.I.; project administration, M.A.M., M.J.A. and Z.I.; funding acquisition, M.F.A. and A.S.A. All authors have read and agreed to the published version of the manuscript.
This research received no external funding.
Institutional Review Board Statement
Informed consent statement, data availability statement, conflicts of interest.
The authors declare no conflict of interest.
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Information
- Author Services
Initiatives
You are accessing a machine-readable page. In order to be human-readable, please install an RSS reader.
All articles published by MDPI are made immediately available worldwide under an open access license. No special permission is required to reuse all or part of the article published by MDPI, including figures and tables. For articles published under an open access Creative Common CC BY license, any part of the article may be reused without permission provided that the original article is clearly cited. For more information, please refer to https://www.mdpi.com/openaccess .
Feature papers represent the most advanced research with significant potential for high impact in the field. A Feature Paper should be a substantial original Article that involves several techniques or approaches, provides an outlook for future research directions and describes possible research applications.
Feature papers are submitted upon individual invitation or recommendation by the scientific editors and must receive positive feedback from the reviewers.
Editor’s Choice articles are based on recommendations by the scientific editors of MDPI journals from around the world. Editors select a small number of articles recently published in the journal that they believe will be particularly interesting to readers, or important in the respective research area. The aim is to provide a snapshot of some of the most exciting work published in the various research areas of the journal.
Original Submission Date Received: .
- Active Journals
- Find a Journal
- Proceedings Series
- For Authors
- For Reviewers
- For Editors
- For Librarians
- For Publishers
- For Societies
- For Conference Organizers
- Open Access Policy
- Institutional Open Access Program
- Special Issues Guidelines
- Editorial Process
- Research and Publication Ethics
- Article Processing Charges
- Testimonials
- Preprints.org
- SciProfiles
- Encyclopedia

Journal Menu
- Cosmetics Home
- Aims & Scope
- Editorial Board
- Reviewer Board
- Topical Advisory Panel
- Instructions for Authors
- Special Issues
- Topical Collections
- Article Processing Charge
- Indexing & Archiving
- Most Cited & Viewed
- Journal Statistics
- Journal History
- Journal Awards
- Editorial Office
- 10th Anniversary
Journal Browser
- arrow_forward_ios Forthcoming issue arrow_forward_ios Current issue
- Vol. 11 (2024)
- Vol. 10 (2023)
- Vol. 9 (2022)
- Vol. 8 (2021)
- Vol. 7 (2020)
- Vol. 6 (2019)
- Vol. 5 (2018)
- Vol. 4 (2017)
- Vol. 3 (2016)
- Vol. 2 (2015)
- Vol. 1 (2014)
Find support for a specific problem in the support section of our website.
Please let us know what you think of our products and services.
Visit our dedicated information section to learn more about MDPI.
Current and Future Trends in Cosmetics Research: The 10th Anniversary of Cosmetics
- Special Issue Editors
Special Issue Information
Benefits of publishing in a special issue.
- Published Papers
A special issue of Cosmetics (ISSN 2079-9284).
Deadline for manuscript submissions: 31 December 2024 | Viewed by 42315
Share This Special Issue
Special issue editor.

Dear Colleagues,
It is my pleasure to celebrate the 10th anniversary of Cosmetics , which, over these years, has become a reference for many scientists interested in this area of research. Ten years ago, we took the first steps towards the creation of a journal covering all the different interests related to the biological, biochemical, and clinical research in the cosmetic world. Today, Cosmetics is a common platform for all scientists willing to communicate and exchange information and scientific ideas in this field. Indeed, cosmetic research is a field that aims to develop new products and methods for enhancing the appearance and health of the skin, hair, nails, and other body parts.
Cosmetic research is driven by various factors, such as consumer demand, technological innovations, environmental concerns, and regulatory requirements.
Among the main trends in cosmetic research is the use of natural and organic ingredients, which are perceived as safer, more sustainable, and more ethical than synthetic ones. Natural and organic cosmetics are made from plant-based, mineral, or animal-derived substances, such as oils, extracts, waxes, pigments, and enzymes.
Customization is another trend in cosmetic research. This refers to the development of personalized and customized products which cater to the specific needs and preferences of individual consumers. Personalized and customized cosmetics are based on factors such as skin type, skin tone, hair color, age, lifestyle, and genetic makeup. Some examples of personalized and customized cosmetics are serums, foundations, hair dyes, and fragrances.
Furthermore, the integration of biotechnology and nanotechnology, which enable the creation of novel and advanced materials and formulations, is another key trend. Biotechnology and nanotechnology are fields that manipulate biological and molecular structures such as cells, proteins, DNA, and nanoparticles at the microscopic level. Some examples of biotechnology and nanotechnology cosmetics may be found in anti-aging creams, sunscreens, hair growth stimulants, and color-changing cosmetics.
I am looking forward to receiving your contributions!
Prof. Dr. Enzo Berardesca Guest Editor
Manuscripts should be submitted online at www.mdpi.com by registering and logging in to this website . Once you are registered, click here to go to the submission form . Manuscripts can be submitted until the deadline. All submissions that pass pre-check are peer-reviewed. Accepted papers will be published continuously in the journal (as soon as accepted) and will be listed together on the special issue website. Research articles, review articles as well as short communications are invited. For planned papers, a title and short abstract (about 100 words) can be sent to the Editorial Office for announcement on this website.
Submitted manuscripts should not have been published previously, nor be under consideration for publication elsewhere (except conference proceedings papers). All manuscripts are thoroughly refereed through a single-blind peer-review process. A guide for authors and other relevant information for submission of manuscripts is available on the Instructions for Authors page. Cosmetics is an international peer-reviewed open access semimonthly journal published by MDPI.
Please visit the Instructions for Authors page before submitting a manuscript. The Article Processing Charge (APC) for publication in this open access journal is 1800 CHF (Swiss Francs). Submitted papers should be well formatted and use good English. Authors may use MDPI's English editing service prior to publication or during author revisions.
- materials and ingredient research related to cosmetics
- therapeutic options for skin, hair and body care
- product formulations and ingredients
- cosmetic olfactory research developments
- technologies in cosmetic product development
- testing of skin and hair products
- toxicological studies of cosmetic products
- in vivo and in vitro testing of cosmetic products
- pure and applied research involved in skin, hair and body cosmetics
- analytical chemistry of essential components involved in cosmetic product formulations
- biomedicine research on biologically active components
- regulatory and ethical issues in cosmetic research
- dermatology, microbiology, anatomy, physiology, immunology and biochemistry of the skin
- facial rejuvenation, laser therapy
- cosmetic surgery and related medicine techniques
- Ease of navigation: Grouping papers by topic helps scholars navigate broad scope journals more efficiently.
- Greater discoverability: Special Issues support the reach and impact of scientific research. Articles in Special Issues are more discoverable and cited more frequently.
- Expansion of research network: Special Issues facilitate connections among authors, fostering scientific collaborations.
- External promotion: Articles in Special Issues are often promoted through the journal's social media, increasing their visibility.
- e-Book format: Special Issues with more than 10 articles can be published as dedicated e-books, ensuring wide and rapid dissemination.
Further information on MDPI's Special Issue polices can be found here .
Published Papers (25 papers)
Jump to: Review

Graphical abstract

Jump to: Research

Further Information
Mdpi initiatives, follow mdpi.

Subscribe to receive issue release notifications and newsletters from MDPI journals
- Browse All Articles
- Newsletter Sign-Up
No results found in Working Knowledge
- Were any results found in one of the other content buckets on the left?
- Try removing some search filters.
- Use different search filters.
- Skip to main content
- Skip to FDA Search
- Skip to in this section menu
- Skip to footer links

The .gov means it’s official. Federal government websites often end in .gov or .mil. Before sharing sensitive information, make sure you're on a federal government site.
The site is secure. The https:// ensures that you are connecting to the official website and that any information you provide is encrypted and transmitted securely.
U.S. Food and Drug Administration
- Search
- Menu
- Cosmetics Science & Research
Cosmetics Research Projects
FDA scientists conduct cosmetic safety research and stay abreast of research by scientists elsewhere, because any action FDA takes on cosmetic safety must be based on reliable information.
Science and FDA’s Role in Cosmetic Safety
Fda scientists: who they are and what they do, fda surveys of cosmetics on the market.
- Additional Information
Under U.S. law, cosmetic products and ingredients do not need FDA approval before they go on the market. The exception is color additives (other than those used in most hair dyes). Companies and individuals who market cosmetics have a legal responsibility to ensure the safety of their products. In order to take action for safety reasons against a cosmetic on the market, we need reliable information showing that it is unsafe when consumers use it according to the directions in the labeling or in the customary or expected way.
FDA scientists involved in cosmetic safety include toxicologists, chemists, biologists, microbiologists, epidemiologists, and physicians. To address safety concerns or provide information to support regulatory actions or guidance related to cosmetics, FDA scientists will--
- conduct research,
- survey cosmetic products on the market and evaluate the findings,
- monitor reports of adverse events associated with cosmetic products,
- collaborate with scientists elsewhere in government and academia, such as the National Center for Natural Products Research at the University of Mississippi, and
- keep up with the research of other scientists.
In evaluating cosmetic safety, FDA scientists consider factors such as--
- whether a cosmetic is likely to be inhaled, swallowed, or absorbed through the skin,
- how often it is generally used,
- how long it stays in contact with the body (for example, do people leave it on their skin or quickly rinse it off?), and
- vulnerable people, such as children, the elderly, or people with compromised immune systems or other significant medical issues.
You can find the work of FDA scientists published in scientific journals such as ACS Nano , Analytical Methods , Biomaterials , Cutaneous and Ocular Toxicology , Food and Chemical Toxicology , Journal of Applied Toxicology , Journal of Chromatography A , Journal of Cosmetic Science , Journal of the American Chemical Society , Journal of the Society of Cosmetic Chemists , Nanoscale , and Soft Matter . For a list of published articles by FDA scientists on topics related to cosmetics, go to Scientific Publications by FDA Staff and enter “cosmetics” in the search field.
FDA periodically buys cosmetics to analyze them, especially if we’re aware of a potential problem. For example, we might be looking for contaminants, such as lead or harmful microorganisms, or monitoring levels of certain ingredients. Sometimes FDA scientists analyze the products, and sometimes the work is done by a contract laboratory. Depending on what we learn, we can use the information to--
- alert consumers
- support regulatory actions
- issue guidance for industry
FDA does not have the resources to sample and analyze all cosmetics on the market. Instead, we focus on particular safety concerns.
Additional Information by Topic
- 1,4-Dioxane
- Alpha Hydroxy Acids
- Bacteriological Analytical Manual (BAM), Chapter 23: Microbiological Methods for Cosmetics
- Color Additives and Cosmetics
- Potential Contaminants
- Diethanolamine
- Limiting Lead in Lipstick and Other Cosmetics
- FDA's Testing of Cosmetics for Arsenic, Cadmium, Chromium, Cobalt, Lead, Mercury, and Nickel Content
- Phthalates
- Tattoos & Permanent Makeup
120 Cosmetics Essay Topic Ideas & Examples
🏆 best cosmetics topic ideas & essay examples, ✍️ cosmetics essay topics for college, 👍 simple & easy cosmetics essay titles, 🥇 good research topics about cosmetics, ❓ research questions about makeup.
- Cosmetic Testing on Animals The surface of the skin or near the eyes of such animals is meant to simulate that of the average human and, as such, is one of easiest methods of determining whether are particular type […]
- Mac Cosmetics Company’s Social Media Use for Customer Engagement The aim of this research is to determine the role of the social media in creating customer engagement to MAC Cosmetics, focusing on the United Arab Emirates’ population.
- Foundation Makeup in the Fashion When choosing a foundation for your skin, look at the bottle, and check if the skin type is listed because it will give you information about the ingredients that will match your oily skin.
- Makeup Techniques: “The Wizard of Oz” by Victor Fleming Also, the color of his face is ochre, which reminds of sacking. In general, Lion is a rather impressive character because of the makeup he has.
- Luxury Cosmetics Branding and Pricing It is considered that “beauty products appeal to the emotions and customers tend to choose based on the product image,” yet luxury brands, such as Chanel, usually emphasize the quality of cosmetics and the technology […]
- Lush Fresh Handmade Cosmetics: Brand Image Thesis: Lush Fresh Handmade Cosmetics maintains the consistency of their brand image of a sustainable, natural, and eco-friendly beauty product by encouraging recycling, using package-less practices, choosing natural and vegan materials to produce their cosmetics, […]
- Testing Makeup Products on Rabbits The use of rabbits to test the effects of body makeup harm the mental health of scientists. Despite these harmful effects of using rabbits as test animals in the screening of beauty products, experimenting chemicals […]
- Organic and Non-Organic Makeup These groups are organic and non-organic. One of the benefits of organic makeup is its apparent safety.
- How Does the Makeup Help to Define the Character For example, a well-known movie “Dorian Gray” tells to the audience the story of the lovely young man Dorian Gray who comes to London, as now he is rich; he has got a huge inheritance […]
- Analysis of Cosmetics as a Consumer Product Improvement of communication and information technology has contributed to advanced forms of advertisement that promote the consumption and exploitation of markets down to the most localized places within the global village.
- Gender and Sexuality in Cosmetic Advertising It also assesses the correctness of truths conveyed to and the effect of these advertising images to the audience. The woman’s position to the back could be interpreted as a sign of feminine subordination.
- Competition in the U.S. Cosmetics Industry At the moment, the cosmetics market in the U. The high level of rivalry peculiar to the beauty and personal care market demands an improved understanding of the competitive landscape.
- Natural Cosmetic Skincare Products Market The trends in the UK market are quite similar to the trends in the European market. There is fierce competition for the market share of natural skincare products.
- Makeup for African American Women and Its Cultural Perspective In assuring innovation in the makeup of African American women, designers must aim to comprehend the significance and comprehension of the African art culture.
- Isopropyl Alcohol in Cosmetics and Medicine Isopropyl is synthesized in two steps: through the reaction of propylene with sulfuric acid and the consequent hydrolysis. In the context of isopropyl alternatives as sanitizers, ethyl alcohol serves as a solid option.
- Racial Discrimination Through the Cosmetics Industry The variety of preconceptions such as the hypersexuality of black women and the perception of their beauty as an unideal version of whites’ one also indicates racism.
- Black Women and the Cosmetics Industry While the industry experienced a significant increase in revenue and scale, the source of this process was the racial discrimination of black women since the marketing campaign emphasized that black women are beautiful.
- Female Consumers on Luxury Brand Clothing Over Cosmetics Consumption The findings of this study suggests that the majority of the respondents preferred to spend more on clothing, although luxury brand clothing preference was notable only to a minority of the respondents.
- Knowing One’s DNA Genetic Makeup: Pros and Cons In addition, the knowledge that one might not get a job or insurance because of their genetic makeup is stressful and depressive.
- Increment of Trans Fats in Dietary Make-Up a Misguided Move Saturated fats are markedly dangerous for human health because they increase the level of the “bad” cholesterol in the body, which increases the risk of heart attacks and other heart diseases.
- AG Hair Cosmetics: Style With Substance We are a company that’s driven by the passion, and determination of our employees and artists spreading the infectious AG philosophy.
- The Chemistry Behind Mineral Make-Up According to the US Food and Drug Administration, cosmetics include all compounds or products that are applied to the body with the aim of beautifying, boosting attractiveness or improving the physical appearance without generally altering […]
- Cultural Artifact Advertisement of Makeup People have accumulated the knowledge, values, beliefs, and attitudes to the whole scope of objects they have ever known in the cultural heritage and have become inherently able to transmit it to successive generations who […]
- Advertisement Impact on Potential Buyers in the Cosmetic Industry Thus, looking at how various products are being advertised, one cannot fail to note the seriousness of the product owners in as far as convincing the consumers to fall for the given product.
- Dr. Eris Cosmetics Company From Poland Here it must be noted that I do not agree to the statement of the Polish manager who states that the small and mid-sized manufacturers have no future in the cosmetics industry.
- Cosmetics Industry and Female Identity While many are willing to pin the blame for this attitude on the women themselves, there is plenty of evidence suggesting it is a concept perpetuated and emphasized by the cosmetics industries through the medium […]
- “Clearing Up Cosmetic Confusion” and “The Great Pretenders?” The author uses a method of observations and personal experience to describe the current state of the corporate regulations within the cosmetics industry.
- Make-Up Throughout the World History In the Fiftiesweb website, the basis of any fifties make-up look is “peaches and cream complexion”. Make-up became more bold and daring in the 1960s.
- A Second Look At Yourself With the Aid of Make-Up It can be inferred now that all make-up dates back to the past and is struck by the ludicrous reasons and ways people made use of make-up.
- Cosmetics as a Decorative Technique Used by Women At the beginning of the 20th century, makeup was used to protect and project a sense of self. For me, makeup means the possibility to create and underline a unique identity and the self.
- Halal Cosmetic Products: Innovation Management Finally, the company has to transparently state its commitment to halal cosmetics in order to attract the attention of customers and enter the competition.
- Celebrity Effect & Price Advantage: Kylie Cosmetics It features a detailed analysis of the possible reasons for it and compares the history of the enterprise to the Estee Lauder Companies, a large business that was established in the middle of the 20th […]
- New Product Campaign Pitch: Women Cosmetic The target market for the marketing campaign of the new women’s cosmetics is mainly women aging from 15 to 45 in Australia.
- La Roche-Posay Cosmetics in European Market It is within this context that the essay examines the European market of LRP by analyzing the beauty market, competition, positioning strategy, identifying brand consumers, examining market trends, and the type of innovations evident in […]
- Biofilm Prevention After Cosmetic Injection The concept of biofilm remains relatively new to dermatology, with few studies available on the formation of biofilm post-cosmetic injections; however, it is needed to explore the ways of preventing biofilm formation from reducing the […]
- Business in the US Cosmetic Retailing Industry US cosmetics retailing industry tends to expand within the domestic and international markets because of the rising level of discretionary income in the developed and developing countries.
- Entrepreneurship in the Organic Cosmetics Sphere According to the article, customers want to be green, and they want to contribute to making the world better. In conclusion, it is possible to note that the article in question provides particular points in […]
- The Peculiar Features of Organic Cosmetics’ Business Gewirtz’s “Organics of Scale” and Traber’s “Experts Sound Alarm on ‘Dirty Dozen’” are the two articles that can be used by people, who want to run a company on organic cosmetics, as these sources provide […]
- Dr. Irena Eris Cosmetics Limited Case Because the Polish cosmetics industry has become saturated with the entry of more players, the firms in the market need to adjust their business strategies both internally and externally, to survive.
- Using Facebook for Multinational Cosmetics Companies In this research proposal, I will discuss the research methodology with the focus on data collection and analysis of the appropriateness of close reading and focus group methods to answer the formulated research questions in […]
- Organic Cosmetics: Shaping Consumer Behavior This will lead to an increase in the market share enjoyed by individual organic cosmetic companies and individual brand owners of organic cosmetic products.
- Ruby & Millie Make-Up Brand Public media and press played essential role in the success of Ruby & Millie brand development as advertising is an important part of products promotion.
- Factors Affecting the Consumption of Men’s Cosmetic Products The main aim of this study was to determine the factors that affect the consumption of cosmetic products in the male population.
- Characteristics of the Skin Care and Cosmetic Industry in China L’Oreal is the market leader in Chinese cosmetic and skin care industry. The success of cosmetic and skin care products companies depends on women consumers.
- Skin Care and Cosmetic Industries in China The skin care and cosmetic industries in China are divided into male and female ones and it is possible to notice the tendency of the men’s skin care industry development increase.
- The Extent of the Cosmetic Industry in Australia The use of advertising in the promotion of the marketability of this product is a strategy that Procter & Gamble has effectively developed across Australia and the rest of the international market.
- Fashion Makeup: Blumarine Fall 2010 Collection In 1980 the Blumarine brand started to become extremely successful, and the first step to popularity, traditionally to all the great brands, was made in Milan.
- Analysis of the Contemporary Cosmetics Industry
- Connected Beauty 2018: Cosmetics of the Future
- Cosmetology: Cosmetics and New Hair Styles
- Fallacies: Cosmetics and Lash Fanatic Mascara
- Math: Cosmetics and Youth Group Leader
- Beauty Product Manufacturers and Eco-Friendly Cosmetics
- The Black Market for Counterfeit Cosmetics Is Booming and Here Is Why
- Avon Cosmetics – Place and Price Analysis
- Analysis of Effects of Using Counterfeit Cosmetics
- The Problem of Animal Testing in Cosmetics
- The Role of Corporate Social Responsibility in the Cosmetic Industry
- Overview and Analysis of Marketing of Cosmetic Product
- The State of the Science of Cosmetics Today
- The Importance of Cosmetics in Today’s World
- Analysis of the Presence of Harmful Chemicals in Cosmetics
- Overview of Social Effects of Cosmetics
- The Current Situation of the Cosmetic Industry in Asia
- Cosmetic Industry: History and Nowadays
- Synthetic Substances in the Cosmetics Industry
- Taking a Look at Beauty and Cosmetics
- M.A.C. Cosmetics: Company Profile and Overview
- FDA’s Credibility in the Cosmetics Industry
- Overview of Marketing Plan for a Cosmetics Company
- The Cosmetic Industry: Selling Illusion
- The Importance of Cosmetic Chemistry in Makeup Products
- Cosmetics and Beauty: The History of the Beauty Industry
- Consumers Beware: Safety in Cosmetic Products
- Why Do Women Use Cosmetics?
- Pros and Cons of Animal Testing on Cosmetics
- Analysis of Black up and Iman Cosmetics
- The History of Cosmetics and the Trends in the Fashion Industry
- Impact of Cosmetic Advertisements on Women
- Mercury Should Be Banned for Cosmetic Products
- We Must Eliminate Animal Testing for Cosmetics
- Chemicals in Beauty Products: The Story of Cosmetics
- Animal Testing in the Cosmetics Industry
- Advantages of Animal Testing for Cosmetics
- Fabulous Cosmetics Company: Marketing Management
- The Cosmetics Industry’s Influence on Women in Society
- Integrated Marketing Communications Plan For CoverGirl Cosmetics
- What Are the Tips and Tricks for Eye Makeup and Eyeshadow?
- How Has Sustainable Design Affected the Cosmetics Industry?
- Why Is the Black Market for Counterfeit Cosmetics Thriving?
- Does Wearing No Makeup Prevent Aging?
- What Are the 5 Main Categories of Cosmetic Products?
- Are Cosmetic Giants Segmenting the Global Cosmetics Market?
- Which Makeup Products Are Best for Summer?
- Is It Profitable to Use Peptides Obtained from Plants and Microalgae as Bioactive Compounds in Cosmetics?
- What Is Consumer Behavior Towards Cosmetics Without Toxic Chemicals?
- How Do Cosmetics Affect Confidence, Body-Esteem, and Self-Worth?
- What Are the Main Ingredients of Cosmetics?
- Should the Makeup Be Lighter or Darker Than the Skin Tone?
- What External Environmental Factors Affect the Cosmetics Industry?
- Is Organic Chemistry Used in Cosmetics?
- Why Should You Use Cosmetics Carefully?
- How Do Cosmetics Contribute to the Portrayal of False Femininity in Fashion?
- What Are the Most Common Makeup Mistakes?
- Do Makeup Courses Pave the Way to a Glamorous Career?
- What Is the Most Serious Skin Disorder Caused by Cosmetics?
- How Are the Cosmetics Industry and the Cultural Obsession with Beauty Related?
- What Are the Different Types of Skin Tones for Makeup?
- Is Going Organic the Future of Cosmetics?
- Which Cosmetics Are Absolute Must-Have for a Glamorous Look?
- Should Women Wear Makeup and Do the Hair for Themselves?
- What Are the Ingredients for Natural Cosmetics?
- Is There a Connection Between Cosmetics and Chronic Diseases?
- What Is the Ugly Truth Behind Cosmetics and Animal Testing?
- Do Cosmetics Cause Skin Disorders?
- Why Do Women’s Fashion and Cosmetics Make up a Large Part of Our Economy?
- Are Women Beautiful Without Makeup?
- Louis Vuitton Essay Topics
- Product Management Ideas
- Marketing Segmentation Questions
- Fashion Design Topics
- Photography Essay Topics
- Fast Fashion Paper Topics
- Self Esteem Research Ideas
- Procter & Gamble Topics
- Chicago (A-D)
- Chicago (N-B)
IvyPanda. (2024, March 13). 120 Cosmetics Essay Topic Ideas & Examples. https://ivypanda.com/essays/topic/cosmetics-essay-topics/
"120 Cosmetics Essay Topic Ideas & Examples." IvyPanda , 13 Mar. 2024, ivypanda.com/essays/topic/cosmetics-essay-topics/.
IvyPanda . (2024) '120 Cosmetics Essay Topic Ideas & Examples'. 13 March.
IvyPanda . 2024. "120 Cosmetics Essay Topic Ideas & Examples." March 13, 2024. https://ivypanda.com/essays/topic/cosmetics-essay-topics/.
1. IvyPanda . "120 Cosmetics Essay Topic Ideas & Examples." March 13, 2024. https://ivypanda.com/essays/topic/cosmetics-essay-topics/.
Bibliography
IvyPanda . "120 Cosmetics Essay Topic Ideas & Examples." March 13, 2024. https://ivypanda.com/essays/topic/cosmetics-essay-topics/.
Home — Essay Samples — Economics — Industry — Cosmetology
Essays on Cosmetology
Brief description of cosmetology.
Cosmetology is the study and application of beauty treatments, including hair styling, skincare, makeup, and nail care. It is a diverse and dynamic field that plays a crucial role in enhancing people's physical appearance and boosting their self-esteem.
Importance of Writing Essays on This Topic
Essays on cosmetology are essential for academic and personal exploration as they provide an opportunity to delve into the history, techniques, and cultural impact of beauty treatments. They also encourage critical thinking and analysis of the ever-evolving trends and practices within the cosmetology industry.
Tips on Choosing a Good Topic
- Focus on a specific area of cosmetology, such as the evolution of makeup trends or the impact of social media on beauty standards.
- Consider the cultural and historical significance of beauty practices in different parts of the world.
- Explore the ethical considerations and environmental impact of beauty products and treatments.
Essay Topics
- The Evolution of Hairstyling Techniques Through History
- The Influence of Social Media on Beauty Standards
- The Cultural Significance of Traditional Makeup Practices
- The Psychological Impact of Cosmetology on Self-Image
- The Ethical Dilemmas in the Beauty Industry
- The Role of Cosmetology in Empowerment and Self-Expression
- The Environmental Impact of Beauty Products
- The Intersection of Fashion and Cosmetology
- The Representation of Diversity in the Beauty Industry
- The Business of Beauty: Marketing and Consumer Behavior
Concluding Thought
Exploring cosmetology through essay writing provides an opportunity to engage critically with the evolving trends and practices within the beauty industry. By choosing a compelling topic and conducting thorough research, students can gain valuable insights into the cultural, historical, and ethical aspects of cosmetology.
Career Technical Education (cte): Cosmetology
Major problems of the cosmetology industry, made-to-order essay as fast as you need it.
Each essay is customized to cater to your unique preferences
+ experts online
Clear and Unbiased Facts About Laser Hair Removal
Therapeutics role of azadirachta indica (neem) and their active constituents in diseases prevention and treatment, effects of using cosmetic that contained mercury, how to get a glowing skin, let us write you an essay from scratch.
- 450+ experts on 30 subjects ready to help
- Custom essay delivered in as few as 3 hours
Hair Transplants: Potentially a Life Saving Procedure
Health benefits of cucumber water, how millennials impact the way spa’s and the skin care industry are doing business, functionalization of melamine sponge to make it oleophobic, get a personalized essay in under 3 hours.
Expert-written essays crafted with your exact needs in mind
How I Started Using Face Masks
Mushrooms ganoderma lucidum for energy, beauty and health, benefits and side effects of sex arousal creams, beauty product manufacturers & eco-friendly cosmetics, eco soap: a pointer to an era of convenience, why i chose cosmetology: a personal journey, the benefits of becoming a cosmetology, career choice: my future career as a cosmetology, relevant topics.
- Real Estate
- Unemployment
- American Dream
- Penny Debate
- Supply and Demand
- Universal Basic Income
- Consumerism
- Macroeconomics
By clicking “Check Writers’ Offers”, you agree to our terms of service and privacy policy . We’ll occasionally send you promo and account related email
No need to pay just yet!
We use cookies to personalyze your web-site experience. By continuing we’ll assume you board with our cookie policy .
- Instructions Followed To The Letter
- Deadlines Met At Every Stage
- Unique And Plagiarism Free
Cosmetology Research Topics

Cosmetologists help people look their best by providing professional assistance with hair care, nails, facials and makeup. You can become a cosmetologist by completing a state-approved training program and passing a state licensing examination for cosmetology. As part of your studies, you may be assigned a research paper, or you might decide to research a topic on your own to enhance classroom learning. Conducting research can also assist you in developing an area of specialization, such as scalp treatments. Spa and salon clients trust and appreciate informed cosmetologists.
Explore this article
- Hairstyle Trends
- Chemical Formulas
- Makeup Applications
- Salon Operations
1 Hairstyle Trends

You may enjoy researching the history of hairstyles. You could explore what made certain styles popular or controversial during past decades, such as long hair on men in the 1970s. You might also investigate emerging trends and what techniques you’ll need to master to create exciting new looks. Another option would be researching what hairstyles and products work best with certain hair textures, such as chemically damaged hair. You may also find it valuable to research how to best serve a culturally diverse clientele, especially if this isn’t covered well in your classes.
2 Chemical Formulas

Researching the many kinds of chemicals used in a salon can be enlightening. For example, you may want to research what chemicals are used in hair products and how they are tested for safety and effectiveness. You may also want to study alternatives to harsh chemicals, such as organic, toxin-free shampoo and conditioners. In addition, you may want to research what chemicals to avoid when working with clients who have chemical sensitivities or medical conditions such as alopecia that cause hair loss.
3 Makeup Applications

Researching new makeup colors, products and techniques can be interesting and practical. Learning how to apply makeup correctly so it’s flattering under varying light conditions is a valuable skill for cosmetologists. Many brides and girls going to prom have their makeup done professionally to look great at the event and in photographs. Or you may wish to research the types of specialized training needed to work outside the traditional salon setting. Although most cosmetologists work in hair salons, some find employment doing hair and makeup in other places, such as mortuaries or movie sets.

4 Salon Operations

Reading books on business management or interviewing successful salon operators are examples of research you can conduct to prepare for a career in cosmetology. Although you’ll likely be introduced to basic business practices in school, research can give you a better understanding of what it takes to run a salon, such as leasing space, obtaining permits, meeting state regulations, advertising, accounting, supervising employees, building a loyal clientele and using appointment software tools. Research can help you decide if you might like to manage a salon someday or work as an independent operator.
- 1 The Princeton Review: Career: Cosmetologist
- 2 U.S. Bureau of Labor Statistics: Occupational Outlook Handbook: How to Become a Barber, Hairdresser, or Cosmetologist
- 3 Lone Star College System: Cosmetology Research Guide
- 4 Santa Monica College: Cosmetology
- 5 Ferguson's Careers in Focus: Cosmetology, Fourth Edition
About the Author
Dr. Mary Dowd is a dean of students whose job includes student conduct, leading the behavioral consultation team, crisis response, retention and the working with the veterans resource center. She enjoys helping parents and students solve problems through advising, teaching and writing online articles that appear on many sites. Dr. Dowd also contributes to scholarly books and journal articles.
Related Articles

List of Supplies Needed for Cosmetology School

How to Purchase Almond Oil at the Local Pharmacy

Wax vs. Gel for Men

How to Get a Bronze Lip Look

How to Mail Donated Hair

What Would a Girl Wear to a "Miami Vice" Party?

How to Become an Amateur Fashion Model

Required Education for Professional Cake Decorators

How to Do a Pompadour Ponytail

Advantages & Disadvantages of a Cosmetologist

Postsecondary Training or Schooling for Cosmetology...

Massage Etiquette for Men

How to Describe an Emo Cut to a Hairstylist

The History of Hair Relaxers

How to Donate Hair for Breast Cancer Patients

How to Make Your Horse's Hair Grow Fast

How to Identify Choctaw Indian Arrowheads

How to Stop a Farmer's Tan

How to Keep Bangs Straight in Humidity
Regardless of how old we are, we never stop learning. Classroom is the educational resource for people of all ages. Whether you’re studying times tables or applying to college, Classroom has the answers.
- Accessibility
- Terms of Use
- Privacy Policy
- Copyright Policy
- Manage Preferences
© 2020 Leaf Group Ltd. / Leaf Group Media, All Rights Reserved. Based on the Word Net lexical database for the English Language. See disclaimer .
Suggestions or feedback?
MIT News | Massachusetts Institute of Technology
- Machine learning
- Sustainability
- Black holes
- Classes and programs
Departments
- Aeronautics and Astronautics
- Brain and Cognitive Sciences
- Architecture
- Political Science
- Mechanical Engineering
Centers, Labs, & Programs
- Abdul Latif Jameel Poverty Action Lab (J-PAL)
- Picower Institute for Learning and Memory
- Lincoln Laboratory
- School of Architecture + Planning
- School of Engineering
- School of Humanities, Arts, and Social Sciences
- Sloan School of Management
- School of Science
- MIT Schwarzman College of Computing
Study reveals the benefits and downside of fasting
Press contact :, media download.
*Terms of Use:
Images for download on the MIT News office website are made available to non-commercial entities, press and the general public under a Creative Commons Attribution Non-Commercial No Derivatives license . You may not alter the images provided, other than to crop them to size. A credit line must be used when reproducing images; if one is not provided below, credit the images to "MIT."
Previous image Next image
Low-calorie diets and intermittent fasting have been shown to have numerous health benefits: They can delay the onset of some age-related diseases and lengthen lifespan, not only in humans but many other organisms.
Many complex mechanisms underlie this phenomenon. Previous work from MIT has shown that one way fasting exerts its beneficial effects is by boosting the regenerative abilities of intestinal stem cells, which helps the intestine recover from injuries or inflammation.
In a study of mice, MIT researchers have now identified the pathway that enables this enhanced regeneration, which is activated once the mice begin “refeeding” after the fast. They also found a downside to this regeneration: When cancerous mutations occurred during the regenerative period, the mice were more likely to develop early-stage intestinal tumors.
“Having more stem cell activity is good for regeneration, but too much of a good thing over time can have less favorable consequences,” says Omer Yilmaz, an MIT associate professor of biology, a member of MIT’s Koch Institute for Integrative Cancer Research, and the senior author of the new study.
Yilmaz adds that further studies are needed before forming any conclusion as to whether fasting has a similar effect in humans.
“We still have a lot to learn, but it is interesting that being in either the state of fasting or refeeding when exposure to mutagen occurs can have a profound impact on the likelihood of developing a cancer in these well-defined mouse models,” he says.
MIT postdocs Shinya Imada and Saleh Khawaled are the lead authors of the paper, which appears today in Nature .
Driving regeneration
For several years, Yilmaz’s lab has been investigating how fasting and low-calorie diets affect intestinal health. In a 2018 study , his team reported that during a fast, intestinal stem cells begin to use lipids as an energy source, instead of carbohydrates. They also showed that fasting led to a significant boost in stem cells’ regenerative ability.
However, unanswered questions remained: How does fasting trigger this boost in regenerative ability, and when does the regeneration begin?
“Since that paper, we’ve really been focused on understanding what is it about fasting that drives regeneration,” Yilmaz says. “Is it fasting itself that’s driving regeneration, or eating after the fast?”
In their new study, the researchers found that stem cell regeneration is suppressed during fasting but then surges during the refeeding period. The researchers followed three groups of mice — one that fasted for 24 hours, another one that fasted for 24 hours and then was allowed to eat whatever they wanted during a 24-hour refeeding period, and a control group that ate whatever they wanted throughout the experiment.
The researchers analyzed intestinal stem cells’ ability to proliferate at different time points and found that the stem cells showed the highest levels of proliferation at the end of the 24-hour refeeding period. These cells were also more proliferative than intestinal stem cells from mice that had not fasted at all.
“We think that fasting and refeeding represent two distinct states,” Imada says. “In the fasted state, the ability of cells to use lipids and fatty acids as an energy source enables them to survive when nutrients are low. And then it’s the postfast refeeding state that really drives the regeneration. When nutrients become available, these stem cells and progenitor cells activate programs that enable them to build cellular mass and repopulate the intestinal lining.”
Further studies revealed that these cells activate a cellular signaling pathway known as mTOR, which is involved in cell growth and metabolism. One of mTOR’s roles is to regulate the translation of messenger RNA into protein, so when it’s activated, cells produce more protein. This protein synthesis is essential for stem cells to proliferate.
The researchers showed that mTOR activation in these stem cells also led to production of large quantities of polyamines — small molecules that help cells to grow and divide.
“In the refed state, you’ve got more proliferation, and you need to build cellular mass. That requires more protein, to build new cells, and those stem cells go on to build more differentiated cells or specialized intestinal cell types that line the intestine,” Khawaled says.
Too much of a good thing
The researchers also found that when stem cells are in this highly regenerative state, they are more prone to become cancerous. Intestinal stem cells are among the most actively dividing cells in the body, as they help the lining of the intestine completely turn over every five to 10 days. Because they divide so frequently, these stem cells are the most common source of precancerous cells in the intestine.
In this study, the researchers discovered that if they turned on a cancer-causing gene in the mice during the refeeding stage, they were much more likely to develop precancerous polyps than if the gene was turned on during the fasting state. Cancer-linked mutations that occurred during the refeeding state were also much more likely to produce polyps than mutations that occurred in mice that did not undergo the cycle of fasting and refeeding.
“I want to emphasize that this was all done in mice, using very well-defined cancer mutations. In humans it’s going to be a much more complex state,” Yilmaz says. “But it does lead us to the following notion: Fasting is very healthy, but if you’re unlucky and you’re refeeding after a fasting, and you get exposed to a mutagen, like a charred steak or something, you might actually be increasing your chances of developing a lesion that can go on to give rise to cancer.”
Yilmaz also noted that the regenerative benefits of fasting could be significant for people who undergo radiation treatment, which can damage the intestinal lining, or other types of intestinal injury. His lab is now studying whether polyamine supplements could help to stimulate this kind of regeneration, without the need to fast.
“This fascinating study provides insights into the complex interplay between food consumption, stem cell biology, and cancer risk,” says Ophir Klein, a professor of medicine at the University of California at San Francisco and Cedars-Sinai Medical Center, who was not involved in the study. “Their work lays a foundation for testing polyamines as compounds that may augment intestinal repair after injuries, and it suggests that careful consideration is needed when planning diet-based strategies for regeneration to avoid increasing cancer risk.”
The research was funded, in part, by a Pew-Stewart Trust Scholar award, the Marble Center for Cancer Nanomedicine, the Koch Institute-Dana Farber/Harvard Cancer Center Bridge Project, and the MIT Stem Cell Initiative.
Share this news article on:
Press mentions, medical news today.
A new study led by researchers at MIT suggests that fasting and then refeeding stimulates cell regeneration in the intestines, reports Katharine Lang for Medical News Today . However, notes Lang, researchers also found that fasting “carries the risk of stimulating the formation of intestinal tumors.”
Prof. Ömer Yilmaz and his colleagues have discovered the potential health benefits and consequences of fasting, reports Max Kozlov for Nature . “There is so much emphasis on fasting and how long to be fasting that we’ve kind of overlooked this whole other side of the equation: what is going on in the refed state,” says Yilmaz.
MIT researchers have discovered how fasting impacts the regenerative abilities of intestinal stem cells, reports Ed Cara for Gizmodo . “The major finding of our current study is that refeeding after fasting is a distinct state from fasting itself,” explain Prof. Ömer Yilmaz and postdocs Shinya Imada and Saleh Khawaled. “Post-fasting refeeding augments the ability of intestinal stem cells to, for example, repair the intestine after injury.”
Previous item Next item
Related Links
- Omer Yilmaz
- Koch Institute
- Department of Biology
Related Topics
Related articles.
How early-stage cancer cells hide from the immune system
Study links certain metabolites to stem cell function in the intestine
Fasting boosts stem cells’ regenerative capacity

How diet influences colon cancer
More mit news.

Study of disordered rock salts leads to battery breakthrough
Read full story →

Toward a code-breaking quantum computer

Uphill battles: Across the country in 75 days

3 Questions: From the bench to the battlefield

Duane Boning named vice provost for international activities

Q&A: Undergraduate admissions in the wake of the 2023 Supreme Court ruling
- More news on MIT News homepage →
Massachusetts Institute of Technology 77 Massachusetts Avenue, Cambridge, MA, USA
- Map (opens in new window)
- Events (opens in new window)
- People (opens in new window)
- Careers (opens in new window)
- Accessibility
- Social Media Hub
- MIT on Facebook
- MIT on YouTube
- MIT on Instagram
- SUGGESTED TOPICS
- The Magazine
- Newsletters
- Managing Yourself
- Managing Teams
- Work-life Balance
- The Big Idea
- Data & Visuals
- Reading Lists
- Case Selections
- HBR Learning
- Topic Feeds
- Account Settings
- Email Preferences
Research: Consumers Spend Loyalty Points and Cash Differently
- So Yeon Chun,
- Freddy Lim,
- Ville Satopää

Your loyalty strategy needs to consider four ways people value points.
Do consumers treat loyalty points the same way that they treat traditional money? And, how do they choose to spend one versus the other? The authors of this article present research findings from their analysis of data describing over 29,000 unique loyalty points earning and spending transactions made during two recent years by 500 airline loyalty program consumers. They found that points users fell into four distinct categories: 1) Money advocates, who prefer cash over points, even when their value is identical in terms of purchasing power; 2) Currency impartialists, who regard points and cash interchangeably, valuing them equally based on their financial worth; 3) Point gamers, who actively seek out the most advantageous point redemption opportunities, opting to spend points particularly when their value significantly surpasses that of cash; and 4) Point lovers, who value points more than money even if their purchase power is the same or lower. This article explores the strategic implications of these findings for companies that manage loyalty programs.
In the years since The Economist spotlighted the astonishing scale of loyalty points — particularly frequent-flyer miles — as a potential global currency rivaling traditional money in 2005, usage has grown rapidly in size and scope. For example, the number of flight redemptions at Southwest Airlines doubled from 5.4 million in 2013 (representing 9.5% of revenue passenger miles) to 10.9 million in 2023 (representing 16.3% of revenue passenger miles).
- SC So Yeon Chun is an Associate Professor of Technology & Operations Management at INSEAD, a global business school with campuses in Abu Dhabi, France, and Singapore.
- FL Freddy Lim is an Assistant Professor of Information Systems and Analytics at the National University of Singapore, School of Computing in Singapore
- VS Ville Satopää is an Associate Professor of Technology and Operations Management at INSEAD, a global business school with campuses in Abu Dhabi, France, and Singapore.
Partner Center
- Alzheimer's disease & dementia
- Arthritis & Rheumatism
- Attention deficit disorders
- Autism spectrum disorders
- Biomedical technology
- Diseases, Conditions, Syndromes
- Endocrinology & Metabolism
- Gastroenterology
- Gerontology & Geriatrics
- Health informatics
- Inflammatory disorders
- Medical economics
- Medical research
- Medications
- Neuroscience
- Obstetrics & gynaecology
- Oncology & Cancer
- Ophthalmology
- Overweight & Obesity
- Parkinson's & Movement disorders
- Psychology & Psychiatry
- Radiology & Imaging
- Sleep disorders
- Sports medicine & Kinesiology
- Vaccination
- Breast cancer
- Cardiovascular disease
- Chronic obstructive pulmonary disease
- Colon cancer
- Coronary artery disease
- Heart attack
- Heart disease
- High blood pressure
- Kidney disease
- Lung cancer
- Multiple sclerosis
- Myocardial infarction
- Ovarian cancer
- Post traumatic stress disorder
- Rheumatoid arthritis
- Schizophrenia
- Skin cancer
- Type 2 diabetes
- Full List »
share this!
August 23, 2024
This article has been reviewed according to Science X's editorial process and policies . Editors have highlighted the following attributes while ensuring the content's credibility:
fact-checked
peer-reviewed publication
trusted source
Research shows drop in unhealthy food ads during kids' TV shows
by Rob Mitchum, University of Illinois at Chicago

Children's exposure to food and drink ads during kids' TV shows has dropped substantially since food and beverage makers pledged to stop advertising unhealthy fare during children's TV shows. Yet, according to research from the University of Illinois Chicago, children under 12 still see more than 1,000 food-related ads a year, most of them for unhealthy products.
For the study , published in JAMA Network Open , researchers analyzed television ratings and advertising data from 2013 through 2022. The study authors found that a dramatic decline in food and drink advertisements during kids' shows did not fully eliminate children's exposure to ads for products high in saturated fat, trans fat, total sugars and sodium.
"Kids are still seeing about a thousand ads per year on other programs, and the majority of ads that kids see are still for unhealthy products," said Lisa Powell, distinguished professor and director of health policy and administration in the UIC School of Public Health. "This is important as the World Health Organization has recognized that reducing children's exposure to unhealthy food and beverage advertisements is a key strategy for improving both children's diets and health."
In 2006, a group of food, beverage and restaurant companies pledged to only advertise healthy products on children's television programming, defined as shows where at least 35% of viewers are under the age of 12. Later revisions in 2014 and 2020 established nutritional criteria for what qualifies as unhealthy and therefore should not be advertised to young audiences.
Using television ratings data from The Nielsen Company, UIC researchers found that following changes in companies' self-regulation, the number of general food and beverage commercials seen during children's programs fell by over 95%. However, they found that 60% of the remaining food and beverage ads were still for unhealthy products.
And overall, kids under the age of 12 still saw more than 1,000 food-related advertisements per year, on average. Because of the steep decline in food and beverage ads during children's shows, as much as 90% of this exposure came from watching shows with lower child-audience shares.
That shift suggests that regulations against advertising unhealthy food and beverages during hours when children are likely to watch television would be more effective than focusing restrictions specifically on children's shows, the authors write.
The researchers also found a persistent racial difference in exposure to food-related advertisements. While the number of these ads seen by both Black and white children declined from 2013 to 2022, Black children saw significantly more advertisements than their white counterparts, due in part to more time spent watching television.
In general, children's time watching television has declined, indicating the need for research on their exposure to advertising on other media. Powell's group is in the early stages of launching a new project to measure the ads children encounter through social media platforms and digital entertainment.
"We know that the media kids consume is changing. They're spending more time on their mobile devices, whether it be a tablet or a phone, and they're seeing a lot of ads," Powell said. "We really need to understand where else the food companies target kids and what they're seeing."
In addition to Powell, UIC co-authors include Julien Leider, Rebecca Schermbeck and Aline Vandenbroeck, along with University of Connecticut co-author Jennifer Harris. The study was supported by grants from Bloomberg Philanthropies and the Robert Wood Johnson Foundation.
Explore further
Feedback to editors

Study identifies metabolic switch essential for generation of memory T cells and anti-tumor immunity
18 hours ago

Multiple sclerosis appears to protect against Alzheimer's disease
Aug 23, 2024

Good sleep habits important for overweight adults, study suggests

Mediterranean diet supplement can affect epigenetics associated with healthy aging

New method for quantifying boredom in the body during temporary stress

Cancer researchers develop new method that uses internal clock inside tumor cells to optimize therapies

Strength training activates cellular waste disposal, interdisciplinary research reveals

Being a 'weekend warrior' could be as good for brain health as exercising throughout the week
Simple blood test for Alzheimer's disease could change how the disease is detected and diagnosed

Chlamydia can settle in the intestine, organoid experiments reveal
Related stories.

Industry self-regulation of food advertising doesn't work, worldwide study shows
May 15, 2019

TV ads still push unhealthy foods at kids
Nov 6, 2017

New study finds fast-food companies spending more on ads, targeting Black and Hispanic youth
Jun 17, 2021

Junk food is promoted online to appeal to kids and target young men, study shows
Jul 12, 2024

New report reveals industry-led initiative restricting products advertised to kids fails to protect them
Apr 5, 2022

Adolescents bombarded with junk food marketing on social media
Jul 21, 2021
Recommended for you

Weight loss drug's heart benefits extend to people with heart failure, study finds
Aug 22, 2024

Mouse model suggests proteins cause damage in fetal abdominal inflammation

Clinical trial in Ireland challenges beliefs about Ozempic and similar new obesity treatments

Most baby foods in US grocery stores are unhealthy, study shows
Aug 21, 2024

Giving an antibiotic to all children under 5 in Africa would save lives, researchers say
Let us know if there is a problem with our content.
Use this form if you have come across a typo, inaccuracy or would like to send an edit request for the content on this page. For general inquiries, please use our contact form . For general feedback, use the public comments section below (please adhere to guidelines ).
Please select the most appropriate category to facilitate processing of your request
Thank you for taking time to provide your feedback to the editors.
Your feedback is important to us. However, we do not guarantee individual replies due to the high volume of messages.
E-mail the story
Your email address is used only to let the recipient know who sent the email. Neither your address nor the recipient's address will be used for any other purpose. The information you enter will appear in your e-mail message and is not retained by Medical Xpress in any form.
Newsletter sign up
Get weekly and/or daily updates delivered to your inbox. You can unsubscribe at any time and we'll never share your details to third parties.
More information Privacy policy
Donate and enjoy an ad-free experience
We keep our content available to everyone. Consider supporting Science X's mission by getting a premium account.
E-mail newsletter
Numbers, Facts and Trends Shaping Your World
Read our research on:
Full Topic List
Regions & Countries
- Publications
- Our Methods
- Short Reads
- Tools & Resources
Read Our Research On:
Race and LGBTQ Issues in K-12 Schools
What teachers, teens and the u.s. public say about current curriculum debates, table of contents.
- What do teachers think students should learn about slavery and gender identity?
- Should parents be able to opt their children out of learning about certain topics?
- How often do topics related to race and LGBTQ issues come up in the classroom?
- How do teachers’ views differ by party?
- What teachers think students should learn about slavery and gender identity
- Should parents be able to opt their children out of learning about race and LGBTQ issues?
- Influence over curriculum
- What teens want to learn about slavery
- What teens want to learn about gender identity
- 4. Public views on parents opting their children out of learning about race and LGBTQ issues
- Acknowledgments
- Teacher survey methodology
- Teen survey methodology
- General public survey methodology
Pew Research Center conducted this study to better understand how public K-12 teachers, teens and the American public see topics related to race, sexual orientation and gender identity playing out in the classroom.
The bulk of the analysis in this report is based on an online survey of 2,531 U.S. public K-12 teachers conducted from Oct. 17 to Nov. 14, 2023. The teachers surveyed are members of RAND’s American Teacher Panel, a nationally representative panel of public school K-12 teachers recruited through MDR Education. Survey data is weighted to state and national teacher characteristics to account for differences in sampling and response to ensure they are representative of the target population.
For the questions for the general public, we surveyed 5,029 U.S. adults from Nov. 9 to Nov. 16, 2023. The adults surveyed are members of the Ipsos KnowledgePanel, a nationally representative online survey panel. Panel members are randomly recruited through probability-based sampling, and households are provided with access to the Internet and hardware if needed. To ensure that the results of this survey reflect a balanced cross section of the nation, the data is weighted to match the U.S. adult population by gender, age, education, race and ethnicity and other categories.
For questions for teens, we conducted an online survey of 1,453 U.S. teens from Sept. 26 to Oct. 23, 2023, through Ipsos. Ipsos recruited the teens via their parents, who were part of its KnowledgePanel. The survey was weighted to be representative of U.S. teens ages 13 to 17 who live with their parents by age, gender, race and ethnicity, household income, and other categories. The survey on teens was reviewed and approved by an external institutional review board (IRB), Advarra, an independent committee of experts specializing in helping to protect the rights of research participants.
Here are the questions used for this report , along with responses, and the survey methodology .
Throughout the report, references to White, Black and Asian adults include those who are not Hispanic and identify as only one race. Hispanics are of any race. The views and experiences of teachers and teens who are Asian American or part of other racial and ethnic groups are not analyzed separately in this report due to sample limitations. Data for these groups is incorporated into the general population figures throughout the report.
All references to party affiliation include those who lean toward that party. Republicans include those who identify as Republicans and those who say they lean toward the Republican Party. Democrats include those who identify as Democrats and those who say they lean toward the Democratic Party.
Political leaning of school districts is based on whether the majority of those residing in the school district voted for Republican Donald Trump or Democrat Joe Biden in the 2020 presidential election.
Amid national debates about what schools are teaching , we asked public K-12 teachers, teens and the American public how they see topics related to race, sexual orientation and gender identity playing out in the classroom.
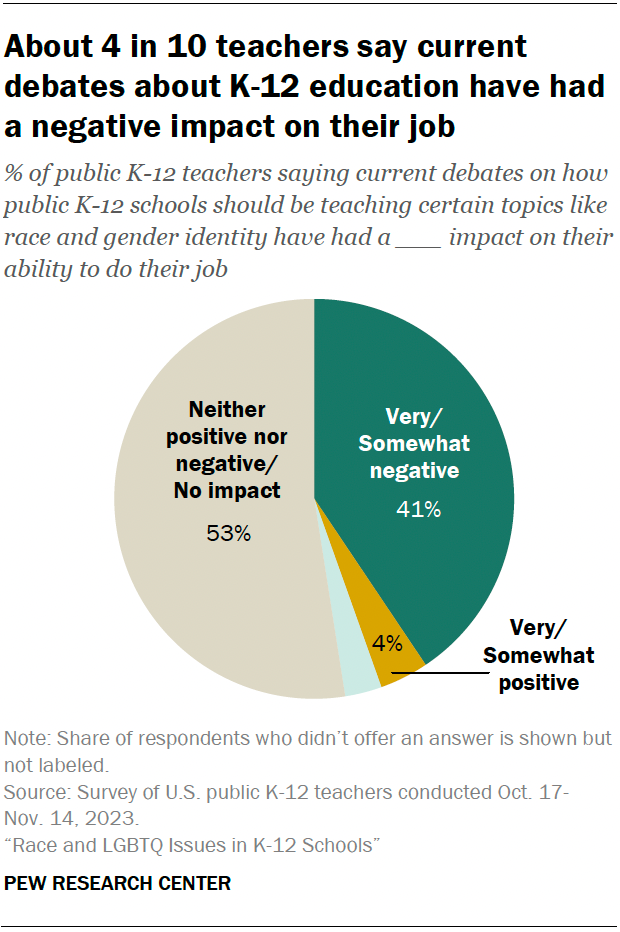
A sizeable share of teachers (41%) say these debates have had a negative impact on their ability to do their job. Just 4% say these debates have had a positive impact, while 53% say the impact has been neither positive nor negative or that these debates have had no impact.
And 71% of teachers say teachers themselves don’t have enough influence over what’s taught in public schools in their area.
In turn, a majority of teachers (58%) say their state government has too much influence over this. And more say the federal government, the local school board and parents have too much influence than say they don’t have enough.
Most of the findings in this report come from a survey of 2,531 U.S. public K-12 teachers conducted Oct. 17-Nov. 14, 2023, using the RAND American Teacher Panel. 1 The survey looks at teachers’ views on:
- Race and LGBTQ issues in the classroom ( Chapter 1 )
- Current debates over what schools should be teaching and the role of key groups ( Chapter 2 )
It follows a fall 2022 survey of K-12 parents that explored similar topics.
This report also includes some findings from a survey of U.S. teens ages 13 to 17 ( Chapter 3 ) and a survey of U.S. adults ( Chapter 4 ). For details about these surveys, refer to the Methodology section of this report. Among the key findings:
- 38% of teens say they feel comfortable when topics related to racism or racial inequality come up in class (among those who say these topics have come up). A smaller share (29%) say they feel comfortable when topics related to sexual orientation or gender identity come up.
- Among the American public , more say parents should be able to opt their children out of learning about LGBTQ issues than say the same about topics related to race (54% vs. 34%).
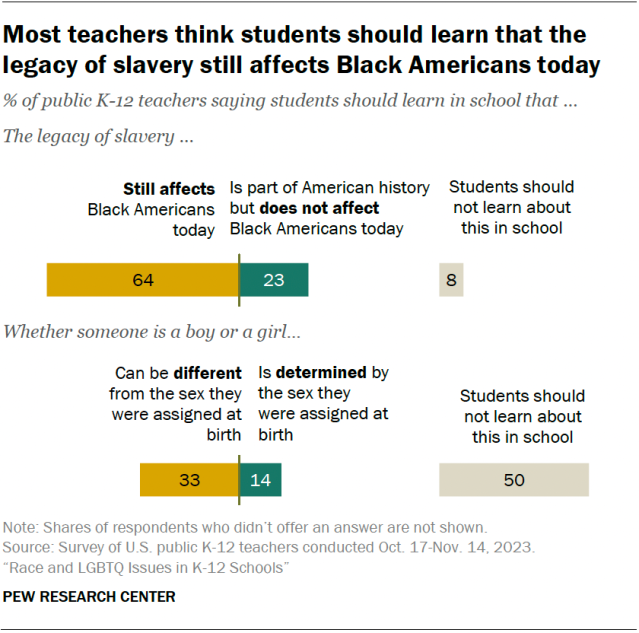
We asked public K-12 teachers what they think students should learn in school about two topics in particular:
- Whether the legacy of slavery still affects the position of Black people in American society today.
- Whether a person’s gender can be different from or is determined by their sex at birth.
For these questions, elementary, middle and high school teachers were asked about elementary, middle and high school students, respectively.
The legacy of slavery
Most teachers (64%) say students should learn that the legacy of slavery still affects the position of Black people in American society today.
About a quarter (23%) say students should learn that slavery is part of American history but no longer affects the position of Black people in American society. Just 8% say students shouldn’t learn about this topic in school at all.
Majorities of elementary, middle and high school teachers say students should learn that the legacy of slavery still has an impact on the lives of Black Americans.
Gender identity
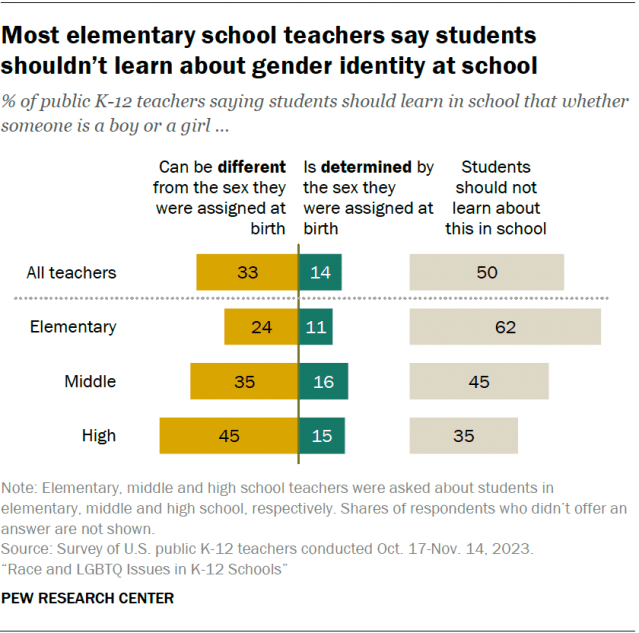
When it comes to teaching about gender identity – specifically whether a person’s gender can be different from or is determined by their sex assigned at birth – half of public K-12 teachers say students shouldn’t learn about this in school.
A third of teachers think students should learn that someone can be a boy or a girl even if that is different from the sex they were assigned at birth.
A smaller share (14%) say students should learn that whether someone is a boy or a girl is determined by their sex at birth.
Views differ among elementary, middle and high school teachers. But teachers across the three levels are more likely to say students should learn that a person’s gender can be different from their sex at birth than to say students should learn gender is determined by sex at birth.
Most elementary school teachers (62%) say students shouldn’t learn about gender identity in school. This is much larger than the shares of middle and high school teachers who say the same (45% and 35%).
What parents and teens say
Parents of K-12 students are more divided on what their children should learn in school about these topics.
In the 2022 survey , 49% of parents said they’d rather their children learn that the legacy of slavery still affects the position of Black people in American society today, while 42% said they’d rather their children learn that slavery no longer affects Black Americans.
When it comes to gender identity, 31% of parents said they’d rather their children learn that gender can be different from sex at birth. An identical share said they would rather their children learn gender is determined by sex at birth. Another 37% of parents said their children shouldn’t learn about gender identity in school.
Teens, like parents, are more divided than teachers on these questions. About half of teens (48%) say they’d rather learn that the legacy of slavery still affects the position of Black Americans today. Four-in-ten would prefer to learn that slavery no longer affects Black Americans.
And teens are about evenly divided when it comes to what they prefer to learn about gender identity. A quarter say they’d rather learn that a person’s gender can be different from their sex at birth; 26% would prefer to learn that gender is determined by sex at birth. About half (48%) say they shouldn’t learn about gender identity in school.
For more on teens’ views about what they prefer to learn in school about each of these topics, read Chapter 3 of this report.
Most public K-12 teachers (60%) say parents should not be able to opt their children out of learning about racism or racial inequality in school, even if the way these topics are taught conflicts with the parents’ beliefs. A quarter say parents should be able to opt their children out of learning about these topics.
In contrast, more say parents should be able to opt their children out of learning about sexual orientation or gender identity (48%) than say parents should not be able to do this (33%).
On topics related to both race and LGBTQ issues, elementary and middle school teachers are more likely than high school teachers to say parents should be able to opt their children out.
How teachers’ views compare with the public’s views
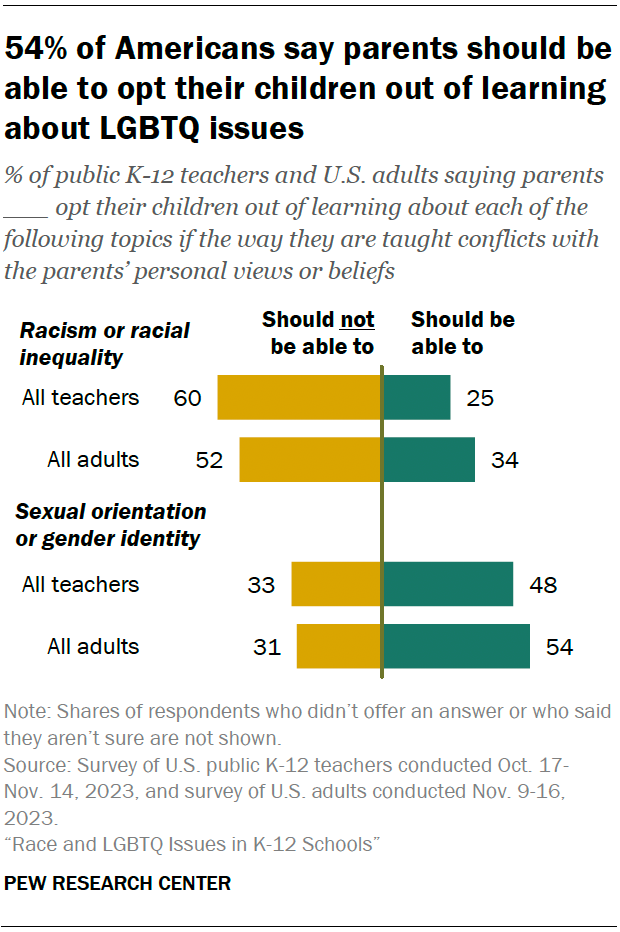
Like teachers, Americans overall are more likely to say parents should be able to opt their children out of learning about sexual orientation or gender identity (54%) than to say they should be able to opt their children out of learning about racism or racial inequality (34%).
Across both issues, Americans overall are somewhat more likely than teachers to say parents should be able to opt their children out.
For more on the public’s views, read Chapter 4 of this report.
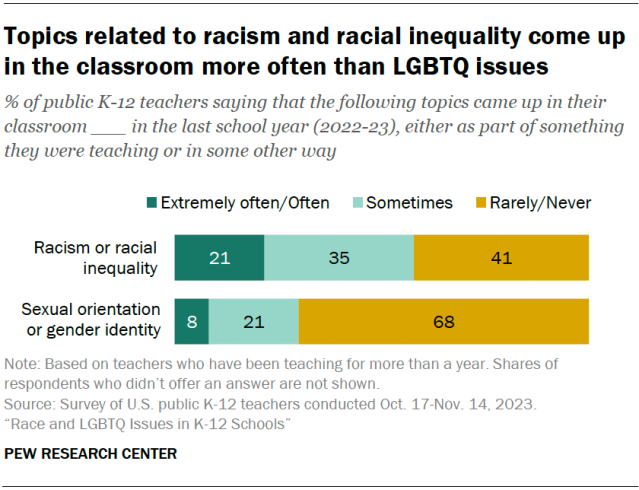
Most teachers who’ve been teaching for more than a year (68%) say the topics of sexual orientation and gender identity rarely or never came up in their classroom in the 2022-23 school year. About one-in-five (21%) say these topics came up sometimes, and 8% say they came up often or extremely often.
Topics related to racism or racial inequality come up more frequently. A majority of teachers (56%) say these topics came up at least sometimes in their classroom, with 21% saying they came up often or extremely often.
These topics are more likely to come up in secondary school than in elementary school classrooms.
As is the case among parents of K-12 students and the general public, teachers’ views on how topics related to race and LGBTQ issues should play out in the classroom differ by political affiliation.
- What students should learn about slavery: 85% of Democratic and Democratic-leaning teachers say students should learn that the legacy of slavery still affects the position of Black people in American society today. This compares with 35% of Republican and Republican-leaning teachers who say the same.
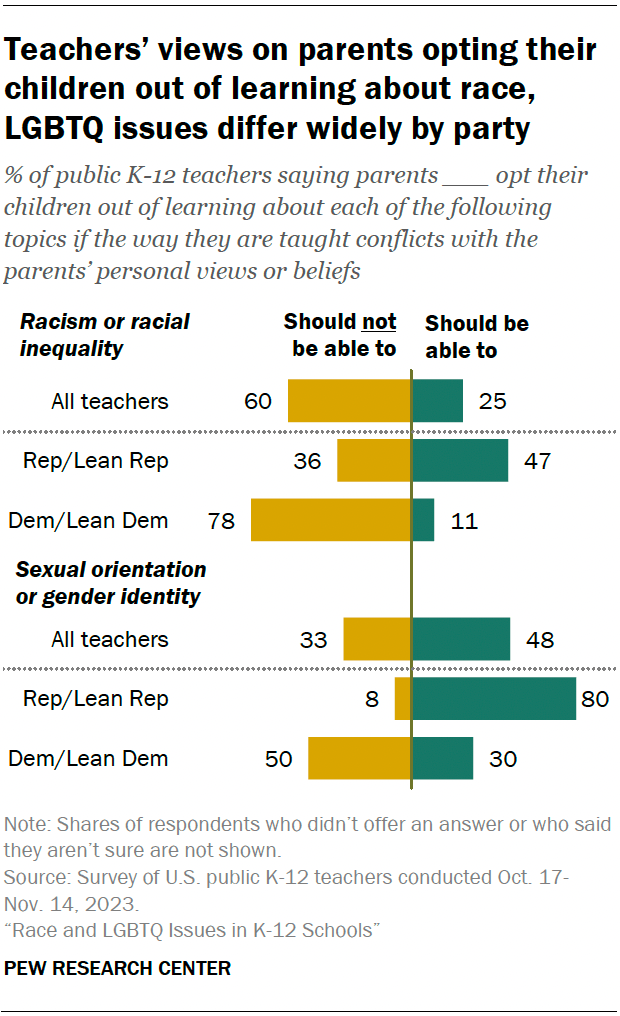
- What students should learn about gender identity: Democratic teachers are far more likely than Republican teachers to say students should learn that a person’s gender can be different from the sex they were assigned at birth (53% vs. 5%). Most Republican teachers (69%) say students shouldn’t learn about gender identity in school.
- Parents opting their children out of learning about these topics: 80% of Republican teachers say parents should be able to opt their children out of learning about LGBTQ issues, compared with 30% of Democratic teachers. And while 47% of Republican teachers say parents should be able to opt their children out of learning about racism and racial inequality, just 11% of Democratic teachers say this.
A majority of public K-12 teachers (58%) identify with or lean toward the Democratic Party. About a third (35%) identify with or lean toward the GOP. Americans overall are more evenly divided: 47% are Democrats or Democratic leaners, and 45% are Republicans or Republican leaners .
- For details, refer to the Methodology section of the report. ↩
Sign up for our weekly newsletter
Fresh data delivery Saturday mornings
Sign up for The Briefing
Weekly updates on the world of news & information
- Education & Gender
- LGBTQ Acceptance
- Race & Ethnicity
Who Are You? The Art and Science of Measuring Identity
Facts about the u.s. black population, 22 states have ever elected a black woman to congress, trust in america: do americans trust the police, black americans have made gains in u.s. political leadership, but gaps remain, most popular, report materials.
901 E St. NW, Suite 300 Washington, DC 20004 USA (+1) 202-419-4300 | Main (+1) 202-857-8562 | Fax (+1) 202-419-4372 | Media Inquiries
Research Topics
- Email Newsletters
ABOUT PEW RESEARCH CENTER Pew Research Center is a nonpartisan fact tank that informs the public about the issues, attitudes and trends shaping the world. It conducts public opinion polling, demographic research, media content analysis and other empirical social science research. Pew Research Center does not take policy positions. It is a subsidiary of The Pew Charitable Trusts .
© 2024 Pew Research Center
Makeup artist
Job posting for makeup artist at san francisco institute of esthetics and cosmetology.
Collective Press is looking for a creative make-up artist / on-camera model / host to join our SF office. We are a digital media startup making online videos in lifestyle verticals : make-up, hair styling, nail painting, cooking, DIY, hacks, and more.
You’ll be immersed in learning the best of digital and social media content.
This is a part-time / full-time position in the SF office.
Responsibilities
- Work on video shoots as stylist / make-up artist & on-camera model / host
- Contribute ideas for new makeup / beauty videos
- Assist with production of styling videos to spec from start to finish : shopping and prepping materials in advance, being in the video (the hands and face ), and video wrap up
- Be able to execute on the make-up techniques both on yourself and on others
- Ability to work with other members of the team to help produce video to spec
- Manage and order all supplies for videos in an organized manner
- Other admin tasks as assigned
Requirements
- Experience and passion for make-up / styling
- Strong, interesting, and dynamic on-camera personality and appearance
- Be able to operate a DSLR camera (or open to learning how)
- Positive, can-do attitude
- Self-starter and team player
- Must be authorized to legally work in the United States.
Please apply with the following :
Qualifications, skills, and all relevant experience needed for this role can be found in the full description below.
2) Sample video demonstrating your make-up skills. This is a requirement. Make a short 1-2 minute video (can be taken with your mobile phone) to show us what you can do makeup-wise.
There must be at least a portion of the video where you are speaking.
3) Samples of your makeup work
About Collective Press
Collective Press is a digital media startup company that was founded in 2014 by serial entrepreneurs from Berkeley, Stanford, and UPenn.
It owns several digital publications spanning a variety of topics : news, trending stories, home, cooking, DIY, and more.
Pursuant to the San Francisco Fair Chance Ordinance, Collective Press will consider for employment qualified applicants with arrest and conviction records.
J-18808-Ljbffr
Last updated : 2024-08-21
Apply for this job
Receive alerts for other Makeup artist job openings
Report this Job
Sign up to receive alerts about other jobs that are on the Makeup artist career path.
Click the checkbox next to the jobs that you are interested in.
Sign up to receive alerts about other jobs with skills like those required for the Makeup artist .
Customer Service Skill
- Server Income Estimation: $20,772 - $27,848
- Blackjack Dealer Income Estimation: $21,027 - $25,942
Sales Promotions Skill
- Spa Attendant Income Estimation: $25,394 - $30,215
- Slot Club Representative Income Estimation: $27,194 - $37,814
Not the job you're looking for? Here are some other Makeup artist jobs in the San Francisco, CA area that may be a better fit.
We don't have any other makeup artist jobs in the san francisco, ca area right now..
Hair And makeup Artist
Quince , San Francisco, CA
Freelance Makeup Artist
Charlotte Tilbury , San Francisco, CA
More than 26 million viewers tuned in to the final night of the DNC convention: Nielsen
Nielsen estimates 26.2 million viewers tuned in to the final night of Democratic National Convention.
The four-day event in Chicago drew audiences of at least 20 million every day, according to event averages calculated by Nielsen, as celebrities including Michelle Obama and Oprah Winfrey took the stage,
On Thursday as Democratic presidential nominee Kamala Harris delivered her acceptance speech , that average went up to 28.9 million, Nielsen said in a Friday press release.
By comparison, the final night of the Republican National Convention drew an average audience of 25.4 million viewers, Nielsen data shows .
Convention viewership has been waning over recent decades, but experts anticipated a small bump for this year's convention due to the tumultuous election cycle.
A special guest at the DNC? These wrong answers (only) were far more entertaining
Looking back at previous RNC and DNC ratings
For convention audience, Nielsen measures the percentage of television-owning households that tuned in. The Nielsen data captured audience from 15 television networks for Thursday night.
The household ratings for the fourth and final night of the 2024 Democratic convention was 15.2, Nielsen's release stated.
Here are the day-by-day Nielsen household ratings for the recent conventions:
- 2024 DNC Day 4 - 15.2
- 2024 DNC Day 3 - 11.9
- 2024 DNC Day 2 - 12.5
- 2024 DNC Day 1 - 12.0
- 2024 RNC Day 4 - 14.3
- 2024 RNC Day 3 - 10.6
- 2024 RNC Day 2 - 8.9
- 2024 RNC Day 1 - 10.6
- 2020 DNC Day 4 - 15.6
- 2020 DNC Day 3 - 14.6
- 2020 DNC Day 2 - 12.6
- 2020 DNC Day 1 - 12.7
- 2020 RNC Day 4 - 14.7
- 2020 RNC Day 3 - 11.0
- 2020 RNC Day 2 - 12.2
- 2020 RNC Day 1 - 10.7
Highlights from the Democratic National Convention
Harris formally accepted the party's nomination in her acceptance speech that capped off the convention. During her remarks, she shared the story of her working class upbringing, warned about a second Trump presidency, and promised to fight for all Americans.
Rumors erupted over a possible surprise guest Thursday night, but it wasn't Beyoncé or Taylor Swift, as many had hoped. One surprise addition to the convention lineup was Oprah Winfrey , who endorsed Harris in an rousing speech Wednesday.
Kerry Washington and Tony Goldwyn, co-stars in Shonda Rhimes' political drama "Scandal," made a surprise on-stage reunion. Pink, The Chicks, Stevie Wonder and John Legend all performed on stage.
Michelle Obama delivered one of the most quoted speeches of the convention, urging the audience to " do something ."

IMAGES
COMMENTS
To help you get started, here are 100 cosmetology essay topic ideas and examples to inspire your writing: The history and evolution of cosmetology. The role of a cosmetologist in society. The importance of professionalism in the cosmetology industry. The impact of social media on beauty standards. The benefits of pursuing a career in ...
Explore the latest full-text research PDFs, articles, conference papers, preprints and more on COSMETOLOGY. Find methods information, sources, references or conduct a literature review on COSMETOLOGY
Notably, we have conducted research on Nobel Prize-winning topics: Estée Lauder was the first cosmetic brand to research skin cells' circadian rhythm and the role of 'clock genes' n ...
In the context of isopropyl alternatives as sanitizers, ethyl alcohol serves as a solid option. Racial Discrimination Through the Cosmetics Industry. The variety of preconceptions such as the hypersexuality of black women and the perception of their beauty as an unideal version of whites' one also indicates racism.
So understood cosmetology is closely related to the modern vision of health promotion, the part of which is health education. 4,5 The literature review shows that cosmetologists undertake talks with their patients on the topics related to health and illness. 6,7 Moreover beauty salons are indicated by many authors as suitable places for ...
Beauty, Skincare, and Cosmetics Challenges. Cosmetic Surgery History and Effects. The Regulation of Chemicals in Cosmetics. Chemical Peel and Microneedling in Cosmetology. Correcting the Form of Eyebrows: Cosmetic Eyebrow Tattooing. Cosmetic Surgery Popularity in the Middle East.
1 - Sunscreen still needed: Protective surgical face masks ineffective against UV protection, says Kao . People still need to protect their skin from ultraviolet sun rays with sunscreen even when wearing protective surgical face masks, according to new research from Kao.. For the first experiment, researchers from the firm's Beauty Research and Creation Center (BRCC) conducted the ...
The present invention describes a composition and process for the preparation and use of nanocosmetics consisting of lipid nanoparticles formulated with carnauba wax and quercetin incorporated into cosmetic formulations in gel, cream, lotion, or gel-cream forms. 5. KR101578466B1. South Korea.
Subject Guide: Cosmetology. Welcome to the Cosmetology Research Guide. You will find a comprehensive range of resources to support your exploration of cosmetology and related topics. In addition to the scholarly journals and databases in this guide, CSM Librarians can help you throughout your research in-person, via chat, or through one-on-one ...
Dear Colleagues, It is my pleasure to celebrate the 10th anniversary of Cosmetics, which, over these years, has become a reference for many scientists interested in this area of research.Ten years ago, we took the first steps towards the creation of a journal covering all the different interests related to the biological, biochemical, and clinical research in the cosmetic world.
This is a research guide created by College of San Mateo Library librarians to help students with research in the area of cosmetology. It provides tools, resources, and tips for getting started researching this topic.
HBS Cases: Beauty Entrepreneur Madam Walker. by Martha Lagace. She may have been the first self-made African American millionaire. Born of emancipated slaves, Madam C.J. Walker traveled from the cotton fields to business fame as a purveyor of hair-care products that offered beauty and dignity.
T he topics most often discussed between the professionals working in the beauty salons and their clients as well as some examples of educational programs implemented in the beauty salons 4-13
For a list of published articles by FDA scientists on topics related to cosmetics, go to Scientific Publications by FDA Staff and enter "cosmetics" in the search field. FDA Surveys of ...
Cosmetology Research Topics. Cosmetology includes the study and practice of a variety of beauty treatments involving the hair, skin, or nails. Cosmetologists often work as hair stylists, hair colorists, makeup artists, waxing specialists, and nail technicians. To practice as a cosmetologist, a person must complete ...
Step 1: Think about your topic! What questions are you trying to answer? Step 2: List terms that best represent the main concepts of your topic. cosmetology. hair coloring. skin cancer. melanoma. basal cell carcinoma. If nothing comes to mind, it is time to do some background research!
An Online Survey to Estimate the Knowledge, Attitude, and Practice among People in Odisha on Proper Drug Disposal Methods along with their Opinion on the Use of E-pharmacy and Drones as Future Dispensing Models for Medicines. View More. World Journal of Cosmetology and Aesthetic Medicine is an international open access journal the basis of ...
Cosmetics as a Decorative Technique Used by Women. At the beginning of the 20th century, makeup was used to protect and project a sense of self. For me, makeup means the possibility to create and underline a unique identity and the self. Halal Cosmetic Products: Innovation Management.
Exploring cosmetology through essay writing provides an opportunity to engage critically with the evolving trends and practices within the beauty industry. By choosing a compelling topic and conducting thorough research, students can gain valuable insights into the cultural, historical, and ethical aspects of cosmetology.
Cosmetologists help people look their best by providing professional assistance with hair care, nails, facials and makeup. You can become a cosmetologist by completing a state-approved training program and passing a state licensing examination for cosmetology.
View our collection of cosmetology essays. Find inspiration for topics, titles, outlines, & craft impactful cosmetology papers. Read our cosmetology papers today! ... Natural ethical sustainabilityThere are different scholars that are presently conducting research on these particular topics. The following is a list of some of the scholars ...
MIT researchers have discovered how fasting impacts the regenerative abilities of intestinal stem cells, reports Ed Cara for Gizmodo.. "The major finding of our current study is that refeeding after fasting is a distinct state from fasting itself," explain Prof. Ömer Yilmaz and postdocs Shinya Imada and Saleh Khawaled.
Do consumers treat loyalty points the same way that they treat traditional money? And, how do they choose to spend one versus the other? The authors of this article present research findings from ...
For example, a Pew Research Center analysis in 2017 found that an influx of Muslim migrants helped increase the share of Europe's overall population that is Muslim from 4% in 2010 to 5% in 2016 and made larger shifts in some individual countries. Syria was the most common origin for Muslims who migrated to Europe during this period, and most ...
Received: 16-02-2021; Revised: 22-04-2021; Accepted: 27-04-2021; Published on: 15-05-2021. ABSTRACT. Cosmetics are a category of health and beauty products that are used to care for the face and ...
And overall, kids under the age of 12 still saw more than 1,000 food-related advertisements per year, on average. Because of the steep decline in food and beverage ads during children's shows, as ...
Nationally, Vice President Kamala Harris and former President Donald Trump are essentially tied among registered voters in the current snapshot of the presidential race: 46% prefer Harris, 45% prefer Trump and 7% prefer Robert F. Kennedy Jr. Following Biden's exit from the race, Trump's support ...
Amid national debates about what schools are teaching, we asked public K-12 teachers, teens and the American public how they see topics related to race, sexual orientation and gender identity playing out in the classroom.. A sizeable share of teachers (41%) say these debates have had a negative impact on their ability to do their job. Just 4% say these debates have had a positive impact, while ...
It owns several digital publications spanning a variety of topics : news, trending stories, home, cooking, DIY, and more. Pursuant to the San Francisco Fair Chance Ordinance, Collective Press will consider for employment qualified applicants with arrest and conviction records. J-18808-Ljbffr. Last updated : 2024-08-21
Nielsen estimates 26.2 million viewers tuned in to the final night of Democratic National Convention. The four-day event in Chicago drew audiences of at least 20 million every day, according to ...