Have a language expert improve your writing
Run a free plagiarism check in 10 minutes, generate accurate citations for free.
- Knowledge Base
- Methodology
- What Is Participant Observation? | Definition & Examples

What Is Participant Observation? | Definition & Examples
Published on March 10, 2023 by Tegan George .
Participant observation is a research method where the researcher immerses themself in a particular social setting or group, observing the behaviors, interactions, and practices of the participants. This can be a valuable method for any research project that seeks to understand the experiences of individuals or groups in a particular social context.
In participant observation, the researcher is called a participant-observer , meaning that they participate in the group’s activities while also observing the group’s behavior and interactions. There is flexibility in the level of participation, ranging from non-participatory (the weakest) to complete participation (the strongest but most intensive.) The goal here is to gain a deep understanding of the group’s culture, beliefs, and practices from an “insider” perspective.
You immerse yourself in this subculture by spending time at skateparks, attending skateboarding events, and engaging with skateboarders. Perhaps you may even learn to skateboard yourself, in order to better understand the experiences of your study participants.
As you observe, you take notes on the behavior, language, norms, and values you witness and also conduct informal unstructured interviews with individual skateboarders to gain further insight into their thoughts and lived experiences.
Typically used in fields like anthropology, sociology, and other social sciences, this method is often used to gather rich and detailed data about social groups or phenomena through ethnographies or other qualitative research .
Table of contents
When to use participant observation, examples of participant observation, how to analyze data from participant observation, advantages and disadvantages of participant observations, other types of research bias, frequently asked questions.
Participant observation is a type of observational study . Like most observational studies, these are primarily qualitative in nature, used to conduct both explanatory research and exploratory research . Participant observation is also often used in conjunction with other types of research, like interviews and surveys .
This type of study is especially well suited for studying social phenomena that are difficult to observe or measure through other methods. As the researcher observes, they typically take detailed notes about their observations and interactions with the group. These are then analyzed to identify patterns and themes using thematic analysis or a similar method.
A participant observation could be a good fit for your research if:
- You are studying subcultures or groups with unique practices or beliefs. Participant observation fosters a deep and intimate understanding of the beliefs, values, and practices of your group or subculture of interest from an insider’s perspective. This can be especially useful when studying marginalized groups or groups that are resistant to observation.
- You are studying complex social interactions . Participant observation can be a powerful tool for studying the complex social interactions that occur within a particular group or community. By immersing yourself in the group and observing these interactions firsthand, you can gain a much more nuanced understanding of how these interactions flow.
- You are studying behaviors or practices that may be difficult to self-report . In some cases, participants may be unwilling or unable to accurately report their own behaviors or practices. Participant observation allows researchers to observe these behaviors directly, allowing for more accuracy in the data collection phase.
Here's why students love Scribbr's proofreading services
Discover proofreading & editing
Participant observation is a common research method in social sciences, with findings often published in research reports used to inform policymakers or other stakeholders.
Over the course of several months, you observe and take notes on the social interactions, customs, and beliefs of the community members, conducting informal interviews with individual residents to gain further insight into their experiences and perspectives. Through your observations, you gain a deep understanding of the community’s culture, including its values, traditions, and social hierarchy.
Participant observations are often also used in sociology to study social groups and related phenomena, like group formation, stratification, or conflict resolution.
Through this participant observation, you soon see that the group is highly stratified, with certain individuals occupying positions of social power and others being marginalized or even largely excluded. You also observe patterns of conformity within the group, alongside complex interpersonal dynamics.
Data analysis in participant observation typically involves a step-by-step process of immersion, categorization, and interpretation.
- After finishing up your observations, you read through your field notes or transcripts multiple times in the immersion phase. This helps you reflect on what you studied, and is well paired with conducting data cleansing to ensure everything is clear and correct prior to proceeding.
- You then create categories or themes to organize the data. This helps with identifying patterns, behaviors, and interactions relevant to your research question or study aims. In turn, these categories help you to form a coding system that labels or “tags” the aspects of the data that you want to focus on. These can be specific behaviors, emotions, or social interactions—whatever helps you to identify connections between different elements of your data.
- Next, your data can be analyzed using a variety of qualitative research methods, such as thematic analysis , grounded theory, or discourse analysis using the coded categories you created. This helps you interpret the data and develop further theories. You may also want to use triangulation , comparing data from multiple sources or methods, to bolster the reliability and validity of your findings.
- Lastly, it’s always a good research practice to seek feedback on your findings from other researchers in your field of study, as well as members of the group you studied. This helps to ensure the accuracy and reliability of your analysis and can mitigate some potential research biases .
Participant observations are a strong fit for some research projects, but with their advantages come their share of disadvantages as well.
Advantages of participant observations
- Participant observations allow you to generate rich and nuanced qualitative data —particularly useful when seeking to develop a deep understanding of a particular social context or experience. By immersing yourself in the group, you can gain an unrivaled insider perspective on the group’s beliefs, values, and practices.
- Participant observation is a flexible research method that can be adapted to fit a variety of research questions and contexts. Metrics like level of participation in the group, the length of the observation period, and the types of data collected all can be adjusted based on research goals and timeline.
- Participant observation is often used in combination with other research methods, such as interviews or surveys , to provide a more complete picture of the phenomenon being studied. This triangulation can help to improve the reliability and validity of the research findings, as participant observations are not particularly strong as a standalone method.
Disadvantages of participant observations
- Like many observational studies, participant observations are at high risk for many research biases , particularly on the side of the researcher. Because participant observation involves the researcher immersing themselves in the group being studied, there is a risk that their own biases could influence the data they collect, leading to observer bias . Likewise, the presence of a researcher in the group being studied can potentially influence the behavior of the participants. This can lead to inaccurate or biased data if participants alter their behavior in response to the researcher’s presence, leading to a Hawthorne effect or social desirability bias .
- Participant observations can be very expensive, time-consuming, and challenging to carry out. They often require a long period of time to build trust and gather sufficient data, with the data usually collected in an intensive, in-person manner. Some participant observations take generations to complete, which can make it difficult to conduct studies with limited time or resources.
- Participant observation can raise ethical concerns , requiring measured ethical consideration on the part of the researcher with regard to informed consent, privacy, and confidentiality. The researcher must take care to protect the privacy and autonomy of the participants and ensure that they are not placed at undue risk by the research.
Receive feedback on language, structure, and formatting
Professional editors proofread and edit your paper by focusing on:
- Academic style
- Vague sentences
- Style consistency
See an example

- Confirmation bias
- Baader–Meinhof phenomenon
- Availability heuristic
- Halo effect
- Hindsight bias
- Ingroup bias
- Outgroup bias
- Perception bias
- Framing effect
- Self-serving bias
- Affect heuristic
- Representativeness heuristic
- Anchoring heuristic
- Primacy bias
- Optimism bias
- Sampling bias
- Ascertainment bias
- Attrition bias
- Self-selection bias
- Survivorship bias
- Nonresponse bias
- Undercoverage bias
- Hawthorne effect
- Observer bias
- Omitted variable bias
- Publication bias
- Conformity bias
- Pygmalion effect
- Recall bias
- Social desirability bias
- Placebo effect
- Actor-observer bias
- Ceiling effect
- Ecological fallacy
- Affinity bias
Ethical considerations in participant observation involve:
- Obtaining informed consent from all participants
- Protecting their privacy and confidentiality
- Ensuring that they are not placed at undue risk by the research, and
- Respecting their autonomy and agency as participants
Researchers should also consider the potential impact of their research on the community being studied and take steps to minimize any negative after-effects.
Participant observation is a type of qualitative research method . It involves active participation on the part of the researcher in the group being studied, usually over a longer period of time.
Other qualitative research methods, such as interviews or focus groups , do not involve the same level of immersion in the research and can be conducted in a less intense manner.
In participant observation , the researcher plays an active role in the social phenomenon, group, or social context being studied. They may move into the community, attend events or activities, or even take on specific roles within the group— fully joining the community over the course of the study. However, the researcher also maintains an observer role here, taking notes on the behavior and interactions of the participants to draw conclusions and guide further research.
Cite this Scribbr article
If you want to cite this source, you can copy and paste the citation or click the “Cite this Scribbr article” button to automatically add the citation to our free Citation Generator.
George, T. (2023, March 10). What Is Participant Observation? | Definition & Examples. Scribbr. Retrieved June 18, 2024, from https://www.scribbr.com/methodology/participant-observation/
Is this article helpful?
Tegan George
Other students also liked, ethical considerations in research | types & examples, what is qualitative research | methods & examples, what is an observational study | guide & examples, get unlimited documents corrected.
✔ Free APA citation check included ✔ Unlimited document corrections ✔ Specialized in correcting academic texts
Participant Selection and Access in Case Study Research
- First Online: 05 January 2019
Cite this chapter

- Zhongyan Wan 4
1598 Accesses
2 Citations
Case study research requires researchers to purposefully select information-rich cases, as they will allow researchers an in-depth understanding of relevant and critical issues under investigation (Patton in Qualitative evaluation and research methods. Sage, Newbury Park, 1990 , Qual Soc Work 1(3):261–283, 2002 ). To gain such insights, purposive sampling is generally believed to contribute to the richness in the range of data collected and help increase the possibilities of uncovering multiple realities (Guba and Lincoln in Handb Qual Res 2(163–194):105, 1994 ). However, dealing with research participants in the case study approach also poses great challenges for novice researchers, as humans can be a difficult factor to control among the variables in research.
This is a preview of subscription content, log in via an institution to check access.
Access this chapter
- Available as PDF
- Read on any device
- Instant download
- Own it forever
- Available as EPUB and PDF
- Compact, lightweight edition
- Dispatched in 3 to 5 business days
- Free shipping worldwide - see info
Tax calculation will be finalised at checkout
Purchases are for personal use only
Institutional subscriptions
Similar content being viewed by others

Questionable Research Practices in Single-Case Experimental Designs: Examples and Possible Solutions

Case Study Research

Alinejad, D. (2011). Mapping homelands through virtual spaces: Transnational embodiment and Iranian diaspora bloggers. Global Networks, 11 (1), 43–62.
Google Scholar
Arcury, T., & Quandt, S. (1999). Participant recruitment for qualitative research: A site-based approach to community research in complex societies. Human Organization, 58 (2), 128–133.
Article Google Scholar
Barbour, R. (2014). Introducing qualitative research: A student’s guide . London: Sage.
Decrop, A. (1999). Qualitative research methods for the study of tourist behaviour. In A. Pizam & Y. Mansfeld. (Eds.), Consumer Behaviour in Travel and Tourism . New York: The Haworth Hospitality Press
Duff, P. (2008). Case study research in applied linguistics . New York: Taylor & Francis.
Feldman, M. S., Bell, J., & Berger, M. T. (2003). Gaining access: A practical and theoretical guide for qualitative researchers . Walnut Creek, CA: Altamira Press.
Gall, M. D., Gall, J. P., & Borg, W. T. (2003). Educational research (7th ed.). White Plains, NY: Pearson Education.
Gerring, J. (2004). What is a case study and what is it good for? American Political Science Review, 98 (02), 341–354.
Guba, E. G., & Lincoln, Y. S. (1994). Competing paradigms in qualitative research. Handbook of Qualitative Research, 2 (163–194), 105.
Gummesson, E. (2000). Qualitative methods in management research . Thousand Oaks, CA: Sage.
Hamilton, R. J., & Bowers, B. J. (2006). Internet recruitment and e-mail interviews in qualitative studies. Qualitative Health Research, 16 (6), 821–835.
Hancock, D. R., & Algozzine, B. (2015). Doing case study research: A practical guide for beginning researchers . New York: Teachers College Press.
Hewson, C. (2014). Qualitative approaches in Internet-mediated research: Opportunities, issues, possibilities. The Oxford Handbook of Qualitative Research , 423–452.
Johl, S. K., & Renganathan, S. (2010). Strategies for gaining access in doing fieldwork: Reflection of two researchers. The Electronic Journal of Business Research Methods, 8 (1), 42–50.
Kothari, C. R. (2004). Research methodology: Methods and techniques . India: New Age International.
Lee, R. (1993). Doing research on sensitive topics . London: Sage.
Merriam, S. (2009). Case study research in education: A qualitative approach . San Francisco: Jossey-Bass.
Mikecz, R. (2012). Interviewing elites addressing methodological issues. Qualitative Inquiry, 18 (6), 482–493.
Miles, M. B., & Huberman, A. M. (1994). Qualitative data analysis: An expanded sourcebook . Thousand Oaks, CA: Sage.
Noy, C. (2008). Sampling knowledge: The hermeneutics of snowball sampling in qualitative research. International Journal of Social Research Methodology, 11 (4), 327–344.
Okumus, F., Altinay, L., & Roper, A. (2007). Gaining access for research: Reflections from experience. Annals of Tourism Research, 34 (1), 7–26.
Patton, M. Q. (1990). Qualitative evaluation and research methods (2nd ed.). Newbury Park, CA: Sage.
Patton, M. Q. (2002). Two decades of developments in qualitative inquiry a personal, experiential perspective. Qualitative Social Work, 1 (3), 261–283.
Punch, K. F. (1998). Introduction to social research . London: Sage.
Seawright, J., & Gerring, J. (2008). Case selection techniques in case study research a menu of qualitative and quantitative options. Political Research Quarterly, 61 (2), 294–308.
Silverman, D. (Ed.). (2016). Qualitative research . London: Sage.
Thomas, G. (2015). How to do your case study . Thousand Oaks, CA: Sage.
Wan, Z. Y. (2016). College teacher beliefs and practices in computer-assisted language learning classrooms in China . Shanghai: Shanghai Jiaotong University Press.
Willis, J. W., Jost, M., & Nilakanta, R. (2007). Foundations of qualitative research: Interpretive and critical approaches . London: Sage.
Book Google Scholar
Wilmot, A. (2005). Designing sampling strategies for qualitative social research: With particular reference to the Office for National Statistics’ Qualitative Respondent Register. Survey Methodology Bulletin-office for National Statistics, 56, 53.
Yin, R. K. (2009). Case study research: Design and methods (4th ed.). Thousand Oaks, CA: Sage.
Yin, R. K. (2013). Case study research: Design and methods (5th ed.). London: Sage publications.
Download references
Author information
Authors and affiliations.
Foreign Language Department, China Pharmaceutical University, Nanjing, China
Zhongyan Wan
You can also search for this author in PubMed Google Scholar
Corresponding author
Correspondence to Zhongyan Wan .
Editor information
Editors and affiliations.
Faculty of Education, Beijing Normal University, Beijing, China
Kwok Kuen Tsang
Faculty of Social Sciences, University of Stavanger, Stavanger, Norway
School of Humanities, Southeast University, Nanjing, Jiangsu, China
Rights and permissions
Reprints and permissions
Copyright information
© 2019 Springer Nature Singapore Pte Ltd.
About this chapter
Wan, Z. (2019). Participant Selection and Access in Case Study Research. In: Tsang, K., Liu, D., Hong, Y. (eds) Challenges and Opportunities in Qualitative Research. Springer, Singapore. https://doi.org/10.1007/978-981-13-5811-1_5
Download citation
DOI : https://doi.org/10.1007/978-981-13-5811-1_5
Published : 05 January 2019
Publisher Name : Springer, Singapore
Print ISBN : 978-981-13-5810-4
Online ISBN : 978-981-13-5811-1
eBook Packages : Social Sciences Social Sciences (R0)
Share this chapter
Anyone you share the following link with will be able to read this content:
Sorry, a shareable link is not currently available for this article.
Provided by the Springer Nature SharedIt content-sharing initiative
- Publish with us
Policies and ethics
- Find a journal
- Track your research
- Privacy Policy

Home » Case Study – Methods, Examples and Guide
Case Study – Methods, Examples and Guide
Table of Contents

A case study is a research method that involves an in-depth examination and analysis of a particular phenomenon or case, such as an individual, organization, community, event, or situation.
It is a qualitative research approach that aims to provide a detailed and comprehensive understanding of the case being studied. Case studies typically involve multiple sources of data, including interviews, observations, documents, and artifacts, which are analyzed using various techniques, such as content analysis, thematic analysis, and grounded theory. The findings of a case study are often used to develop theories, inform policy or practice, or generate new research questions.
Types of Case Study
Types and Methods of Case Study are as follows:
Single-Case Study
A single-case study is an in-depth analysis of a single case. This type of case study is useful when the researcher wants to understand a specific phenomenon in detail.
For Example , A researcher might conduct a single-case study on a particular individual to understand their experiences with a particular health condition or a specific organization to explore their management practices. The researcher collects data from multiple sources, such as interviews, observations, and documents, and uses various techniques to analyze the data, such as content analysis or thematic analysis. The findings of a single-case study are often used to generate new research questions, develop theories, or inform policy or practice.
Multiple-Case Study
A multiple-case study involves the analysis of several cases that are similar in nature. This type of case study is useful when the researcher wants to identify similarities and differences between the cases.
For Example, a researcher might conduct a multiple-case study on several companies to explore the factors that contribute to their success or failure. The researcher collects data from each case, compares and contrasts the findings, and uses various techniques to analyze the data, such as comparative analysis or pattern-matching. The findings of a multiple-case study can be used to develop theories, inform policy or practice, or generate new research questions.
Exploratory Case Study
An exploratory case study is used to explore a new or understudied phenomenon. This type of case study is useful when the researcher wants to generate hypotheses or theories about the phenomenon.
For Example, a researcher might conduct an exploratory case study on a new technology to understand its potential impact on society. The researcher collects data from multiple sources, such as interviews, observations, and documents, and uses various techniques to analyze the data, such as grounded theory or content analysis. The findings of an exploratory case study can be used to generate new research questions, develop theories, or inform policy or practice.
Descriptive Case Study
A descriptive case study is used to describe a particular phenomenon in detail. This type of case study is useful when the researcher wants to provide a comprehensive account of the phenomenon.
For Example, a researcher might conduct a descriptive case study on a particular community to understand its social and economic characteristics. The researcher collects data from multiple sources, such as interviews, observations, and documents, and uses various techniques to analyze the data, such as content analysis or thematic analysis. The findings of a descriptive case study can be used to inform policy or practice or generate new research questions.
Instrumental Case Study
An instrumental case study is used to understand a particular phenomenon that is instrumental in achieving a particular goal. This type of case study is useful when the researcher wants to understand the role of the phenomenon in achieving the goal.
For Example, a researcher might conduct an instrumental case study on a particular policy to understand its impact on achieving a particular goal, such as reducing poverty. The researcher collects data from multiple sources, such as interviews, observations, and documents, and uses various techniques to analyze the data, such as content analysis or thematic analysis. The findings of an instrumental case study can be used to inform policy or practice or generate new research questions.
Case Study Data Collection Methods
Here are some common data collection methods for case studies:
Interviews involve asking questions to individuals who have knowledge or experience relevant to the case study. Interviews can be structured (where the same questions are asked to all participants) or unstructured (where the interviewer follows up on the responses with further questions). Interviews can be conducted in person, over the phone, or through video conferencing.
Observations
Observations involve watching and recording the behavior and activities of individuals or groups relevant to the case study. Observations can be participant (where the researcher actively participates in the activities) or non-participant (where the researcher observes from a distance). Observations can be recorded using notes, audio or video recordings, or photographs.
Documents can be used as a source of information for case studies. Documents can include reports, memos, emails, letters, and other written materials related to the case study. Documents can be collected from the case study participants or from public sources.
Surveys involve asking a set of questions to a sample of individuals relevant to the case study. Surveys can be administered in person, over the phone, through mail or email, or online. Surveys can be used to gather information on attitudes, opinions, or behaviors related to the case study.
Artifacts are physical objects relevant to the case study. Artifacts can include tools, equipment, products, or other objects that provide insights into the case study phenomenon.
How to conduct Case Study Research
Conducting a case study research involves several steps that need to be followed to ensure the quality and rigor of the study. Here are the steps to conduct case study research:
- Define the research questions: The first step in conducting a case study research is to define the research questions. The research questions should be specific, measurable, and relevant to the case study phenomenon under investigation.
- Select the case: The next step is to select the case or cases to be studied. The case should be relevant to the research questions and should provide rich and diverse data that can be used to answer the research questions.
- Collect data: Data can be collected using various methods, such as interviews, observations, documents, surveys, and artifacts. The data collection method should be selected based on the research questions and the nature of the case study phenomenon.
- Analyze the data: The data collected from the case study should be analyzed using various techniques, such as content analysis, thematic analysis, or grounded theory. The analysis should be guided by the research questions and should aim to provide insights and conclusions relevant to the research questions.
- Draw conclusions: The conclusions drawn from the case study should be based on the data analysis and should be relevant to the research questions. The conclusions should be supported by evidence and should be clearly stated.
- Validate the findings: The findings of the case study should be validated by reviewing the data and the analysis with participants or other experts in the field. This helps to ensure the validity and reliability of the findings.
- Write the report: The final step is to write the report of the case study research. The report should provide a clear description of the case study phenomenon, the research questions, the data collection methods, the data analysis, the findings, and the conclusions. The report should be written in a clear and concise manner and should follow the guidelines for academic writing.
Examples of Case Study
Here are some examples of case study research:
- The Hawthorne Studies : Conducted between 1924 and 1932, the Hawthorne Studies were a series of case studies conducted by Elton Mayo and his colleagues to examine the impact of work environment on employee productivity. The studies were conducted at the Hawthorne Works plant of the Western Electric Company in Chicago and included interviews, observations, and experiments.
- The Stanford Prison Experiment: Conducted in 1971, the Stanford Prison Experiment was a case study conducted by Philip Zimbardo to examine the psychological effects of power and authority. The study involved simulating a prison environment and assigning participants to the role of guards or prisoners. The study was controversial due to the ethical issues it raised.
- The Challenger Disaster: The Challenger Disaster was a case study conducted to examine the causes of the Space Shuttle Challenger explosion in 1986. The study included interviews, observations, and analysis of data to identify the technical, organizational, and cultural factors that contributed to the disaster.
- The Enron Scandal: The Enron Scandal was a case study conducted to examine the causes of the Enron Corporation’s bankruptcy in 2001. The study included interviews, analysis of financial data, and review of documents to identify the accounting practices, corporate culture, and ethical issues that led to the company’s downfall.
- The Fukushima Nuclear Disaster : The Fukushima Nuclear Disaster was a case study conducted to examine the causes of the nuclear accident that occurred at the Fukushima Daiichi Nuclear Power Plant in Japan in 2011. The study included interviews, analysis of data, and review of documents to identify the technical, organizational, and cultural factors that contributed to the disaster.
Application of Case Study
Case studies have a wide range of applications across various fields and industries. Here are some examples:
Business and Management
Case studies are widely used in business and management to examine real-life situations and develop problem-solving skills. Case studies can help students and professionals to develop a deep understanding of business concepts, theories, and best practices.
Case studies are used in healthcare to examine patient care, treatment options, and outcomes. Case studies can help healthcare professionals to develop critical thinking skills, diagnose complex medical conditions, and develop effective treatment plans.
Case studies are used in education to examine teaching and learning practices. Case studies can help educators to develop effective teaching strategies, evaluate student progress, and identify areas for improvement.
Social Sciences
Case studies are widely used in social sciences to examine human behavior, social phenomena, and cultural practices. Case studies can help researchers to develop theories, test hypotheses, and gain insights into complex social issues.
Law and Ethics
Case studies are used in law and ethics to examine legal and ethical dilemmas. Case studies can help lawyers, policymakers, and ethical professionals to develop critical thinking skills, analyze complex cases, and make informed decisions.
Purpose of Case Study
The purpose of a case study is to provide a detailed analysis of a specific phenomenon, issue, or problem in its real-life context. A case study is a qualitative research method that involves the in-depth exploration and analysis of a particular case, which can be an individual, group, organization, event, or community.
The primary purpose of a case study is to generate a comprehensive and nuanced understanding of the case, including its history, context, and dynamics. Case studies can help researchers to identify and examine the underlying factors, processes, and mechanisms that contribute to the case and its outcomes. This can help to develop a more accurate and detailed understanding of the case, which can inform future research, practice, or policy.
Case studies can also serve other purposes, including:
- Illustrating a theory or concept: Case studies can be used to illustrate and explain theoretical concepts and frameworks, providing concrete examples of how they can be applied in real-life situations.
- Developing hypotheses: Case studies can help to generate hypotheses about the causal relationships between different factors and outcomes, which can be tested through further research.
- Providing insight into complex issues: Case studies can provide insights into complex and multifaceted issues, which may be difficult to understand through other research methods.
- Informing practice or policy: Case studies can be used to inform practice or policy by identifying best practices, lessons learned, or areas for improvement.
Advantages of Case Study Research
There are several advantages of case study research, including:
- In-depth exploration: Case study research allows for a detailed exploration and analysis of a specific phenomenon, issue, or problem in its real-life context. This can provide a comprehensive understanding of the case and its dynamics, which may not be possible through other research methods.
- Rich data: Case study research can generate rich and detailed data, including qualitative data such as interviews, observations, and documents. This can provide a nuanced understanding of the case and its complexity.
- Holistic perspective: Case study research allows for a holistic perspective of the case, taking into account the various factors, processes, and mechanisms that contribute to the case and its outcomes. This can help to develop a more accurate and comprehensive understanding of the case.
- Theory development: Case study research can help to develop and refine theories and concepts by providing empirical evidence and concrete examples of how they can be applied in real-life situations.
- Practical application: Case study research can inform practice or policy by identifying best practices, lessons learned, or areas for improvement.
- Contextualization: Case study research takes into account the specific context in which the case is situated, which can help to understand how the case is influenced by the social, cultural, and historical factors of its environment.
Limitations of Case Study Research
There are several limitations of case study research, including:
- Limited generalizability : Case studies are typically focused on a single case or a small number of cases, which limits the generalizability of the findings. The unique characteristics of the case may not be applicable to other contexts or populations, which may limit the external validity of the research.
- Biased sampling: Case studies may rely on purposive or convenience sampling, which can introduce bias into the sample selection process. This may limit the representativeness of the sample and the generalizability of the findings.
- Subjectivity: Case studies rely on the interpretation of the researcher, which can introduce subjectivity into the analysis. The researcher’s own biases, assumptions, and perspectives may influence the findings, which may limit the objectivity of the research.
- Limited control: Case studies are typically conducted in naturalistic settings, which limits the control that the researcher has over the environment and the variables being studied. This may limit the ability to establish causal relationships between variables.
- Time-consuming: Case studies can be time-consuming to conduct, as they typically involve a detailed exploration and analysis of a specific case. This may limit the feasibility of conducting multiple case studies or conducting case studies in a timely manner.
- Resource-intensive: Case studies may require significant resources, including time, funding, and expertise. This may limit the ability of researchers to conduct case studies in resource-constrained settings.
About the author
Muhammad Hassan
Researcher, Academic Writer, Web developer
You may also like

Survey Research – Types, Methods, Examples

Mixed Methods Research – Types & Analysis

Transformative Design – Methods, Types, Guide

Qualitative Research – Methods, Analysis Types...

Exploratory Research – Types, Methods and...

Observational Research – Methods and Guide
Case Studies
This guide examines case studies, a form of qualitative descriptive research that is used to look at individuals, a small group of participants, or a group as a whole. Researchers collect data about participants using participant and direct observations, interviews, protocols, tests, examinations of records, and collections of writing samples. Starting with a definition of the case study, the guide moves to a brief history of this research method. Using several well documented case studies, the guide then looks at applications and methods including data collection and analysis. A discussion of ways to handle validity, reliability, and generalizability follows, with special attention to case studies as they are applied to composition studies. Finally, this guide examines the strengths and weaknesses of case studies.
Definition and Overview
Case study refers to the collection and presentation of detailed information about a particular participant or small group, frequently including the accounts of subjects themselves. A form of qualitative descriptive research, the case study looks intensely at an individual or small participant pool, drawing conclusions only about that participant or group and only in that specific context. Researchers do not focus on the discovery of a universal, generalizable truth, nor do they typically look for cause-effect relationships; instead, emphasis is placed on exploration and description.
Case studies typically examine the interplay of all variables in order to provide as complete an understanding of an event or situation as possible. This type of comprehensive understanding is arrived at through a process known as thick description, which involves an in-depth description of the entity being evaluated, the circumstances under which it is used, the characteristics of the people involved in it, and the nature of the community in which it is located. Thick description also involves interpreting the meaning of demographic and descriptive data such as cultural norms and mores, community values, ingrained attitudes, and motives.
Unlike quantitative methods of research, like the survey, which focus on the questions of who, what, where, how much, and how many, and archival analysis, which often situates the participant in some form of historical context, case studies are the preferred strategy when how or why questions are asked. Likewise, they are the preferred method when the researcher has little control over the events, and when there is a contemporary focus within a real life context. In addition, unlike more specifically directed experiments, case studies require a problem that seeks a holistic understanding of the event or situation in question using inductive logic--reasoning from specific to more general terms.
In scholarly circles, case studies are frequently discussed within the context of qualitative research and naturalistic inquiry. Case studies are often referred to interchangeably with ethnography, field study, and participant observation. The underlying philosophical assumptions in the case are similar to these types of qualitative research because each takes place in a natural setting (such as a classroom, neighborhood, or private home), and strives for a more holistic interpretation of the event or situation under study.
Unlike more statistically-based studies which search for quantifiable data, the goal of a case study is to offer new variables and questions for further research. F.H. Giddings, a sociologist in the early part of the century, compares statistical methods to the case study on the basis that the former are concerned with the distribution of a particular trait, or a small number of traits, in a population, whereas the case study is concerned with the whole variety of traits to be found in a particular instance" (Hammersley 95).
Case studies are not a new form of research; naturalistic inquiry was the primary research tool until the development of the scientific method. The fields of sociology and anthropology are credited with the primary shaping of the concept as we know it today. However, case study research has drawn from a number of other areas as well: the clinical methods of doctors; the casework technique being developed by social workers; the methods of historians and anthropologists, plus the qualitative descriptions provided by quantitative researchers like LePlay; and, in the case of Robert Park, the techniques of newspaper reporters and novelists.
Park was an ex-newspaper reporter and editor who became very influential in developing sociological case studies at the University of Chicago in the 1920s. As a newspaper professional he coined the term "scientific" or "depth" reporting: the description of local events in a way that pointed to major social trends. Park viewed the sociologist as "merely a more accurate, responsible, and scientific reporter." Park stressed the variety and value of human experience. He believed that sociology sought to arrive at natural, but fluid, laws and generalizations in regard to human nature and society. These laws weren't static laws of the kind sought by many positivists and natural law theorists, but rather, they were laws of becoming--with a constant possibility of change. Park encouraged students to get out of the library, to quit looking at papers and books, and to view the constant experiment of human experience. He writes, "Go and sit in the lounges of the luxury hotels and on the doorsteps of the flophouses; sit on the Gold Coast settees and on the slum shakedowns; sit in the Orchestra Hall and in the Star and Garter Burlesque. In short, gentlemen [sic], go get the seats of your pants dirty in real research."
But over the years, case studies have drawn their share of criticism. In fact, the method had its detractors from the start. In the 1920s, the debate between pro-qualitative and pro-quantitative became quite heated. Case studies, when compared to statistics, were considered by many to be unscientific. From the 1930's on, the rise of positivism had a growing influence on quantitative methods in sociology. People wanted static, generalizable laws in science. The sociological positivists were looking for stable laws of social phenomena. They criticized case study research because it failed to provide evidence of inter subjective agreement. Also, they condemned it because of the few number of cases studied and that the under-standardized character of their descriptions made generalization impossible. By the 1950s, quantitative methods, in the form of survey research, had become the dominant sociological approach and case study had become a minority practice.
Educational Applications
The 1950's marked the dawning of a new era in case study research, namely that of the utilization of the case study as a teaching method. "Instituted at Harvard Business School in the 1950s as a primary method of teaching, cases have since been used in classrooms and lecture halls alike, either as part of a course of study or as the main focus of the course to which other teaching material is added" (Armisted 1984). The basic purpose of instituting the case method as a teaching strategy was "to transfer much of the responsibility for learning from the teacher on to the student, whose role, as a result, shifts away from passive absorption toward active construction" (Boehrer 1990). Through careful examination and discussion of various cases, "students learn to identify actual problems, to recognize key players and their agendas, and to become aware of those aspects of the situation that contribute to the problem" (Merseth 1991). In addition, students are encouraged to "generate their own analysis of the problems under consideration, to develop their own solutions, and to practically apply their own knowledge of theory to these problems" (Boyce 1993). Along the way, students also develop "the power to analyze and to master a tangled circumstance by identifying and delineating important factors; the ability to utilize ideas, to test them against facts, and to throw them into fresh combinations" (Merseth 1991).
In addition to the practical application and testing of scholarly knowledge, case discussions can also help students prepare for real-world problems, situations and crises by providing an approximation of various professional environments (i.e. classroom, board room, courtroom, or hospital). Thus, through the examination of specific cases, students are given the opportunity to work out their own professional issues through the trials, tribulations, experiences, and research findings of others. An obvious advantage to this mode of instruction is that it allows students the exposure to settings and contexts that they might not otherwise experience. For example, a student interested in studying the effects of poverty on minority secondary student's grade point averages and S.A.T. scores could access and analyze information from schools as geographically diverse as Los Angeles, New York City, Miami, and New Mexico without ever having to leave the classroom.
The case study method also incorporates the idea that students can learn from one another "by engaging with each other and with each other's ideas, by asserting something and then having it questioned, challenged and thrown back at them so that they can reflect on what they hear, and then refine what they say" (Boehrer 1990). In summary, students can direct their own learning by formulating questions and taking responsibility for the study.
Types and Design Concerns
Researchers use multiple methods and approaches to conduct case studies.
Types of Case Studies
Under the more generalized category of case study exist several subdivisions, each of which is custom selected for use depending upon the goals and/or objectives of the investigator. These types of case study include the following:
Illustrative Case Studies These are primarily descriptive studies. They typically utilize one or two instances of an event to show what a situation is like. Illustrative case studies serve primarily to make the unfamiliar familiar and to give readers a common language about the topic in question.
Exploratory (or pilot) Case Studies These are condensed case studies performed before implementing a large scale investigation. Their basic function is to help identify questions and select types of measurement prior to the main investigation. The primary pitfall of this type of study is that initial findings may seem convincing enough to be released prematurely as conclusions.
Cumulative Case Studies These serve to aggregate information from several sites collected at different times. The idea behind these studies is the collection of past studies will allow for greater generalization without additional cost or time being expended on new, possibly repetitive studies.
Critical Instance Case Studies These examine one or more sites for either the purpose of examining a situation of unique interest with little to no interest in generalizability, or to call into question or challenge a highly generalized or universal assertion. This method is useful for answering cause and effect questions.
Identifying a Theoretical Perspective
Much of the case study's design is inherently determined for researchers, depending on the field from which they are working. In composition studies, researchers are typically working from a qualitative, descriptive standpoint. In contrast, physicists will approach their research from a more quantitative perspective. Still, in designing the study, researchers need to make explicit the questions to be explored and the theoretical perspective from which they will approach the case. The three most commonly adopted theories are listed below:
Individual Theories These focus primarily on the individual development, cognitive behavior, personality, learning and disability, and interpersonal interactions of a particular subject.
Organizational Theories These focus on bureaucracies, institutions, organizational structure and functions, or excellence in organizational performance.
Social Theories These focus on urban development, group behavior, cultural institutions, or marketplace functions.
Two examples of case studies are used consistently throughout this chapter. The first, a study produced by Berkenkotter, Huckin, and Ackerman (1988), looks at a first year graduate student's initiation into an academic writing program. The study uses participant-observer and linguistic data collecting techniques to assess the student's knowledge of appropriate discourse conventions. Using the pseudonym Nate to refer to the subject, the study sought to illuminate the particular experience rather than to generalize about the experience of fledgling academic writers collectively.
For example, in Berkenkotter, Huckin, and Ackerman's (1988) study we are told that the researchers are interested in disciplinary communities. In the first paragraph, they ask what constitutes membership in a disciplinary community and how achieving membership might affect a writer's understanding and production of texts. In the third paragraph they state that researchers must negotiate their claims "within the context of his sub specialty's accepted knowledge and methodology." In the next paragraph they ask, "How is literacy acquired? What is the process through which novices gain community membership? And what factors either aid or hinder students learning the requisite linguistic behaviors?" This introductory section ends with a paragraph in which the study's authors claim that during the course of the study, the subject, Nate, successfully makes the transition from "skilled novice" to become an initiated member of the academic discourse community and that his texts exhibit linguistic changes which indicate this transition. In the next section the authors make explicit the sociolinguistic theoretical and methodological assumptions on which the study is based (1988). Thus the reader has a good understanding of the authors' theoretical background and purpose in conducting the study even before it is explicitly stated on the fourth page of the study. "Our purpose was to examine the effects of the educational context on one graduate student's production of texts as he wrote in different courses and for different faculty members over the academic year 1984-85." The goal of the study then, was to explore the idea that writers must be initiated into a writing community, and that this initiation will change the way one writes.
The second example is Janet Emig's (1971) study of the composing process of a group of twelfth graders. In this study, Emig seeks to answer the question of what happens to the self as a result educational stimuli in terms of academic writing. The case study used methods such as protocol analysis, tape-recorded interviews, and discourse analysis.
In the case of Janet Emig's (1971) study of the composing process of eight twelfth graders, four specific hypotheses were made:
- Twelfth grade writers engage in two modes of composing: reflexive and extensive.
- These differences can be ascertained and characterized through having the writers compose aloud their composition process.
- A set of implied stylistic principles governs the writing process.
- For twelfth grade writers, extensive writing occurs chiefly as a school-sponsored activity, or reflexive, as a self-sponsored activity.
In this study, the chief distinction is between the two dominant modes of composing among older, secondary school students. The distinctions are:
- The reflexive mode, which focuses on the writer's thoughts and feelings.
- The extensive mode, which focuses on conveying a message.
Emig also outlines the specific questions which guided the research in the opening pages of her Review of Literature , preceding the report.
Designing a Case Study
After considering the different sub categories of case study and identifying a theoretical perspective, researchers can begin to design their study. Research design is the string of logic that ultimately links the data to be collected and the conclusions to be drawn to the initial questions of the study. Typically, research designs deal with at least four problems:
- What questions to study
- What data are relevant
- What data to collect
- How to analyze that data
In other words, a research design is basically a blueprint for getting from the beginning to the end of a study. The beginning is an initial set of questions to be answered, and the end is some set of conclusions about those questions.
Because case studies are conducted on topics as diverse as Anglo-Saxon Literature (Thrane 1986) and AIDS prevention (Van Vugt 1994), it is virtually impossible to outline any strict or universal method or design for conducting the case study. However, Robert K. Yin (1993) does offer five basic components of a research design:
- A study's questions.
- A study's propositions (if any).
- A study's units of analysis.
- The logic that links the data to the propositions.
- The criteria for interpreting the findings.
In addition to these five basic components, Yin also stresses the importance of clearly articulating one's theoretical perspective, determining the goals of the study, selecting one's subject(s), selecting the appropriate method(s) of collecting data, and providing some considerations to the composition of the final report.
Conducting Case Studies
To obtain as complete a picture of the participant as possible, case study researchers can employ a variety of approaches and methods. These approaches, methods, and related issues are discussed in depth in this section.
Method: Single or Multi-modal?
To obtain as complete a picture of the participant as possible, case study researchers can employ a variety of methods. Some common methods include interviews , protocol analyses, field studies, and participant-observations. Emig (1971) chose to use several methods of data collection. Her sources included conversations with the students, protocol analysis, discrete observations of actual composition, writing samples from each student, and school records (Lauer and Asher 1988).
Berkenkotter, Huckin, and Ackerman (1988) collected data by observing classrooms, conducting faculty and student interviews, collecting self reports from the subject, and by looking at the subject's written work.
A study that was criticized for using a single method model was done by Flower and Hayes (1984). In this study that explores the ways in which writers use different forms of knowing to create space, the authors used only protocol analysis to gather data. The study came under heavy fire because of their decision to use only one method.
Participant Selection
Case studies can use one participant, or a small group of participants. However, it is important that the participant pool remain relatively small. The participants can represent a diverse cross section of society, but this isn't necessary.
For example, the Berkenkotter, Huckin, and Ackerman (1988) study looked at just one participant, Nate. By contrast, in Janet Emig's (1971) study of the composition process of twelfth graders, eight participants were selected representing a diverse cross section of the community, with volunteers from an all-white upper-middle-class suburban school, an all-black inner-city school, a racially mixed lower-middle-class school, an economically and racially mixed school, and a university school.
Often, a brief "case history" is done on the participants of the study in order to provide researchers with a clearer understanding of their participants, as well as some insight as to how their own personal histories might affect the outcome of the study. For instance, in Emig's study, the investigator had access to the school records of five of the participants, and to standardized test scores for the remaining three. Also made available to the researcher was the information that three of the eight students were selected as NCTE Achievement Award winners. These personal histories can be useful in later stages of the study when data are being analyzed and conclusions drawn.
Data Collection
There are six types of data collected in case studies:
- Archival records.
- Interviews.
- Direct observation.
- Participant observation.
In the field of composition research, these six sources might be:
- A writer's drafts.
- School records of student writers.
- Transcripts of interviews with a writer.
- Transcripts of conversations between writers (and protocols).
- Videotapes and notes from direct field observations.
- Hard copies of a writer's work on computer.
Depending on whether researchers have chosen to use a single or multi-modal approach for the case study, they may choose to collect data from one or any combination of these sources.
Protocols, that is, transcriptions of participants talking aloud about what they are doing as they do it, have been particularly common in composition case studies. For example, in Emig's (1971) study, the students were asked, in four different sessions, to give oral autobiographies of their writing experiences and to compose aloud three themes in the presence of a tape recorder and the investigator.
In some studies, only one method of data collection is conducted. For example, the Flower and Hayes (1981) report on the cognitive process theory of writing depends on protocol analysis alone. However, using multiple sources of evidence to increase the reliability and validity of the data can be advantageous.
Case studies are likely to be much more convincing and accurate if they are based on several different sources of information, following a corroborating mode. This conclusion is echoed among many composition researchers. For example, in her study of predrafting processes of high and low-apprehensive writers, Cynthia Selfe (1985) argues that because "methods of indirect observation provide only an incomplete reflection of the complex set of processes involved in composing, a combination of several such methods should be used to gather data in any one study." Thus, in this study, Selfe collected her data from protocols, observations of students role playing their writing processes, audio taped interviews with the students, and videotaped observations of the students in the process of composing.
It can be said then, that cross checking data from multiple sources can help provide a multidimensional profile of composing activities in a particular setting. Sharan Merriam (1985) suggests "checking, verifying, testing, probing, and confirming collected data as you go, arguing that this process will follow in a funnel-like design resulting in less data gathering in later phases of the study along with a congruent increase in analysis checking, verifying, and confirming."
It is important to note that in case studies, as in any qualitative descriptive research, while researchers begin their studies with one or several questions driving the inquiry (which influence the key factors the researcher will be looking for during data collection), a researcher may find new key factors emerging during data collection. These might be unexpected patterns or linguistic features which become evident only during the course of the research. While not bearing directly on the researcher's guiding questions, these variables may become the basis for new questions asked at the end of the report, thus linking to the possibility of further research.
Data Analysis
As the information is collected, researchers strive to make sense of their data. Generally, researchers interpret their data in one of two ways: holistically or through coding. Holistic analysis does not attempt to break the evidence into parts, but rather to draw conclusions based on the text as a whole. Flower and Hayes (1981), for example, make inferences from entire sections of their students' protocols, rather than searching through the transcripts to look for isolatable characteristics.
However, composition researchers commonly interpret their data by coding, that is by systematically searching data to identify and/or categorize specific observable actions or characteristics. These observable actions then become the key variables in the study. Sharan Merriam (1988) suggests seven analytic frameworks for the organization and presentation of data:
- The role of participants.
- The network analysis of formal and informal exchanges among groups.
- Historical.
- Thematical.
- Ritual and symbolism.
- Critical incidents that challenge or reinforce fundamental beliefs, practices, and values.
There are two purposes of these frameworks: to look for patterns among the data and to look for patterns that give meaning to the case study.
As stated above, while most researchers begin their case studies expecting to look for particular observable characteristics, it is not unusual for key variables to emerge during data collection. Typical variables coded in case studies of writers include pauses writers make in the production of a text, the use of specific linguistic units (such as nouns or verbs), and writing processes (planning, drafting, revising, and editing). In the Berkenkotter, Huckin, and Ackerman (1988) study, for example, researchers coded the participant's texts for use of connectives, discourse demonstratives, average sentence length, off-register words, use of the first person pronoun, and the ratio of definite articles to indefinite articles.
Since coding is inherently subjective, more than one coder is usually employed. In the Berkenkotter, Huckin, and Ackerman (1988) study, for example, three rhetoricians were employed to code the participant's texts for off-register phrases. The researchers established the agreement among the coders before concluding that the participant used fewer off-register words as the graduate program progressed.
Composing the Case Study Report
In the many forms it can take, "a case study is generically a story; it presents the concrete narrative detail of actual, or at least realistic events, it has a plot, exposition, characters, and sometimes even dialogue" (Boehrer 1990). Generally, case study reports are extensively descriptive, with "the most problematic issue often referred to as being the determination of the right combination of description and analysis" (1990). Typically, authors address each step of the research process, and attempt to give the reader as much context as possible for the decisions made in the research design and for the conclusions drawn.
This contextualization usually includes a detailed explanation of the researchers' theoretical positions, of how those theories drove the inquiry or led to the guiding research questions, of the participants' backgrounds, of the processes of data collection, of the training and limitations of the coders, along with a strong attempt to make connections between the data and the conclusions evident.
Although the Berkenkotter, Huckin, and Ackerman (1988) study does not, case study reports often include the reactions of the participants to the study or to the researchers' conclusions. Because case studies tend to be exploratory, most end with implications for further study. Here researchers may identify significant variables that emerged during the research and suggest studies related to these, or the authors may suggest further general questions that their case study generated.
For example, Emig's (1971) study concludes with a section dedicated solely to the topic of implications for further research, in which she suggests several means by which this particular study could have been improved, as well as questions and ideas raised by this study which other researchers might like to address, such as: is there a correlation between a certain personality and a certain composing process profile (e.g. is there a positive correlation between ego strength and persistence in revising)?
Also included in Emig's study is a section dedicated to implications for teaching, which outlines the pedagogical ramifications of the study's findings for teachers currently involved in high school writing programs.
Sharan Merriam (1985) also offers several suggestions for alternative presentations of data:
- Prepare specialized condensations for appropriate groups.
- Replace narrative sections with a series of answers to open-ended questions.
- Present "skimmer's" summaries at beginning of each section.
- Incorporate headlines that encapsulate information from text.
- Prepare analytic summaries with supporting data appendixes.
- Present data in colorful and/or unique graphic representations.
Issues of Validity and Reliability
Once key variables have been identified, they can be analyzed. Reliability becomes a key concern at this stage, and many case study researchers go to great lengths to ensure that their interpretations of the data will be both reliable and valid. Because issues of validity and reliability are an important part of any study in the social sciences, it is important to identify some ways of dealing with results.
Multi-modal case study researchers often balance the results of their coding with data from interviews or writer's reflections upon their own work. Consequently, the researchers' conclusions become highly contextualized. For example, in a case study which looked at the time spent in different stages of the writing process, Berkenkotter concluded that her participant, Donald Murray, spent more time planning his essays than in other writing stages. The report of this case study is followed by Murray's reply, wherein he agrees with some of Berkenkotter's conclusions and disagrees with others.
As is the case with other research methodologies, issues of external validity, construct validity, and reliability need to be carefully considered.
Commentary on Case Studies
Researchers often debate the relative merits of particular methods, among them case study. In this section, we comment on two key issues. To read the commentaries, choose any of the items below:
Strengths and Weaknesses of Case Studies
Most case study advocates point out that case studies produce much more detailed information than what is available through a statistical analysis. Advocates will also hold that while statistical methods might be able to deal with situations where behavior is homogeneous and routine, case studies are needed to deal with creativity, innovation, and context. Detractors argue that case studies are difficult to generalize because of inherent subjectivity and because they are based on qualitative subjective data, generalizable only to a particular context.
Flexibility
The case study approach is a comparatively flexible method of scientific research. Because its project designs seem to emphasize exploration rather than prescription or prediction, researchers are comparatively freer to discover and address issues as they arise in their experiments. In addition, the looser format of case studies allows researchers to begin with broad questions and narrow their focus as their experiment progresses rather than attempt to predict every possible outcome before the experiment is conducted.
Emphasis on Context
By seeking to understand as much as possible about a single subject or small group of subjects, case studies specialize in "deep data," or "thick description"--information based on particular contexts that can give research results a more human face. This emphasis can help bridge the gap between abstract research and concrete practice by allowing researchers to compare their firsthand observations with the quantitative results obtained through other methods of research.
Inherent Subjectivity
"The case study has long been stereotyped as the weak sibling among social science methods," and is often criticized as being too subjective and even pseudo-scientific. Likewise, "investigators who do case studies are often regarded as having deviated from their academic disciplines, and their investigations as having insufficient precision (that is, quantification), objectivity and rigor" (Yin 1989). Opponents cite opportunities for subjectivity in the implementation, presentation, and evaluation of case study research. The approach relies on personal interpretation of data and inferences. Results may not be generalizable, are difficult to test for validity, and rarely offer a problem-solving prescription. Simply put, relying on one or a few subjects as a basis for cognitive extrapolations runs the risk of inferring too much from what might be circumstance.
High Investment
Case studies can involve learning more about the subjects being tested than most researchers would care to know--their educational background, emotional background, perceptions of themselves and their surroundings, their likes, dislikes, and so on. Because of its emphasis on "deep data," the case study is out of reach for many large-scale research projects which look at a subject pool in the tens of thousands. A budget request of $10,000 to examine 200 subjects sounds more efficient than a similar request to examine four subjects.
Ethical Considerations
Researchers conducting case studies should consider certain ethical issues. For example, many educational case studies are often financed by people who have, either directly or indirectly, power over both those being studied and those conducting the investigation (1985). This conflict of interests can hinder the credibility of the study.
The personal integrity, sensitivity, and possible prejudices and/or biases of the investigators need to be taken into consideration as well. Personal biases can creep into how the research is conducted, alternative research methods used, and the preparation of surveys and questionnaires.
A common complaint in case study research is that investigators change direction during the course of the study unaware that their original research design was inadequate for the revised investigation. Thus, the researchers leave unknown gaps and biases in the study. To avoid this, researchers should report preliminary findings so that the likelihood of bias will be reduced.
Concerns about Reliability, Validity, and Generalizability
Merriam (1985) offers several suggestions for how case study researchers might actively combat the popular attacks on the validity, reliability, and generalizability of case studies:
- Prolong the Processes of Data Gathering on Site: This will help to insure the accuracy of the findings by providing the researcher with more concrete information upon which to formulate interpretations.
- Employ the Process of "Triangulation": Use a variety of data sources as opposed to relying solely upon one avenue of observation. One example of such a data check would be what McClintock, Brannon, and Maynard (1985) refer to as a "case cluster method," that is, when a single unit within a larger case is randomly sampled, and that data treated quantitatively." For instance, in Emig's (1971) study, the case cluster method was employed, singling out the productivity of a single student named Lynn. This cluster profile included an advanced case history of the subject, specific examination and analysis of individual compositions and protocols, and extensive interview sessions. The seven remaining students were then compared with the case of Lynn, to ascertain if there are any shared, or unique dimensions to the composing process engaged in by these eight students.
- Conduct Member Checks: Initiate and maintain an active corroboration on the interpretation of data between the researcher and those who provided the data. In other words, talk to your subjects.
- Collect Referential Materials: Complement the file of materials from the actual site with additional document support. For example, Emig (1971) supports her initial propositions with historical accounts by writers such as T.S. Eliot, James Joyce, and D.H. Lawrence. Emig also cites examples of theoretical research done with regards to the creative process, as well as examples of empirical research dealing with the writing of adolescents. Specific attention is then given to the four stages description of the composing process delineated by Helmoltz, Wallas, and Cowley, as it serves as the focal point in this study.
- Engage in Peer Consultation: Prior to composing the final draft of the report, researchers should consult with colleagues in order to establish validity through pooled judgment.
Although little can be done to combat challenges concerning the generalizability of case studies, "most writers suggest that qualitative research should be judged as credible and confirmable as opposed to valid and reliable" (Merriam 1985). Likewise, it has been argued that "rather than transplanting statistical, quantitative notions of generalizability and thus finding qualitative research inadequate, it makes more sense to develop an understanding of generalization that is congruent with the basic characteristics of qualitative inquiry" (1985). After all, criticizing the case study method for being ungeneralizable is comparable to criticizing a washing machine for not being able to tell the correct time. In other words, it is unjust to criticize a method for not being able to do something which it was never originally designed to do in the first place.
Annotated Bibliography
Armisted, C. (1984). How Useful are Case Studies. Training and Development Journal, 38 (2), 75-77.
This article looks at eight types of case studies, offers pros and cons of using case studies in the classroom, and gives suggestions for successfully writing and using case studies.
Bardovi-Harlig, K. (1997). Beyond Methods: Components of Second Language Teacher Education . New York: McGraw-Hill.
A compilation of various research essays which address issues of language teacher education. Essays included are: "Non-native reading research and theory" by Lee, "The case for Psycholinguistics" by VanPatten, and "Assessment and Second Language Teaching" by Gradman and Reed.
Bartlett, L. (1989). A Question of Good Judgment; Interpretation Theory and Qualitative Enquiry Address. 70th Annual Meeting of the American Educational Research Association. San Francisco.
Bartlett selected "quasi-historical" methodology, which focuses on the "truth" found in case records, as one that will provide "good judgments" in educational inquiry. He argues that although the method is not comprehensive, it can try to connect theory with practice.
Baydere, S. et. al. (1993). Multimedia conferencing as a tool for collaborative writing: a case study in Computer Supported Collaborative Writing. New York: Springer-Verlag.
The case study by Baydere et. al. is just one of the many essays in this book found in the series "Computer Supported Cooperative Work." Denley, Witefield and May explore similar issues in their essay, "A case study in task analysis for the design of a collaborative document production system."
Berkenkotter, C., Huckin, T., N., & Ackerman J. (1988). Conventions, Conversations, and the Writer: Case Study of a Student in a Rhetoric Ph.D. Program. Research in the Teaching of English, 22, 9-44.
The authors focused on how the writing of their subject, Nate or Ackerman, changed as he became more acquainted or familiar with his field's discourse community.
Berninger, V., W., and Gans, B., M. (1986). Language Profiles in Nonspeaking Individuals of Normal Intelligence with Severe Cerebral Palsy. Augmentative and Alternative Communication, 2, 45-50.
Argues that generalizations about language abilities in patients with severe cerebral palsy (CP) should be avoided. Standardized tests of different levels of processing oral language, of processing written language, and of producing written language were administered to 3 male participants (aged 9, 16, and 40 yrs).
Bockman, J., R., and Couture, B. (1984). The Case Method in Technical Communication: Theory and Models. Texas: Association of Teachers of Technical Writing.
Examines the study and teaching of technical writing, communication of technical information, and the case method in terms of those applications.
Boehrer, J. (1990). Teaching With Cases: Learning to Question. New Directions for Teaching and Learning, 42 41-57.
This article discusses the origins of the case method, looks at the question of what is a case, gives ideas about learning in case teaching, the purposes it can serve in the classroom, the ground rules for the case discussion, including the role of the question, and new directions for case teaching.
Bowman, W. R. (1993). Evaluating JTPA Programs for Economically Disadvantaged Adults: A Case Study of Utah and General Findings . Washington: National Commission for Employment Policy.
"To encourage state-level evaluations of JTPA, the Commission and the State of Utah co-sponsored this report on the effectiveness of JTPA Title II programs for adults in Utah. The technique used is non-experimental and the comparison group was selected from registrants with Utah's Employment Security. In a step-by-step approach, the report documents how non-experimental techniques can be applied and several specific technical issues can be addressed."
Boyce, A. (1993) The Case Study Approach for Pedagogists. Annual Meeting of the American Alliance for Health, Physical Education, Recreation and Dance. (Address). Washington DC.
This paper addresses how case studies 1) bridge the gap between teaching theory and application, 2) enable students to analyze problems and develop solutions for situations that will be encountered in the real world of teaching, and 3) helps students to evaluate the feasibility of alternatives and to understand the ramifications of a particular course of action.
Carson, J. (1993) The Case Study: Ideal Home of WAC Quantitative and Qualitative Data. Annual Meeting of the Conference on College Composition and Communication. (Address). San Diego.
"Increasingly, one of the most pressing questions for WAC advocates is how to keep [WAC] programs going in the face of numerous difficulties. Case histories offer the best chance for fashioning rhetorical arguments to keep WAC programs going because they offer the opportunity to provide a coherent narrative that contextualizes all documents and data, including what is generally considered scientific data. A case study of the WAC program, . . . at Robert Morris College in Pittsburgh demonstrates the advantages of this research method. Such studies are ideal homes for both naturalistic and positivistic data as well as both quantitative and qualitative information."
---. (1991). A Cognitive Process Theory of Writing. College Composition and Communication. 32. 365-87.
No abstract available.
Cromer, R. (1994) A Case Study of Dissociations Between Language and Cognition. Constraints on Language Acquisition: Studies of Atypical Children . Hillsdale: Lawrence Erlbaum Associates, 141-153.
Crossley, M. (1983) Case Study in Comparative and International Education: An Approach to Bridging the Theory-Practice Gap. Proceedings of the 11th Annual Conference of the Australian Comparative and International Education Society. Hamilton, NZ.
Case study research, as presented here, helps bridge the theory-practice gap in comparative and international research studies of education because it focuses on the practical, day-to-day context rather than on the national arena. The paper asserts that the case study method can be valuable at all levels of research, formation, and verification of theories in education.
Daillak, R., H., and Alkin, M., C. (1982). Qualitative Studies in Context: Reflections on the CSE Studies of Evaluation Use . California: EDRS
The report shows how the Center of the Study of Evaluation (CSE) applied qualitative techniques to a study of evaluation information use in local, Los Angeles schools. It critiques the effectiveness and the limitations of using case study, evaluation, field study, and user interview survey methodologies.
Davey, L. (1991). The Application of Case Study Evaluations. ERIC/TM Digest.
This article examines six types of case studies, the type of evaluation questions that can be answered, the functions served, some design features, and some pitfalls of the method.
Deutch, C. E. (1996). A course in research ethics for graduate students. College Teaching, 44, 2, 56-60.
This article describes a one-credit discussion course in research ethics for graduate students in biology. Case studies are focused on within the four parts of the course: 1) major issues, 2 )practical issues in scholarly work, 3) ownership of research results, and 4) training and personal decisions.
DeVoss, G. (1981). Ethics in Fieldwork Research. RIE 27p. (ERIC)
This article examines four of the ethical problems that can happen when conducting case study research: acquiring permission to do research, knowing when to stop digging, the pitfalls of doing collaborative research, and preserving the integrity of the participants.
Driscoll, A. (1985). Case Study of a Research Intervention: the University of Utah’s Collaborative Approach . San Francisco: Far West Library for Educational Research Development.
Paper presented at the annual meeting of the American Association of Colleges of Teacher Education, Denver, CO, March 1985. Offers information of in-service training, specifically case studies application.
Ellram, L. M. (1996). The Use of the Case Study Method in Logistics Research. Journal of Business Logistics, 17, 2, 93.
This article discusses the increased use of case study in business research, and the lack of understanding of when and how to use case study methodology in business.
Emig, J. (1971) The Composing Processes of Twelfth Graders . Urbana: NTCE.
This case study uses observation, tape recordings, writing samples, and school records to show that writing in reflexive and extensive situations caused different lengths of discourse and different clusterings of the components of the writing process.
Feagin, J. R. (1991). A Case For the Case Study . Chapel Hill: The University of North Carolina Press.
This book discusses the nature, characteristics, and basic methodological issues of the case study as a research method.
Feldman, H., Holland, A., & Keefe, K. (1989) Language Abilities after Left Hemisphere Brain Injury: A Case Study of Twins. Topics in Early Childhood Special Education, 9, 32-47.
"Describes the language abilities of 2 twin pairs in which 1 twin (the experimental) suffered brain injury to the left cerebral hemisphere around the time of birth and1 twin (the control) did not. One pair of twins was initially assessed at age 23 mo. and the other at about 30 mo.; they were subsequently evaluated in their homes 3 times at about 6-mo intervals."
Fidel, R. (1984). The Case Study Method: A Case Study. Library and Information Science Research, 6.
The article describes the use of case study methodology to systematically develop a model of online searching behavior in which study design is flexible, subject manner determines data gathering and analyses, and procedures adapt to the study's progressive change.
Flower, L., & Hayes, J. R. (1984). Images, Plans and Prose: The Representation of Meaning in Writing. Written Communication, 1, 120-160.
Explores the ways in which writers actually use different forms of knowing to create prose.
Frey, L. R. (1992). Interpreting Communication Research: A Case Study Approach Englewood Cliffs, N.J.: Prentice Hall.
The book discusses research methodologies in the Communication field. It focuses on how case studies bridge the gap between communication research, theory, and practice.
Gilbert, V. K. (1981). The Case Study as a Research Methodology: Difficulties and Advantages of Integrating the Positivistic, Phenomenological and Grounded Theory Approaches . The Annual Meeting of the Canadian Association for the Study of Educational Administration. (Address) Halifax, NS, Can.
This study on an innovative secondary school in England shows how a "low-profile" participant-observer case study was crucial to the initial observation, the testing of hypotheses, the interpretive approach, and the grounded theory.
Gilgun, J. F. (1994). A Case for Case Studies in Social Work Research. Social Work, 39, 4, 371-381.
This article defines case study research, presents guidelines for evaluation of case studies, and shows the relevance of case studies to social work research. It also looks at issues such as evaluation and interpretations of case studies.
Glennan, S. L., Sharp-Bittner, M. A. & Tullos, D. C. (1991). Augmentative and Alternative Communication Training with a Nonspeaking Adult: Lessons from MH. Augmentative and Alternative Communication, 7, 240-7.
"A response-guided case study documented changes in a nonspeaking 36-yr-old man's ability to communicate using 3 trained augmentative communication modes. . . . Data were collected in videotaped interaction sessions between the nonspeaking adult and a series of adult speaking."
Graves, D. (1981). An Examination of the Writing Processes of Seven Year Old Children. Research in the Teaching of English, 15, 113-134.
Hamel, J. (1993). Case Study Methods . Newbury Park: Sage. .
"In a most economical fashion, Hamel provides a practical guide for producing theoretically sharp and empirically sound sociological case studies. A central idea put forth by Hamel is that case studies must "locate the global in the local" thus making the careful selection of the research site the most critical decision in the analytic process."
Karthigesu, R. (1986, July). Television as a Tool for Nation-Building in the Third World: A Post-Colonial Pattern, Using Malaysia as a Case-Study. International Television Studies Conference. (Address). London, 10-12.
"The extent to which Television Malaysia, as a national mass media organization, has been able to play a role in nation building in the post-colonial period is . . . studied in two parts: how the choice of a model of nation building determines the character of the organization; and how the character of the organization influences the output of the organization."
Kenny, R. (1984). Making the Case for the Case Study. Journal of Curriculum Studies, 16, (1), 37-51.
The article looks at how and why the case study is justified as a viable and valuable approach to educational research and program evaluation.
Knirk, F. (1991). Case Materials: Research and Practice. Performance Improvement Quarterly, 4 (1 ), 73-81.
The article addresses the effectiveness of case studies, subject areas where case studies are commonly used, recent examples of their use, and case study design considerations.
Klos, D. (1976). Students as Case Writers. Teaching of Psychology, 3.2, 63-66.
This article reviews a course in which students gather data for an original case study of another person. The task requires the students to design the study, collect the data, write the narrative, and interpret the findings.
Leftwich, A. (1981). The Politics of Case Study: Problems of Innovation in University Education. Higher Education Review, 13.2, 38-64.
The article discusses the use of case studies as a teaching method. Emphasis is on the instructional materials, interdisciplinarity, and the complex relationships within the university that help or hinder the method.
Mabrito, M. (1991, Oct.). Electronic Mail as a Vehicle for Peer Response: Conversations of High and Low Apprehensive Writers. Written Communication, 509-32.
McCarthy, S., J. (1955). The Influence of Classroom Discourse on Student Texts: The Case of Ella . East Lansing: Institute for Research on Teaching.
A look at how students of color become marginalized within traditional classroom discourse. The essay follows the struggles of one black student: Ella.
Matsuhashi, A., ed. (1987). Writing in Real Time: Modeling Production Processes Norwood, NJ: Ablex Publishing Corporation.
Investigates how writers plan to produce discourse for different purposes to report, to generalize, and to persuade, as well as how writers plan for sentence level units of language. To learn about planning, an observational measure of pause time was used" (ERIC).
Merriam, S. B. (1985). The Case Study in Educational Research: A Review of Selected Literature. Journal of Educational Thought, 19.3, 204-17.
The article examines the characteristics of, philosophical assumptions underlying the case study, the mechanics of conducting a case study, and the concerns about the reliability, validity, and generalizability of the method.
---. (1988). Case Study Research in Education: A Qualitative Approach San Francisco: Jossey Bass.
Merry, S. E., & Milner, N. eds. (1993). The Possibility of Popular Justice: A Case Study of Community Mediation in the United States . Ann Arbor: U of Michigan.
". . . this volume presents a case study of one experiment in popular justice, the San Francisco Community Boards. This program has made an explicit claim to create an alternative justice, or new justice, in the midst of a society ordered by state law. The contributors to this volume explore the history and experience of the program and compare it to other versions of popular justice in the United States, Europe, and the Third World."
Merseth, K. K. (1991). The Case for Cases in Teacher Education. RIE. 42p. (ERIC).
This monograph argues that the case method of instruction offers unique potential for revitalizing the field of teacher education.
Michaels, S. (1987). Text and Context: A New Approach to the Study of Classroom Writing. Discourse Processes, 10, 321-346.
"This paper argues for and illustrates an approach to the study of writing that integrates ethnographic analysis of classroom interaction with linguistic analysis of written texts and teacher/student conversational exchanges. The approach is illustrated through a case study of writing in a single sixth grade classroom during a single writing assignment."
Milburn, G. (1995). Deciphering a Code or Unraveling a Riddle: A Case Study in the Application of a Humanistic Metaphor to the Reporting of Social Studies Teaching. Theory and Research in Education, 13.
This citation serves as an example of how case studies document learning procedures in a senior-level economics course.
Milley, J. E. (1979). An Investigation of Case Study as an Approach to Program Evaluation. 19th Annual Forum of the Association for Institutional Research. (Address). San Diego.
The case study method merged a narrative report focusing on the evaluator as participant-observer with document review, interview, content analysis, attitude questionnaire survey, and sociogram analysis. Milley argues that case study program evaluation has great potential for widespread use.
Minnis, J. R. (1985, Sept.). Ethnography, Case Study, Grounded Theory, and Distance Education Research. Distance Education, 6.2.
This article describes and defines the strengths and weaknesses of ethnography, case study, and grounded theory.
Nunan, D. (1992). Collaborative language learning and teaching . New York: Cambridge University Press.
Included in this series of essays is Peter Sturman’s "Team Teaching: a case study from Japan" and David Nunan’s own "Toward a collaborative approach to curriculum development: a case study."
Nystrand, M., ed. (1982). What Writers Know: The Language, Process, and Structure of Written Discourse . New York: Academic Press.
Owenby, P. H. (1992). Making Case Studies Come Alive. Training, 29, (1), 43-46. (ERIC)
This article provides tips for writing more effective case studies.
---. (1981). Pausing and Planning: The Tempo of Writer Discourse Production. Research in the Teaching of English, 15 (2),113-34.
Perl, S. (1979). The Composing Processes of Unskilled College Writers. Research in the Teaching of English, 13, 317-336.
"Summarizes a study of five unskilled college writers, focusing especially on one of the five, and discusses the findings in light of current pedagogical practice and research design."
Pilcher J. and A. Coffey. eds. (1996). Gender and Qualitative Research . Brookfield: Aldershot, Hants, England.
This book provides a series of essays which look at gender identity research, qualitative research and applications of case study to questions of gendered pedagogy.
Pirie, B. S. (1993). The Case of Morty: A Four Year Study. Gifted Education International, 9 (2), 105-109.
This case study describes a boy from kindergarten through third grade with above average intelligence but difficulty in learning to read, write, and spell.
Popkewitz, T. (1993). Changing Patterns of Power: Social Regulation and Teacher Education Reform. Albany: SUNY Press.
Popkewitz edits this series of essays that address case studies on educational change and the training of teachers. The essays vary in terms of discipline and scope. Also, several authors include case studies of educational practices in countries other than the United States.
---. (1984). The Predrafting Processes of Four High- and Four Low Apprehensive Writers. Research in the Teaching of English, 18, (1), 45-64.
Rasmussen, P. (1985, March) A Case Study on the Evaluation of Research at the Technical University of Denmark. International Journal of Institutional Management in Higher Education, 9 (1).
This is an example of a case study methodology used to evaluate the chemistry and chemical engineering departments at the University of Denmark.
Roth, K. J. (1986). Curriculum Materials, Teacher Talk, and Student Learning: Case Studies in Fifth-Grade Science Teaching . East Lansing: Institute for Research on Teaching.
Roth offers case studies on elementary teachers, elementary school teaching, science studies and teaching, and verbal learning.
Selfe, C. L. (1985). An Apprehensive Writer Composes. When a Writer Can't Write: Studies in Writer's Block and Other Composing-Process Problems . (pp. 83-95). Ed. Mike Rose. NMY: Guilford.
Smith-Lewis, M., R. and Ford, A. (1987). A User's Perspective on Augmentative Communication. Augmentative and Alternative Communication, 3, 12-7.
"During a series of in-depth interviews, a 25-yr-old woman with cerebral palsy who utilized augmentative communication reflected on the effectiveness of the devices designed for her during her school career."
St. Pierre, R., G. (1980, April). Follow Through: A Case Study in Metaevaluation Research . 64th Annual Meeting of the American Educational Research Association. (Address).
The three approaches to metaevaluation are evaluation of primary evaluations, integrative meta-analysis with combined primary evaluation results, and re-analysis of the raw data from a primary evaluation.
Stahler, T., M. (1996, Feb.) Early Field Experiences: A Model That Worked. ERIC.
"This case study of a field and theory class examines a model designed to provide meaningful field experiences for preservice teachers while remaining consistent with the instructor's beliefs about the role of teacher education in preparing teachers for the classroom."
Stake, R. E. (1995). The Art of Case Study Research. Thousand Oaks: Sage Publications.
This book examines case study research in education and case study methodology.
Stiegelbauer, S. (1984) Community, Context, and Co-curriculum: Situational Factors Influencing School Improvements in a Study of High Schools. Presented at the annual meeting of the American Educational Research Association, New Orleans, LA.
Discussion of several case studies: one looking at high school environments, another examining educational innovations.
Stolovitch, H. (1990). Case Study Method. Performance And Instruction, 29, (9), 35-37.
This article describes the case study method as a form of simulation and presents guidelines for their use in professional training situations.
Thaller, E. (1994). Bibliography for the Case Method: Using Case Studies in Teacher Education. RIE. 37 p.
This bibliography presents approximately 450 citations on the use of case studies in teacher education from 1921-1993.
Thrane, T. (1986). On Delimiting the Senses of Near-Synonyms in Historical Semantics: A Case Study of Adjectives of 'Moral Sufficiency' in the Old English Andreas. Linguistics Across Historical and Geographical Boundaries: In Honor of Jacek Fisiak on the Occasion of his Fiftieth Birthday . Berlin: Mouton de Gruyter.
United Nations. (1975). Food and Agriculture Organization. Report on the FAO/UNFPA Seminar on Methodology, Research and Country: Case Studies on Population, Employment and Productivity . Rome: United Nations.
This example case study shows how the methodology can be used in a demographic and psychographic evaluation. At the same time, it discusses the formation and instigation of the case study methodology itself.
Van Vugt, J. P., ed. (1994). Aids Prevention and Services: Community Based Research . Westport: Bergin and Garvey.
"This volume has been five years in the making. In the process, some of the policy applications called for have met with limited success, such as free needle exchange programs in a limited number of American cities, providing condoms to prison inmates, and advertisements that depict same-sex couples. Rather than dating our chapters that deal with such subjects, such policy applications are verifications of the type of research demonstrated here. Furthermore, they indicate the critical need to continue community based research in the various communities threatened by acquired immuno-deficiency syndrome (AIDS) . . . "
Welch, W., ed. (1981, May). Case Study Methodology in Educational Evaluation. Proceedings of the Minnesota Evaluation Conference. Minnesota. (Address).
The four papers in these proceedings provide a comprehensive picture of the rationale, methodology, strengths, and limitations of case studies.
Williams, G. (1987). The Case Method: An Approach to Teaching and Learning in Educational Administration. RIE, 31p.
This paper examines the viability of the case method as a teaching and learning strategy in instructional systems geared toward the training of personnel of the administration of various aspects of educational systems.
Yin, R. K. (1993). Advancing Rigorous Methodologies: A Review of 'Towards Rigor in Reviews of Multivocal Literatures.' Review of Educational Research, 61, (3).
"R. T. Ogawa and B. Malen's article does not meet its own recommended standards for rigorous testing and presentation of its own conclusions. Use of the exploratory case study to analyze multivocal literatures is not supported, and the claim of grounded theory to analyze multivocal literatures may be stronger."
---. (1989). Case Study Research: Design and Methods. London: Sage Publications Inc.
This book discusses in great detail, the entire design process of the case study, including entire chapters on collecting evidence, analyzing evidence, composing the case study report, and designing single and multiple case studies.
Related Links
Consider the following list of related Web sites for more information on the topic of case study research. Note: although many of the links cover the general category of qualitative research, all have sections that address issues of case studies.
- Sage Publications on Qualitative Methodology: Search here for a comprehensive list of new books being published about "Qualitative Methodology" http://www.sagepub.co.uk/
- The International Journal of Qualitative Studies in Education: An on-line journal "to enhance the theory and practice of qualitative research in education." On-line submissions are welcome. http://www.tandf.co.uk/journals/tf/09518398.html
- Qualitative Research Resources on the Internet: From syllabi to home pages to bibliographies. All links relate somehow to qualitative research. http://www.nova.edu/ssss/QR/qualres.html
Citation Information
Bronwyn Becker, Patrick Dawson, Karen Devine, Carla Hannum, Steve Hill, Jon Leydens, Debbie Matuskevich, Carol Traver, and Mike Palmquist. (1994-2024). Case Studies. The WAC Clearinghouse. Colorado State University. Available at https://wac.colostate.edu/repository/writing/guides/.
Copyright Information
Copyright © 1994-2024 Colorado State University and/or this site's authors, developers, and contributors . Some material displayed on this site is used with permission.
- Bipolar Disorder
- Therapy Center
- When To See a Therapist
- Types of Therapy
- Best Online Therapy
- Best Couples Therapy
- Best Family Therapy
- Managing Stress
- Sleep and Dreaming
- Understanding Emotions
- Self-Improvement
- Healthy Relationships
- Student Resources
- Personality Types
- Guided Meditations
- Verywell Mind Insights
- 2024 Verywell Mind 25
- Mental Health in the Classroom
- Editorial Process
- Meet Our Review Board
- Crisis Support
What Is a Case Study?
Weighing the pros and cons of this method of research
Kendra Cherry, MS, is a psychosocial rehabilitation specialist, psychology educator, and author of the "Everything Psychology Book."
:max_bytes(150000):strip_icc():format(webp)/IMG_9791-89504ab694d54b66bbd72cb84ffb860e.jpg)
Cara Lustik is a fact-checker and copywriter.
:max_bytes(150000):strip_icc():format(webp)/Cara-Lustik-1000-77abe13cf6c14a34a58c2a0ffb7297da.jpg)
Verywell / Colleen Tighe
- Pros and Cons
What Types of Case Studies Are Out There?
Where do you find data for a case study, how do i write a psychology case study.
A case study is an in-depth study of one person, group, or event. In a case study, nearly every aspect of the subject's life and history is analyzed to seek patterns and causes of behavior. Case studies can be used in many different fields, including psychology, medicine, education, anthropology, political science, and social work.
The point of a case study is to learn as much as possible about an individual or group so that the information can be generalized to many others. Unfortunately, case studies tend to be highly subjective, and it is sometimes difficult to generalize results to a larger population.
While case studies focus on a single individual or group, they follow a format similar to other types of psychology writing. If you are writing a case study, we got you—here are some rules of APA format to reference.
At a Glance
A case study, or an in-depth study of a person, group, or event, can be a useful research tool when used wisely. In many cases, case studies are best used in situations where it would be difficult or impossible for you to conduct an experiment. They are helpful for looking at unique situations and allow researchers to gather a lot of˜ information about a specific individual or group of people. However, it's important to be cautious of any bias we draw from them as they are highly subjective.
What Are the Benefits and Limitations of Case Studies?
A case study can have its strengths and weaknesses. Researchers must consider these pros and cons before deciding if this type of study is appropriate for their needs.
One of the greatest advantages of a case study is that it allows researchers to investigate things that are often difficult or impossible to replicate in a lab. Some other benefits of a case study:
- Allows researchers to capture information on the 'how,' 'what,' and 'why,' of something that's implemented
- Gives researchers the chance to collect information on why one strategy might be chosen over another
- Permits researchers to develop hypotheses that can be explored in experimental research
On the other hand, a case study can have some drawbacks:
- It cannot necessarily be generalized to the larger population
- Cannot demonstrate cause and effect
- It may not be scientifically rigorous
- It can lead to bias
Researchers may choose to perform a case study if they want to explore a unique or recently discovered phenomenon. Through their insights, researchers develop additional ideas and study questions that might be explored in future studies.
It's important to remember that the insights from case studies cannot be used to determine cause-and-effect relationships between variables. However, case studies may be used to develop hypotheses that can then be addressed in experimental research.
Case Study Examples
There have been a number of notable case studies in the history of psychology. Much of Freud's work and theories were developed through individual case studies. Some great examples of case studies in psychology include:
- Anna O : Anna O. was a pseudonym of a woman named Bertha Pappenheim, a patient of a physician named Josef Breuer. While she was never a patient of Freud's, Freud and Breuer discussed her case extensively. The woman was experiencing symptoms of a condition that was then known as hysteria and found that talking about her problems helped relieve her symptoms. Her case played an important part in the development of talk therapy as an approach to mental health treatment.
- Phineas Gage : Phineas Gage was a railroad employee who experienced a terrible accident in which an explosion sent a metal rod through his skull, damaging important portions of his brain. Gage recovered from his accident but was left with serious changes in both personality and behavior.
- Genie : Genie was a young girl subjected to horrific abuse and isolation. The case study of Genie allowed researchers to study whether language learning was possible, even after missing critical periods for language development. Her case also served as an example of how scientific research may interfere with treatment and lead to further abuse of vulnerable individuals.
Such cases demonstrate how case research can be used to study things that researchers could not replicate in experimental settings. In Genie's case, her horrific abuse denied her the opportunity to learn a language at critical points in her development.
This is clearly not something researchers could ethically replicate, but conducting a case study on Genie allowed researchers to study phenomena that are otherwise impossible to reproduce.
There are a few different types of case studies that psychologists and other researchers might use:
- Collective case studies : These involve studying a group of individuals. Researchers might study a group of people in a certain setting or look at an entire community. For example, psychologists might explore how access to resources in a community has affected the collective mental well-being of those who live there.
- Descriptive case studies : These involve starting with a descriptive theory. The subjects are then observed, and the information gathered is compared to the pre-existing theory.
- Explanatory case studies : These are often used to do causal investigations. In other words, researchers are interested in looking at factors that may have caused certain things to occur.
- Exploratory case studies : These are sometimes used as a prelude to further, more in-depth research. This allows researchers to gather more information before developing their research questions and hypotheses .
- Instrumental case studies : These occur when the individual or group allows researchers to understand more than what is initially obvious to observers.
- Intrinsic case studies : This type of case study is when the researcher has a personal interest in the case. Jean Piaget's observations of his own children are good examples of how an intrinsic case study can contribute to the development of a psychological theory.
The three main case study types often used are intrinsic, instrumental, and collective. Intrinsic case studies are useful for learning about unique cases. Instrumental case studies help look at an individual to learn more about a broader issue. A collective case study can be useful for looking at several cases simultaneously.
The type of case study that psychology researchers use depends on the unique characteristics of the situation and the case itself.
There are a number of different sources and methods that researchers can use to gather information about an individual or group. Six major sources that have been identified by researchers are:
- Archival records : Census records, survey records, and name lists are examples of archival records.
- Direct observation : This strategy involves observing the subject, often in a natural setting . While an individual observer is sometimes used, it is more common to utilize a group of observers.
- Documents : Letters, newspaper articles, administrative records, etc., are the types of documents often used as sources.
- Interviews : Interviews are one of the most important methods for gathering information in case studies. An interview can involve structured survey questions or more open-ended questions.
- Participant observation : When the researcher serves as a participant in events and observes the actions and outcomes, it is called participant observation.
- Physical artifacts : Tools, objects, instruments, and other artifacts are often observed during a direct observation of the subject.
If you have been directed to write a case study for a psychology course, be sure to check with your instructor for any specific guidelines you need to follow. If you are writing your case study for a professional publication, check with the publisher for their specific guidelines for submitting a case study.
Here is a general outline of what should be included in a case study.
Section 1: A Case History
This section will have the following structure and content:
Background information : The first section of your paper will present your client's background. Include factors such as age, gender, work, health status, family mental health history, family and social relationships, drug and alcohol history, life difficulties, goals, and coping skills and weaknesses.
Description of the presenting problem : In the next section of your case study, you will describe the problem or symptoms that the client presented with.
Describe any physical, emotional, or sensory symptoms reported by the client. Thoughts, feelings, and perceptions related to the symptoms should also be noted. Any screening or diagnostic assessments that are used should also be described in detail and all scores reported.
Your diagnosis : Provide your diagnosis and give the appropriate Diagnostic and Statistical Manual code. Explain how you reached your diagnosis, how the client's symptoms fit the diagnostic criteria for the disorder(s), or any possible difficulties in reaching a diagnosis.
Section 2: Treatment Plan
This portion of the paper will address the chosen treatment for the condition. This might also include the theoretical basis for the chosen treatment or any other evidence that might exist to support why this approach was chosen.
- Cognitive behavioral approach : Explain how a cognitive behavioral therapist would approach treatment. Offer background information on cognitive behavioral therapy and describe the treatment sessions, client response, and outcome of this type of treatment. Make note of any difficulties or successes encountered by your client during treatment.
- Humanistic approach : Describe a humanistic approach that could be used to treat your client, such as client-centered therapy . Provide information on the type of treatment you chose, the client's reaction to the treatment, and the end result of this approach. Explain why the treatment was successful or unsuccessful.
- Psychoanalytic approach : Describe how a psychoanalytic therapist would view the client's problem. Provide some background on the psychoanalytic approach and cite relevant references. Explain how psychoanalytic therapy would be used to treat the client, how the client would respond to therapy, and the effectiveness of this treatment approach.
- Pharmacological approach : If treatment primarily involves the use of medications, explain which medications were used and why. Provide background on the effectiveness of these medications and how monotherapy may compare with an approach that combines medications with therapy or other treatments.
This section of a case study should also include information about the treatment goals, process, and outcomes.
When you are writing a case study, you should also include a section where you discuss the case study itself, including the strengths and limitiations of the study. You should note how the findings of your case study might support previous research.
In your discussion section, you should also describe some of the implications of your case study. What ideas or findings might require further exploration? How might researchers go about exploring some of these questions in additional studies?
Need More Tips?
Here are a few additional pointers to keep in mind when formatting your case study:
- Never refer to the subject of your case study as "the client." Instead, use their name or a pseudonym.
- Read examples of case studies to gain an idea about the style and format.
- Remember to use APA format when citing references .
Crowe S, Cresswell K, Robertson A, Huby G, Avery A, Sheikh A. The case study approach . BMC Med Res Methodol . 2011;11:100.
Crowe S, Cresswell K, Robertson A, Huby G, Avery A, Sheikh A. The case study approach . BMC Med Res Methodol . 2011 Jun 27;11:100. doi:10.1186/1471-2288-11-100
Gagnon, Yves-Chantal. The Case Study as Research Method: A Practical Handbook . Canada, Chicago Review Press Incorporated DBA Independent Pub Group, 2010.
Yin, Robert K. Case Study Research and Applications: Design and Methods . United States, SAGE Publications, 2017.
By Kendra Cherry, MSEd Kendra Cherry, MS, is a psychosocial rehabilitation specialist, psychology educator, and author of the "Everything Psychology Book."
- Skip to main content
- Skip to primary sidebar
- Skip to footer
- QuestionPro

- Solutions Industries Gaming Automotive Sports and events Education Government Travel & Hospitality Financial Services Healthcare Cannabis Technology Use Case NPS+ Communities Audience Contactless surveys Mobile LivePolls Member Experience GDPR Positive People Science 360 Feedback Surveys
- Resources Blog eBooks Survey Templates Case Studies Training Help center
Home Market Research
Participant observation: What it is, types & uses

Participant observation has been widely used in disciplines such as anthropology, sociology, communication studies, political science, social psychology, and even in market research.
To help you get a broad overview of this methodology, in this article we have compiled its most important characteristics, its importance, and some of the types of participant observation that you can put into practice for your next study.
LEARN ABOUT: Behavioral Research
What is participant observation?
Participant observation is a qualitative research methodology in which the researcher studies a group not only through observation, but also by participating in its activities.
In this qualitative observation methodology, the researcher immerses himself in the daily activities of the participants in order to record the behavior in as many scenarios as possible.
Thanks to the immersion in the study place, the researchers can observe the daily life of the people: their exchanges with each other, their formal and informal conversations, habits, etc.
It offers researchers the opportunity to collect honest and intimate information about people. However, this information is filtered through the perspective of researchers who, by using this method, run the risk of losing their objectivity and altering with their presence the behavior of the groups they study.
LEARN ABOUT: Research Process Steps

Importance of participant observation
Participant observation is a method that helps you see and understand what people are doing and compare it with what they say. In this way, you help researchers know if the people with whom you are conducting a study act differently from what they are described.
It also allows the researcher to better understand what is happening in a given group and its cultural environment, giving greater credibility to their interpretations of the observation.
In addition, it allows the researcher to collect qualitative data through various types of interviews and quantitative data through surveys and different quantitative observation techniques.
Characteristics of participant observation
Participant observation has historically been associated with a form of field research in which the researcher resides for long periods of time in a small community.
Today, this methodology is used in a wide variety of settings and for widely varying periods of time, from a single interaction to many years. But it is usually characterized by the following points:
- The long-term nature of the interaction between the researcher and the participants as part of the fieldwork process.
- A wide range of relationship dynamics that it studies, such as differences in status between the two parties, differences in power and educational differences, as well as degrees of formality. Differences in power can have their origin in gender, social class, health and other aspects.
- The variety of settings, from close interpersonal interactions to observing public gatherings and actual participation in social events.
- In many cases, research takes place in settings unfamiliar to the researcher, which will make her presentation and interaction with others especially sensitive.
- There may be different ethical codes between the groups studied and those of the researcher’s country or institution of origin. They may also differ from the ethical principles followed by the host government, non-governmental organizations in the area, or agencies funding the research .
- The changing nature of the researcher’s roles and relationships with the studied group over time.
- The use of technology to document observations, including mapping, photography, and video and audio recording.
LEARN ABOUT: Social Communication Questionnaire
Types of participant observation
Now that you know what this method is and what its most common characteristics are, we will introduce you to the types that exist.
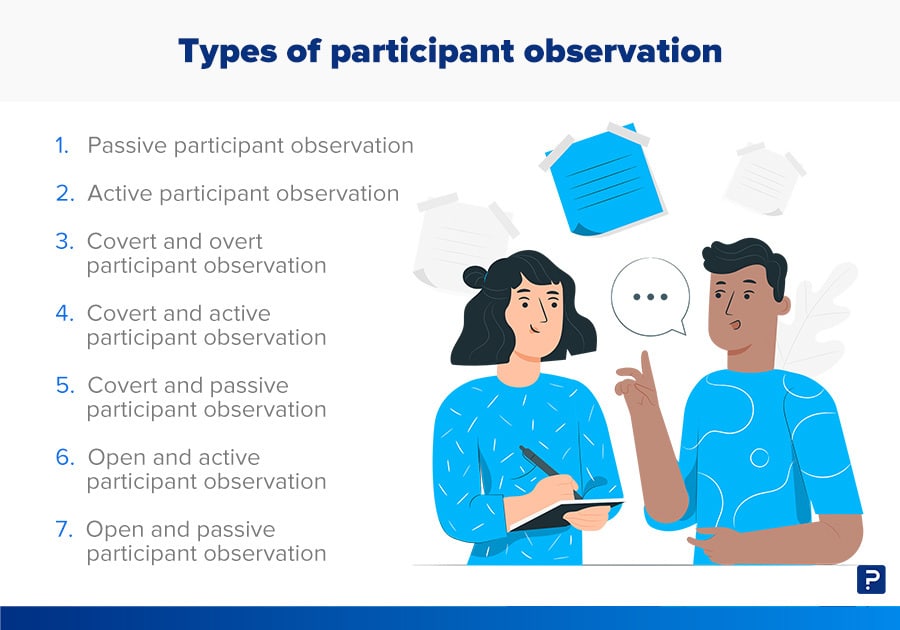
Passive participant observation
Researchers observe and record the behaviors of their subjects in their own environment without conversing or interacting with them in any way.
Many of the studies that use this form of participant observation are studies in which researchers observe people’s behavior and communications in public places, such as restaurants, coffee shops, transportation hubs, and even on the Internet through innovative methods such as netnography .
Active participant observation
In this way, researchers converse with their subjects and participate in the daily life of the groups they study, including their activities, c ustoms, rituals, routines, etc.
The degree of commitment of researchers to these groups varies. Some researchers limit their interactions to interviews, while others engage in all aspects of their subjects’ lives.
Examples of this form of participant observation are studies in which researchers lived for long periods of time among different ethnic, cultural, or religious communities.
Covert and overt
In covert participant observation, researchers do not make their presence known to their subjects and, if they do, they do not identify themselves as investigators, whereas in open participant observation they do.
However, even when the investigation is open, investigators often do not inform the people they meet in the course of their investigation of the specific purpose of the investigation, nor do they inform everyone they meet that they are researchers, as this could unnecessarily interrupt conversations and events being observed.
Covert and active
Covert and active participant observation has several advantages. In this type of participant observation, researchers can have access to a group that they would not otherwise have the opportunity to observe, and they can experience the practices of the group as they are experienced by the members of the group.
Generally, researchers can alter group behavior by their presence, but in this form of participant observation, groups would not consciously change their behavior in response to the researcher’s presence because they are not aware of being observed.
Covert and passive
In the case of covert and passive participant observation, researchers are not likely to alter the behaviors of their subjects, since the researchers do not actively engage with their subjects and because the subjects are also not aware that they are being observed.
However, since observation is passive, researchers do not have the opportunity to experience the lives of their subjects for themselves.
Open and active
If observation is open and active, people can participate in and experience their subjects’ activities as their subjects would, but they run the risk of both changing the behavior of their subjects through their interactions with them, and that their subjects change their behavior by themselves knowing that they are being studied.
Open and passive
As in the case of covert and passive participant observation, researchers do not run the risk that their presence alters the behavior of the groups they study through their interactions with them.
However, the guinea pig effect is a problem for this form of observation, unlike the case of covert and passive participant observation, because the participants are aware that they are being studied. Furthermore, researchers cannot experience the world as it is as subjects would.
As you can see, participant observation is a research method that provides valuable information about the social and cultural relationships of a group or community over time.
Uncover the insights that matter the most. Use QuestionPro’s market research platform to uncover complex insights that can propel your business to the forefront of your industry.
MORE LIKE THIS

QuestionPro Thrive: A Space to Visualize & Share the Future of Technology
Jun 18, 2024

Relationship NPS Fails to Understand Customer Experiences — Tuesday CX

CX Platform: Top 13 CX Platforms to Drive Customer Success
Jun 17, 2024

How to Know Whether Your Employee Initiatives are Working
Other categories.
- Academic Research
- Artificial Intelligence
- Assessments
- Brand Awareness
- Case Studies
- Communities
- Consumer Insights
- Customer effort score
- Customer Engagement
- Customer Experience
- Customer Loyalty
- Customer Research
- Customer Satisfaction
- Employee Benefits
- Employee Engagement
- Employee Retention
- Friday Five
- General Data Protection Regulation
- Insights Hub
- Life@QuestionPro
- Market Research
- Mobile diaries
- Mobile Surveys
- New Features
- Online Communities
- Question Types
- Questionnaire
- QuestionPro Products
- Release Notes
- Research Tools and Apps
- Revenue at Risk
- Survey Templates
- Training Tips
- Tuesday CX Thoughts (TCXT)
- Uncategorized
- Video Learning Series
- What’s Coming Up
- Workforce Intelligence

Reporting Participant Characteristics in a Research Paper
A report on a scientific study using human participants will include a description of the participant characteristics. This is included as a subsection of the “Methods” section, usually called “Participants” or “Participant Characteristics.” The purpose is to give readers information on the number and type of study participants, as a way of clarifying to whom the study findings apply and shedding light on the generalizability of the findings as well as any possible limitations. Accurate reporting is needed for replication studies that might be carried out in the future.
The “Participants” subsection should be fairly short and should tell readers about the population pool, how many participants were included in the study sample, and what kind of sample they represent, such as random, snowball, etc. There is no need to give a lengthy description of the method used to select or recruit the participants, as these topics belong in a separate “Procedures” subsection that is also under “Methods.” The subsection on “Participant Characteristics” only needs to provide facts on the participants themselves.
Report the participants’ genders (how many male and female participants) and ages (the age range and, if appropriate, the standard deviation). In particular, if you are writing for an international audience, specify the country and region or cities where the participants lived. If the study invited only participants with certain characteristics, report this, too. For example, tell readers if the participants all had autism, were left-handed, or had participated in sports within the past year.
Related: Finished preparing the methods sections for your research paper ? Find out why the “Methods” section is so important now!
Next, use your judgment to identify other pieces of information that are relevant to the study. For a detailed tutorial on reporting “Participant Characteristics,” see Alice Frye’s “Method Section: Describing participants.” Frye reminds authors to mention if only people with certain characteristics or backgrounds were included in the study. Did all the participants work at the same company? Were the students at the same school? Did they represent a range of socioeconomic backgrounds? Did they come from both urban and rural backgrounds? Were they physically and emotionally healthy? Similarly, mention if the study sample excluded people with certain characteristics.
If you are going to examine any participant characteristics as factors in the analysis, include a description of these. For instance, if you plan to examine the influence of teachers’ years of experience on their attitude toward new technology, then you should report the range of the teachers’ years of experience. If you plan to study how children’s socioeconomic level relates to their test scores, you should briefly mention that the children in the sample came from low, middle, and high-income backgrounds. Finally, mention whether the participants participated voluntarily. Include information on whether they gave informed consent (if the participants were children, mention that their parents consented to their participation). Also, mention if the participants received any sort of compensation or benefit for their participation, such as money or course credit.
Case Studies and Qualitative Reports
Case studies and qualitative reports may have only a few participants or even a single participant. If there is space to do so, you can write a brief background of each participant in the “Participants” section and include relevant information on the participant’s birthplace, current place of residence, language, and any life experience that is relevant to the study theme. If you have permission to use the participant’s name, do so. Otherwise, use a different name and add a note to readers that the name is a pseudonym. Alternatively, you might label the participants with numbers (e.g., Student 1, Student 2) or letters (e.g., Doctor A, Doctor B, etc.), or use initials to identify them (e.g., KY, JM).
Use Past Tense
Remember to use past tense when writing the “Participants” section . This is because you are describing what the participants’ characteristics were at the time of data collection . By the time your article is published, the participants’ characteristics may have changed. For example, they may be a year older and have more work experience. Their socioeconomic level may have changed since the study. In some cases, participants may even have passed away. While characteristics like gender and race are either unlikely or impossible to change, the whole section is written in the past tense to maintain a consistent style and to avoid making unsupported claims about what the participants’ current status is.
Rate this article Cancel Reply
Your email address will not be published.

Enago Academy's Most Popular Articles

- AI in Academia
- Infographic
- Manuscripts & Grants
- Reporting Research
- Trending Now
Can AI Tools Prepare a Research Manuscript From Scratch? — A comprehensive guide
As technology continues to advance, the question of whether artificial intelligence (AI) tools can prepare…

Abstract Vs. Introduction — Do you know the difference?
Ross wants to publish his research. Feeling positive about his research outcomes, he begins to…

- Old Webinars
- Webinar Mobile App
Demystifying Research Methodology With Field Experts
Choosing research methodology Research design and methodology Evidence-based research approach How RAxter can assist researchers

- Manuscript Preparation
- Publishing Research
How to Choose Best Research Methodology for Your Study
Successful research conduction requires proper planning and execution. While there are multiple reasons and aspects…

Top 5 Key Differences Between Methods and Methodology
While burning the midnight oil during literature review, most researchers do not realize that the…
How to Draft the Acknowledgment Section of a Manuscript
Discussion Vs. Conclusion: Know the Difference Before Drafting Manuscripts

Sign-up to read more
Subscribe for free to get unrestricted access to all our resources on research writing and academic publishing including:
- 2000+ blog articles
- 50+ Webinars
- 10+ Expert podcasts
- 50+ Infographics
- 10+ Checklists
- Research Guides
We hate spam too. We promise to protect your privacy and never spam you.
I am looking for Editing/ Proofreading services for my manuscript Tentative date of next journal submission:

What would be most effective in reducing research misconduct?
Some (Relatively) Recent Examples of Participant Observation Studies
Table of Contents
Last Updated on August 11, 2020 by
Participant observation is one the main research methods on the A level sociology syllabus, but many of the examples in the main text books are painfully out of date. This post provides some more recent examples of research studies which employed participant observation as their main research method.
Covert Participant Observation
Pearson’s (2009) covert participant observation study of blackpool football club’s supporters.
Pearson carried out covert participant observation of supporters of Blackpool Football Club between 1995 and 1998. He was known to other supporters as a student pursuing a degree in law, but his status as an academic researcher was unknown to them. His approach was to meet up with them in the pub before a match or sometimes on entering the stadium, and to meet up with them afterwards for a drink. He attended seventy-eight matches but notes that because he did not live in the area, he was unable to observe the supporters outside of a football context.
He chose Blackpool F.C. because it was close to Lancaster, where he was a student, and because of its reputation as having problems with football hooliganism. He seems to have been able to gradually insinuate himself into the supporters’ world by being recognised as a regular fan. Pearson played up his knowledge of the game and the club and was able to integrate himself into their world.
Pearson says of his research…’ whilst it was possible to avoid committing some individual offences, a refusal to commit crimes on a regular basis would have aroused suspicions and reduced research opportunities. As a result I committed ‘minor’ offences (which I tentatively defined as those would not cause direct physical harm to a research subject). My strategy was to commit only the offences which the majority of the research subjects were committing and that I considered necessary to carry out the research. Furthermore, whilst I would commit lesser offences with regularity, I would, if possible, avoid more serious ones.’ (Pearson, 2009).
You can read an interview with Dr Geoff Pearson here .
Overt Participant Observation
Khan’s (2011, 2014) ethnography of an elite high school in the united states.
The majority of ethnographic work seems to have been carried out with (on?) the poor and the marginalised, Khan’s work provides us with a rare ethnographic study of an elite institution.
Khan says: ‘ethnography is a method wherein the scholar embeds himself in the relations under study, spending long periods of time with research subjects. For me, it meant getting a job at St. Paul’s School… I moved into an apartment on campus, and… observed the daily life of the school. After my years at St. Paul’s I returned many times, and I sought out alumni to interview and discuss some of the things I’d learned (Khan 2014).
In contrast to Pearson’s research, this ethnography by Khan illustrates one of the main advantages overt participant observation has over covert: you can carry on collecting data from the respondents afterwards!
Mears’s (2011) ethnography of the world of the fashion model

In contrast to Khan’s research, Mears explicitly puts the observation before the participation, which suggests she is less immersed in the day to day life of her group than Kahn was.
Sampson’s (2013) ethnographic research on international seafarers
This final example is what Bryman refers to as a ‘participating’ observer’ rather than a ‘full member’ – Sampson is working for the shipping company with the men on a very temporary basis.
Anna Lora-Wainwright (2018) Resigned Activism – Living with Pollution in Rural China
Lora-Wainwright spent from 2009-2013 studying how people in rural China cope when they know severe pollution is having a severely detrimental effect on their health.
Lora-Wainwright focused on how people responded when they knew they were being subjected to a significant cancer risk from pollution – how they organised and protested, but also how they just coped on a day to day basis -living with things such as polluted water that’s going to give you cancer if you drink it.
Share this:
Leave a reply cancel reply.
This site uses Akismet to reduce spam. Learn how your comment data is processed .
Discover more from ReviseSociology
The Advantages and Limitations of Single Case Study Analysis

As Andrew Bennett and Colin Elman have recently noted, qualitative research methods presently enjoy “an almost unprecedented popularity and vitality… in the international relations sub-field”, such that they are now “indisputably prominent, if not pre-eminent” (2010: 499). This is, they suggest, due in no small part to the considerable advantages that case study methods in particular have to offer in studying the “complex and relatively unstructured and infrequent phenomena that lie at the heart of the subfield” (Bennett and Elman, 2007: 171). Using selected examples from within the International Relations literature[1], this paper aims to provide a brief overview of the main principles and distinctive advantages and limitations of single case study analysis. Divided into three inter-related sections, the paper therefore begins by first identifying the underlying principles that serve to constitute the case study as a particular research strategy, noting the somewhat contested nature of the approach in ontological, epistemological, and methodological terms. The second part then looks to the principal single case study types and their associated advantages, including those from within the recent ‘third generation’ of qualitative International Relations (IR) research. The final section of the paper then discusses the most commonly articulated limitations of single case studies; while accepting their susceptibility to criticism, it is however suggested that such weaknesses are somewhat exaggerated. The paper concludes that single case study analysis has a great deal to offer as a means of both understanding and explaining contemporary international relations.
The term ‘case study’, John Gerring has suggested, is “a definitional morass… Evidently, researchers have many different things in mind when they talk about case study research” (2006a: 17). It is possible, however, to distil some of the more commonly-agreed principles. One of the most prominent advocates of case study research, Robert Yin (2009: 14) defines it as “an empirical enquiry that investigates a contemporary phenomenon in depth and within its real-life context, especially when the boundaries between phenomenon and context are not clearly evident”. What this definition usefully captures is that case studies are intended – unlike more superficial and generalising methods – to provide a level of detail and understanding, similar to the ethnographer Clifford Geertz’s (1973) notion of ‘thick description’, that allows for the thorough analysis of the complex and particularistic nature of distinct phenomena. Another frequently cited proponent of the approach, Robert Stake, notes that as a form of research the case study “is defined by interest in an individual case, not by the methods of inquiry used”, and that “the object of study is a specific, unique, bounded system” (2008: 443, 445). As such, three key points can be derived from this – respectively concerning issues of ontology, epistemology, and methodology – that are central to the principles of single case study research.
First, the vital notion of ‘boundedness’ when it comes to the particular unit of analysis means that defining principles should incorporate both the synchronic (spatial) and diachronic (temporal) elements of any so-called ‘case’. As Gerring puts it, a case study should be “an intensive study of a single unit… a spatially bounded phenomenon – e.g. a nation-state, revolution, political party, election, or person – observed at a single point in time or over some delimited period of time” (2004: 342). It is important to note, however, that – whereas Gerring refers to a single unit of analysis – it may be that attention also necessarily be given to particular sub-units. This points to the important difference between what Yin refers to as an ‘holistic’ case design, with a single unit of analysis, and an ’embedded’ case design with multiple units of analysis (Yin, 2009: 50-52). The former, for example, would examine only the overall nature of an international organization, whereas the latter would also look to specific departments, programmes, or policies etc.
Secondly, as Tim May notes of the case study approach, “even the most fervent advocates acknowledge that the term has entered into understandings with little specification or discussion of purpose and process” (2011: 220). One of the principal reasons for this, he argues, is the relationship between the use of case studies in social research and the differing epistemological traditions – positivist, interpretivist, and others – within which it has been utilised. Philosophy of science concerns are obviously a complex issue, and beyond the scope of much of this paper. That said, the issue of how it is that we know what we know – of whether or not a single independent reality exists of which we as researchers can seek to provide explanation – does lead us to an important distinction to be made between so-called idiographic and nomothetic case studies (Gerring, 2006b). The former refers to those which purport to explain only a single case, are concerned with particularisation, and hence are typically (although not exclusively) associated with more interpretivist approaches. The latter are those focused studies that reflect upon a larger population and are more concerned with generalisation, as is often so with more positivist approaches[2]. The importance of this distinction, and its relation to the advantages and limitations of single case study analysis, is returned to below.
Thirdly, in methodological terms, given that the case study has often been seen as more of an interpretivist and idiographic tool, it has also been associated with a distinctly qualitative approach (Bryman, 2009: 67-68). However, as Yin notes, case studies can – like all forms of social science research – be exploratory, descriptive, and/or explanatory in nature. It is “a common misconception”, he notes, “that the various research methods should be arrayed hierarchically… many social scientists still deeply believe that case studies are only appropriate for the exploratory phase of an investigation” (Yin, 2009: 6). If case studies can reliably perform any or all three of these roles – and given that their in-depth approach may also require multiple sources of data and the within-case triangulation of methods – then it becomes readily apparent that they should not be limited to only one research paradigm. Exploratory and descriptive studies usually tend toward the qualitative and inductive, whereas explanatory studies are more often quantitative and deductive (David and Sutton, 2011: 165-166). As such, the association of case study analysis with a qualitative approach is a “methodological affinity, not a definitional requirement” (Gerring, 2006a: 36). It is perhaps better to think of case studies as transparadigmatic; it is mistaken to assume single case study analysis to adhere exclusively to a qualitative methodology (or an interpretivist epistemology) even if it – or rather, practitioners of it – may be so inclined. By extension, this also implies that single case study analysis therefore remains an option for a multitude of IR theories and issue areas; it is how this can be put to researchers’ advantage that is the subject of the next section.
Having elucidated the defining principles of the single case study approach, the paper now turns to an overview of its main benefits. As noted above, a lack of consensus still exists within the wider social science literature on the principles and purposes – and by extension the advantages and limitations – of case study research. Given that this paper is directed towards the particular sub-field of International Relations, it suggests Bennett and Elman’s (2010) more discipline-specific understanding of contemporary case study methods as an analytical framework. It begins however, by discussing Harry Eckstein’s seminal (1975) contribution to the potential advantages of the case study approach within the wider social sciences.
Eckstein proposed a taxonomy which usefully identified what he considered to be the five most relevant types of case study. Firstly were so-called configurative-idiographic studies, distinctly interpretivist in orientation and predicated on the assumption that “one cannot attain prediction and control in the natural science sense, but only understanding ( verstehen )… subjective values and modes of cognition are crucial” (1975: 132). Eckstein’s own sceptical view was that any interpreter ‘simply’ considers a body of observations that are not self-explanatory and “without hard rules of interpretation, may discern in them any number of patterns that are more or less equally plausible” (1975: 134). Those of a more post-modernist bent, of course – sharing an “incredulity towards meta-narratives”, in Lyotard’s (1994: xxiv) evocative phrase – would instead suggest that this more free-form approach actually be advantageous in delving into the subtleties and particularities of individual cases.
Eckstein’s four other types of case study, meanwhile, promote a more nomothetic (and positivist) usage. As described, disciplined-configurative studies were essentially about the use of pre-existing general theories, with a case acting “passively, in the main, as a receptacle for putting theories to work” (Eckstein, 1975: 136). As opposed to the opportunity this presented primarily for theory application, Eckstein identified heuristic case studies as explicit theoretical stimulants – thus having instead the intended advantage of theory-building. So-called p lausibility probes entailed preliminary attempts to determine whether initial hypotheses should be considered sound enough to warrant more rigorous and extensive testing. Finally, and perhaps most notably, Eckstein then outlined the idea of crucial case studies , within which he also included the idea of ‘most-likely’ and ‘least-likely’ cases; the essential characteristic of crucial cases being their specific theory-testing function.
Whilst Eckstein’s was an early contribution to refining the case study approach, Yin’s (2009: 47-52) more recent delineation of possible single case designs similarly assigns them roles in the applying, testing, or building of theory, as well as in the study of unique cases[3]. As a subset of the latter, however, Jack Levy (2008) notes that the advantages of idiographic cases are actually twofold. Firstly, as inductive/descriptive cases – akin to Eckstein’s configurative-idiographic cases – whereby they are highly descriptive, lacking in an explicit theoretical framework and therefore taking the form of “total history”. Secondly, they can operate as theory-guided case studies, but ones that seek only to explain or interpret a single historical episode rather than generalise beyond the case. Not only does this therefore incorporate ‘single-outcome’ studies concerned with establishing causal inference (Gerring, 2006b), it also provides room for the more postmodern approaches within IR theory, such as discourse analysis, that may have developed a distinct methodology but do not seek traditional social scientific forms of explanation.
Applying specifically to the state of the field in contemporary IR, Bennett and Elman identify a ‘third generation’ of mainstream qualitative scholars – rooted in a pragmatic scientific realist epistemology and advocating a pluralistic approach to methodology – that have, over the last fifteen years, “revised or added to essentially every aspect of traditional case study research methods” (2010: 502). They identify ‘process tracing’ as having emerged from this as a central method of within-case analysis. As Bennett and Checkel observe, this carries the advantage of offering a methodologically rigorous “analysis of evidence on processes, sequences, and conjunctures of events within a case, for the purposes of either developing or testing hypotheses about causal mechanisms that might causally explain the case” (2012: 10).
Harnessing various methods, process tracing may entail the inductive use of evidence from within a case to develop explanatory hypotheses, and deductive examination of the observable implications of hypothesised causal mechanisms to test their explanatory capability[4]. It involves providing not only a coherent explanation of the key sequential steps in a hypothesised process, but also sensitivity to alternative explanations as well as potential biases in the available evidence (Bennett and Elman 2010: 503-504). John Owen (1994), for example, demonstrates the advantages of process tracing in analysing whether the causal factors underpinning democratic peace theory are – as liberalism suggests – not epiphenomenal, but variously normative, institutional, or some given combination of the two or other unexplained mechanism inherent to liberal states. Within-case process tracing has also been identified as advantageous in addressing the complexity of path-dependent explanations and critical junctures – as for example with the development of political regime types – and their constituent elements of causal possibility, contingency, closure, and constraint (Bennett and Elman, 2006b).
Bennett and Elman (2010: 505-506) also identify the advantages of single case studies that are implicitly comparative: deviant, most-likely, least-likely, and crucial cases. Of these, so-called deviant cases are those whose outcome does not fit with prior theoretical expectations or wider empirical patterns – again, the use of inductive process tracing has the advantage of potentially generating new hypotheses from these, either particular to that individual case or potentially generalisable to a broader population. A classic example here is that of post-independence India as an outlier to the standard modernisation theory of democratisation, which holds that higher levels of socio-economic development are typically required for the transition to, and consolidation of, democratic rule (Lipset, 1959; Diamond, 1992). Absent these factors, MacMillan’s single case study analysis (2008) suggests the particularistic importance of the British colonial heritage, the ideology and leadership of the Indian National Congress, and the size and heterogeneity of the federal state.
Most-likely cases, as per Eckstein above, are those in which a theory is to be considered likely to provide a good explanation if it is to have any application at all, whereas least-likely cases are ‘tough test’ ones in which the posited theory is unlikely to provide good explanation (Bennett and Elman, 2010: 505). Levy (2008) neatly refers to the inferential logic of the least-likely case as the ‘Sinatra inference’ – if a theory can make it here, it can make it anywhere. Conversely, if a theory cannot pass a most-likely case, it is seriously impugned. Single case analysis can therefore be valuable for the testing of theoretical propositions, provided that predictions are relatively precise and measurement error is low (Levy, 2008: 12-13). As Gerring rightly observes of this potential for falsification:
“a positivist orientation toward the work of social science militates toward a greater appreciation of the case study format, not a denigration of that format, as is usually supposed” (Gerring, 2007: 247, emphasis added).
In summary, the various forms of single case study analysis can – through the application of multiple qualitative and/or quantitative research methods – provide a nuanced, empirically-rich, holistic account of specific phenomena. This may be particularly appropriate for those phenomena that are simply less amenable to more superficial measures and tests (or indeed any substantive form of quantification) as well as those for which our reasons for understanding and/or explaining them are irreducibly subjective – as, for example, with many of the normative and ethical issues associated with the practice of international relations. From various epistemological and analytical standpoints, single case study analysis can incorporate both idiographic sui generis cases and, where the potential for generalisation may exist, nomothetic case studies suitable for the testing and building of causal hypotheses. Finally, it should not be ignored that a signal advantage of the case study – with particular relevance to international relations – also exists at a more practical rather than theoretical level. This is, as Eckstein noted, “that it is economical for all resources: money, manpower, time, effort… especially important, of course, if studies are inherently costly, as they are if units are complex collective individuals ” (1975: 149-150, emphasis added).
Limitations
Single case study analysis has, however, been subject to a number of criticisms, the most common of which concern the inter-related issues of methodological rigour, researcher subjectivity, and external validity. With regard to the first point, the prototypical view here is that of Zeev Maoz (2002: 164-165), who suggests that “the use of the case study absolves the author from any kind of methodological considerations. Case studies have become in many cases a synonym for freeform research where anything goes”. The absence of systematic procedures for case study research is something that Yin (2009: 14-15) sees as traditionally the greatest concern due to a relative absence of methodological guidelines. As the previous section suggests, this critique seems somewhat unfair; many contemporary case study practitioners – and representing various strands of IR theory – have increasingly sought to clarify and develop their methodological techniques and epistemological grounding (Bennett and Elman, 2010: 499-500).
A second issue, again also incorporating issues of construct validity, concerns that of the reliability and replicability of various forms of single case study analysis. This is usually tied to a broader critique of qualitative research methods as a whole. However, whereas the latter obviously tend toward an explicitly-acknowledged interpretive basis for meanings, reasons, and understandings:
“quantitative measures appear objective, but only so long as we don’t ask questions about where and how the data were produced… pure objectivity is not a meaningful concept if the goal is to measure intangibles [as] these concepts only exist because we can interpret them” (Berg and Lune, 2010: 340).
The question of researcher subjectivity is a valid one, and it may be intended only as a methodological critique of what are obviously less formalised and researcher-independent methods (Verschuren, 2003). Owen (1994) and Layne’s (1994) contradictory process tracing results of interdemocratic war-avoidance during the Anglo-American crisis of 1861 to 1863 – from liberal and realist standpoints respectively – are a useful example. However, it does also rest on certain assumptions that can raise deeper and potentially irreconcilable ontological and epistemological issues. There are, regardless, plenty such as Bent Flyvbjerg (2006: 237) who suggest that the case study contains no greater bias toward verification than other methods of inquiry, and that “on the contrary, experience indicates that the case study contains a greater bias toward falsification of preconceived notions than toward verification”.
The third and arguably most prominent critique of single case study analysis is the issue of external validity or generalisability. How is it that one case can reliably offer anything beyond the particular? “We always do better (or, in the extreme, no worse) with more observation as the basis of our generalization”, as King et al write; “in all social science research and all prediction, it is important that we be as explicit as possible about the degree of uncertainty that accompanies out prediction” (1994: 212). This is an unavoidably valid criticism. It may be that theories which pass a single crucial case study test, for example, require rare antecedent conditions and therefore actually have little explanatory range. These conditions may emerge more clearly, as Van Evera (1997: 51-54) notes, from large-N studies in which cases that lack them present themselves as outliers exhibiting a theory’s cause but without its predicted outcome. As with the case of Indian democratisation above, it would logically be preferable to conduct large-N analysis beforehand to identify that state’s non-representative nature in relation to the broader population.
There are, however, three important qualifiers to the argument about generalisation that deserve particular mention here. The first is that with regard to an idiographic single-outcome case study, as Eckstein notes, the criticism is “mitigated by the fact that its capability to do so [is] never claimed by its exponents; in fact it is often explicitly repudiated” (1975: 134). Criticism of generalisability is of little relevance when the intention is one of particularisation. A second qualifier relates to the difference between statistical and analytical generalisation; single case studies are clearly less appropriate for the former but arguably retain significant utility for the latter – the difference also between explanatory and exploratory, or theory-testing and theory-building, as discussed above. As Gerring puts it, “theory confirmation/disconfirmation is not the case study’s strong suit” (2004: 350). A third qualification relates to the issue of case selection. As Seawright and Gerring (2008) note, the generalisability of case studies can be increased by the strategic selection of cases. Representative or random samples may not be the most appropriate, given that they may not provide the richest insight (or indeed, that a random and unknown deviant case may appear). Instead, and properly used , atypical or extreme cases “often reveal more information because they activate more actors… and more basic mechanisms in the situation studied” (Flyvbjerg, 2006). Of course, this also points to the very serious limitation, as hinted at with the case of India above, that poor case selection may alternatively lead to overgeneralisation and/or grievous misunderstandings of the relationship between variables or processes (Bennett and Elman, 2006a: 460-463).
As Tim May (2011: 226) notes, “the goal for many proponents of case studies […] is to overcome dichotomies between generalizing and particularizing, quantitative and qualitative, deductive and inductive techniques”. Research aims should drive methodological choices, rather than narrow and dogmatic preconceived approaches. As demonstrated above, there are various advantages to both idiographic and nomothetic single case study analyses – notably the empirically-rich, context-specific, holistic accounts that they have to offer, and their contribution to theory-building and, to a lesser extent, that of theory-testing. Furthermore, while they do possess clear limitations, any research method involves necessary trade-offs; the inherent weaknesses of any one method, however, can potentially be offset by situating them within a broader, pluralistic mixed-method research strategy. Whether or not single case studies are used in this fashion, they clearly have a great deal to offer.
References
Bennett, A. and Checkel, J. T. (2012) ‘Process Tracing: From Philosophical Roots to Best Practice’, Simons Papers in Security and Development, No. 21/2012, School for International Studies, Simon Fraser University: Vancouver.
Bennett, A. and Elman, C. (2006a) ‘Qualitative Research: Recent Developments in Case Study Methods’, Annual Review of Political Science , 9, 455-476.
Bennett, A. and Elman, C. (2006b) ‘Complex Causal Relations and Case Study Methods: The Example of Path Dependence’, Political Analysis , 14, 3, 250-267.
Bennett, A. and Elman, C. (2007) ‘Case Study Methods in the International Relations Subfield’, Comparative Political Studies , 40, 2, 170-195.
Bennett, A. and Elman, C. (2010) Case Study Methods. In C. Reus-Smit and D. Snidal (eds) The Oxford Handbook of International Relations . Oxford University Press: Oxford. Ch. 29.
Berg, B. and Lune, H. (2012) Qualitative Research Methods for the Social Sciences . Pearson: London.
Bryman, A. (2012) Social Research Methods . Oxford University Press: Oxford.
David, M. and Sutton, C. D. (2011) Social Research: An Introduction . SAGE Publications Ltd: London.
Diamond, J. (1992) ‘Economic development and democracy reconsidered’, American Behavioral Scientist , 35, 4/5, 450-499.
Eckstein, H. (1975) Case Study and Theory in Political Science. In R. Gomm, M. Hammersley, and P. Foster (eds) Case Study Method . SAGE Publications Ltd: London.
Flyvbjerg, B. (2006) ‘Five Misunderstandings About Case-Study Research’, Qualitative Inquiry , 12, 2, 219-245.
Geertz, C. (1973) The Interpretation of Cultures: Selected Essays by Clifford Geertz . Basic Books Inc: New York.
Gerring, J. (2004) ‘What is a Case Study and What Is It Good for?’, American Political Science Review , 98, 2, 341-354.
Gerring, J. (2006a) Case Study Research: Principles and Practices . Cambridge University Press: Cambridge.
Gerring, J. (2006b) ‘Single-Outcome Studies: A Methodological Primer’, International Sociology , 21, 5, 707-734.
Gerring, J. (2007) ‘Is There a (Viable) Crucial-Case Method?’, Comparative Political Studies , 40, 3, 231-253.
King, G., Keohane, R. O. and Verba, S. (1994) Designing Social Inquiry: Scientific Inference in Qualitative Research . Princeton University Press: Chichester.
Layne, C. (1994) ‘Kant or Cant: The Myth of the Democratic Peace’, International Security , 19, 2, 5-49.
Levy, J. S. (2008) ‘Case Studies: Types, Designs, and Logics of Inference’, Conflict Management and Peace Science , 25, 1-18.
Lipset, S. M. (1959) ‘Some Social Requisites of Democracy: Economic Development and Political Legitimacy’, The American Political Science Review , 53, 1, 69-105.
Lyotard, J-F. (1984) The Postmodern Condition: A Report on Knowledge . University of Minnesota Press: Minneapolis.
MacMillan, A. (2008) ‘Deviant Democratization in India’, Democratization , 15, 4, 733-749.
Maoz, Z. (2002) Case study methodology in international studies: from storytelling to hypothesis testing. In F. P. Harvey and M. Brecher (eds) Evaluating Methodology in International Studies . University of Michigan Press: Ann Arbor.
May, T. (2011) Social Research: Issues, Methods and Process . Open University Press: Maidenhead.
Owen, J. M. (1994) ‘How Liberalism Produces Democratic Peace’, International Security , 19, 2, 87-125.
Seawright, J. and Gerring, J. (2008) ‘Case Selection Techniques in Case Study Research: A Menu of Qualitative and Quantitative Options’, Political Research Quarterly , 61, 2, 294-308.
Stake, R. E. (2008) Qualitative Case Studies. In N. K. Denzin and Y. S. Lincoln (eds) Strategies of Qualitative Inquiry . Sage Publications: Los Angeles. Ch. 17.
Van Evera, S. (1997) Guide to Methods for Students of Political Science . Cornell University Press: Ithaca.
Verschuren, P. J. M. (2003) ‘Case study as a research strategy: some ambiguities and opportunities’, International Journal of Social Research Methodology , 6, 2, 121-139.
Yin, R. K. (2009) Case Study Research: Design and Methods . SAGE Publications Ltd: London.
[1] The paper follows convention by differentiating between ‘International Relations’ as the academic discipline and ‘international relations’ as the subject of study.
[2] There is some similarity here with Stake’s (2008: 445-447) notion of intrinsic cases, those undertaken for a better understanding of the particular case, and instrumental ones that provide insight for the purposes of a wider external interest.
[3] These may be unique in the idiographic sense, or in nomothetic terms as an exception to the generalising suppositions of either probabilistic or deterministic theories (as per deviant cases, below).
[4] Although there are “philosophical hurdles to mount”, according to Bennett and Checkel, there exists no a priori reason as to why process tracing (as typically grounded in scientific realism) is fundamentally incompatible with various strands of positivism or interpretivism (2012: 18-19). By extension, it can therefore be incorporated by a range of contemporary mainstream IR theories.
— Written by: Ben Willis Written at: University of Plymouth Written for: David Brockington Date written: January 2013
Further Reading on E-International Relations
- Identity in International Conflicts: A Case Study of the Cuban Missile Crisis
- Imperialism’s Legacy in the Study of Contemporary Politics: The Case of Hegemonic Stability Theory
- Recreating a Nation’s Identity Through Symbolism: A Chinese Case Study
- Ontological Insecurity: A Case Study on Israeli-Palestinian Conflict in Jerusalem
- Terrorists or Freedom Fighters: A Case Study of ETA
- A Critical Assessment of Eco-Marxism: A Ghanaian Case Study
Please Consider Donating
Before you download your free e-book, please consider donating to support open access publishing.
E-IR is an independent non-profit publisher run by an all volunteer team. Your donations allow us to invest in new open access titles and pay our bandwidth bills to ensure we keep our existing titles free to view. Any amount, in any currency, is appreciated. Many thanks!
Donations are voluntary and not required to download the e-book - your link to download is below.
- Subscribe to journal Subscribe
- Get new issue alerts Get alerts
Secondary Logo
Journal logo.
Colleague's E-mail is Invalid
Your message has been successfully sent to your colleague.
Save my selection
Selection of the Study Participants
Capili, Bernadette PhD, NP-C
Bernadette Capili is director of the Heilbrunn Family Center for Research Nursing at Rockefeller University, New York City. This manuscript was supported in part by grant No. UL1TR001866 from the National Institutes of Health's National Center for Advancing Translational Sciences Clinical and Translational Science Awards Program. Contact author: [email protected] . The author has disclosed no potential conflicts of interest, financial or otherwise. A podcast with the author is available at www.ajnonline.com .

Editor's note: This is the second article in a new series on clinical research by nurses. The series is designed to give nurses the knowledge and skills they need to participate in research, step by step. Each column will present the concepts that underpin evidence-based practice—from research design to data interpretation. The articles will also be accompanied by a podcast offering more insight and context from the author.
The first article in this series (“How Does Research Start?” October 2020) was about how to identify a topic of interest and develop a good research question. This article is an introduction to designing study eligibility criteria and recruiting study participants.
Defining the target population is an essential part of protocol development because it ensures the study participants are well suited to the research question. The target population is the group of people who share a common condition (disease process) or characteristic the researcher is interested in studying. The accessible population is the geographically and temporally classified subset who are available for study participant recruitment. The clinical and demographic characteristics of the accessible population are established using predetermined eligibility criteria. The choice of study participants also involves factors related to feasibility, including the time and money needed to obtain the required number of participants.
DESIGNING THE ELIGIBILITY CRITERIA
All clinical research protocols must have an eligibility criteria section that includes both inclusion criteria and exclusion criteria . Common elements that determine suitability for participation in a study are age, sex, medical condition, specific disease examined, and medications taken. The development of eligibility criteria must also include a review of the potential safety concerns of an intervention, such as interference with a participant's current medication or concurrent health condition. The eligibility criteria should be designed to select a representative sample (the accessible population) from the target population. From this smaller sample, researchers can generalize findings to the larger target population in order to improve understanding of disease processes, new treatment options, and sociodemographic trends.
To illustrate the development of the eligibility criteria, see Table 1 . 1 Discussed below is the rationale for the selection of the inclusion and exclusion criteria.
| Criteria | Design Feature | Example |
|---|---|---|
| Inclusion criteria | Specifying populations relevant to the research question and efficient for study: | |
| Demographic characteristics | Men and women 18-60 years | |
| Clinical characteristics | HIV positive; fasting serum triglyceride level, 150-500 mg/dL; CD4+ cell count, ≥ 300 cells/mm ; body mass index, 20-29 kg/m | |
| Geographic characteristics | Lives in the New York City area | |
| Temporal characteristics | On a stable HIV drug regimen for a minimum of six months | |
| Able to provide verbal and written consent | Able and willing to sign informed consent | |
| Exclusion criteria | Specifying subsets of the population that will not be studied because of: | |
| A high likelihood of loss to follow-up | Active user of illicit drugs and alcohol | |
| An inability to provide good data | Scores less than an established cut-off point on the Mini-Mental State Examination | |
| At high risk for possible adverse effects | Has diabetes mellitus, familial hyperlipidemia, or opportunistic infections; uses fish oil supplements |
Inclusion criteria considerations . The inclusion criteria describe potential study participants who can best provide an answer to the clinical research question. Common elements of the inclusion criteria include demographic characteristics, clinical attributes, geographic factors, and temporal considerations. Defining the inclusion criteria can help to maintain a degree of homogeneity among the accessible population and narrow the available pool of eligible study participants. The inclusion criteria should also address the capacity of the participant to understand the study goals, the study requirements (such as completing questionnaires and taking study medications), and the potential risks associated with joining a study.
For example, as shown in the table, the inclusion criteria for the study on the efficacy of fish oil in lowering triglyceride levels in adults with HIV contain an age limitation, since advanced age is associated with an accelerated loss of CD4+ cells and fish oil can reduce CD4+ cell count. 1 A triglyceride parameter (fasting serum triglycerides between 150 and 500 mg/dL) is also required to capture participants with high triglyceride levels, yet such a range necessarily leaves out those with extremely high triglycerides who may otherwise be eligible for prescription fish oil treatment. Additionally, a parameter for participants' CD4+ cell count is necessary to ensure the participants' immune systems are stable.
The possibility that participants will be able to travel easily to the study site is another vital consideration: geographic limitations are essential if virtual study visits are not possible and face-to-face interactions are necessary. An example of a temporal study criterion is the requirement that a participant wait a minimum of six months or more after starting a new HIV drug regimen. The waiting period reduces the likelihood that an elevated triglyceride level is a temporary drug side effect that will return to normal once the proper dosage of the new HIV drug is achieved.
Finally, the informed consent process is also among the inclusion criteria. A future article in this series will discuss the ethics of patient participation, including such issues as the potential for coercion or undue influence and the ability of the study participant to provide verbal and written consent.
Exclusion criteria considerations . The exclusion criteria describe potential study participants who meet the inclusion criteria but have characteristics that could affect the successful outcome of the study or lead to an increased risk of adverse effects. In this way, the exclusion criteria act as a mechanism to reduce potential study risks and confounding variables. 2 Confounding variables may affect the outcome of interest (the dependent variable). For instance, in the example study, the triglyceride level is the dependent variable; since the study is examining the effect of fish oil on reducing triglycerides, potential participants already using fish oil are not eligible to join.
In formulating the exclusion criteria, it's important to consider factors that would increase the likelihood of participants' loss to follow-up or inability to provide necessary data. For instance, participants actively abusing alcohol and illicit drugs were not eligible to join the HIV and high triglycerides study because substance abuse could have limited a participant's ability to comprehend and comply with study requirements. The use of a screening tool such as the Mini-Mental State Examination, which measures attention, recall, and calculation, can be used to exclude participants who score below a certain threshold (in the example study, the threshold was 24). 1, 3 Screening for cognitive impairment in this way may increase the likelihood of obtaining the required data. It's also important to anticipate the high risk of adverse effects related to comorbidities or current treatments. Participants with diabetes mellitus were also excluded from the example study, as fish oil can raise blood glucose levels and may not be safe for some participants.
Practical considerations for designing the eligibility criteria . When formulating the eligibility criteria, it can be helpful for prospective authors of a new clinical research study to review published studies with similar study participants and objectives. Examine the approaches used in such investigations. Assess the data on the retention of the participants and evaluate the reasons for participant loss to follow-up. Much can be learned about the success or potential flaws of study methods from the experiences of researchers in previously conducted studies.
Defining the eligibility criteria requires a delicate balance between restrictive and less restrictive. A restrictive criterion can make participant recruitment and enrollment more difficult by reducing the eligible participant pool. Additionally, a restrictive criterion might increase the time frame required for study completion, as well as the workforce needed to recruit and enroll study participants. On the other hand, a less stringent criterion may make it easier to recruit and enroll participants, which can increase the generalizability of a study's findings; however, it can also result in more confounding variables and an overly heterogeneous participant pool.
Obtaining IRB approval . During the development of the eligibility criteria, review the Code of Federal Regulations Title 45, Part 46 (45 CFR 46), which focuses on the protection of human subjects. 4 Since institutional review board (IRB) approval is mandatory for all clinical research studies, IRBs will assess study protocols to ensure they meet the 45 CFR 46 requirements. The focus of 45 CFR 46 is on the unbiased selection of study participants centered around sex, race, ethnicity, religion, nationality, and other factors. It stipulates that all eligible participants should be considered unless there is a compelling safety or scientific rationale against it. For example, a prostate cancer study can reasonably exclude women, since the disease is specific to men. In another example, a case to exclude pregnant women could be made for studies with known risks to maternal and fetal health.
In writing the eligibility criteria, use bullets or a numbered list. This format makes it easier for IRB review and implementation. A printed (or an electronic) version of the eligibility criteria placed near the telephone, computer, or participant screening area can provide study team members with a copy they can easily review to reduce the chances of enrolling ineligible participants.
STUDY PARTICIPANT RECRUITMENT
Participant recruitment is one of the most challenging aspects of conducting clinical research. It's estimated that a lack of enrollment accounts for delays or cancellations of up to 60% of clinical trials 5 ; therefore, a well-thought-out and feasible recruitment plan is vital.
The study protocol provides details of the procedures the research team will follow to recruit and enroll study participants. In designing the recruitment plan, consider the anticipated participant accrual rate—the number of participants per month or year who are enrolled and meet all study requirements—and how long it will take to complete the recruitment goals. Describing the specifics of how and where to obtain eligible study participants is an essential component of the recruitment plan. The researchers must specify the methods that will be used to find and approach potential study participants and the setting in which they plan to do so, such as a clinic or a research site. For example, in recruiting the study of HIV-positive adults with high triglyceride levels, flyers and brochures were developed and sent to local HIV clinics in the New York City area. 1 Potential participants then called the research center to inquire about the study; in this way, the participants initiated the recruitment process.
Recruitment materials should include the title of the protocol, IRB study number, sponsoring institution, contact information, and purpose of the research (in our example, the purpose was to investigate the use of fish oil as a treatment for HIV-positive adults with high triglyceride levels), and indicate whether study participants will receive compensation for their time and effort. 6 All methods to advertise or promote the study require IRB approval before implementation. Some examples of recruitment materials include the telephone script used to discuss the study with potential participants, as well as flyers, posters, postcards, newspaper advertisements, press releases, website advertisements, electronic mailings, and social media posts. 5, 6 The IRB may require specific information about recruitment materials before granting approval. Most institutions publish their IRB requirements online; to ensure a smoother review process, read the guidelines associated with each IRB institution.
IMPLEMENTING A SUCCESSFUL STUDY
The development of the eligibility criteria is a complex process that can affect participant recruitment, participant enrollment, and study completion. Reviewing the literature for research studies with populations similar to the proposed target population of a study is an excellent way to identify appropriate eligibility criteria. Designing a feasible recruitment plan that includes the selection of a suitable participant pool and developing approaches to recruiting and enrolling eligible participants is critical to implementing a successful study. The next column will discuss sampling design, including probability and nonprobability sampling methods.
- Cited Here |
- Google Scholar
- + Favorites
- View in Gallery
Readers Of this Article Also Read
Sampling design in nursing research, measurement in nursing research, the architecture of a research study, selection and implementation of outcome measurements, interpretive methodologies in qualitative nursing research.

- Request new password
- Create a new account
Social Research Methods: Qualitative, Quantitative and Mixed Methods Approaches
Student resources, case studies.
Participant and Nonparticipant Observation: A Study of Instructional Support Liaisons
The case presents an example of a research project in which the researchers struggled to negotiate participant and nonparticipant observation roles. The project and the data collection efforts including mistakes and eventual solutions are described. The case presents the benefits and challenges of engaging in participant and nonparticipant observation, specifically for those in the field of education. Finally, this case highlights the importance of thoughtfully selecting one’s role in observational data collection.
1: What are the differences between participant and nonparticipant observation?
2: Why is it important to reflect on the researchers’ roles in participant observation?
Developing and Executing a Data Collection Plan for Archival Research
This case details the process of planning and carrying out archival research on municipal associations, and it discusses the challenges and opportunities that arise from researching an understudied field, including the need to identify, locate, and collect primary data. The case discusses the process of developing a Data Collecting Plan and executing a plan of archival research to conduct original, empirical analysis. By carrying out this process, the researcher found and developed the data needed to carry out mixed-methods research, and to analyse the relationship between municipal associations’ memberships and their behaviour in intergovernmental relations.
1: Why is it useful to develop a data collection plan in archival research?
2: How can documents from archives be used in mixed methods research?
Importance of Adapting to Unexpected Circumstances in Qualitative Data Collection
This case is a reflection on a qualitative research project based on focus groups for evaluating the effectiveness of a sports centre for canoe/kayak in Oklahoma City in the US. The purpose of the project was to make sure that what the centre was doing was actually working. The case highlights the importance of being prepared, anticipating problems, and being flexible enough to overcome unforeseen circumstances.
1: Why is proper planning necessary in qualitative data collection?
2: What are the strengths and limitations of using focus group in this kind of evaluation research?
A Mixed-Method Design for Developing a Measure of Entrepreneurial Openness
This mixed methods case explains the development of the concept and measurement scale of entrepreneurial openness – a personality characteristic that helps to understand the impact of an entrepreneur’s personality on a small firm’s performance. The case describes several procedures that were applied in the scale development process: interdisciplinary literature review, interviews, focus group, pilot study, and two large-scale studies with questionnaires and data analysis for assessing each measures reliability and validity.
1: What are the benefits of a mixed-method design in a scale development process?
2: Why is the combination of different methods useful for assessing and ensuring the reliability and validity of the data?
Warning: The NCBI web site requires JavaScript to function. more...
An official website of the United States government
The .gov means it's official. Federal government websites often end in .gov or .mil. Before sharing sensitive information, make sure you're on a federal government site.
The site is secure. The https:// ensures that you are connecting to the official website and that any information you provide is encrypted and transmitted securely.
- Publications
- Account settings
- Browse Titles
NCBI Bookshelf. A service of the National Library of Medicine, National Institutes of Health.
Pollock K, Wilson E, Caswell G, et al. Family and health-care professionals managing medicines for patients with serious and terminal illness at home: a qualitative study. Southampton (UK): NIHR Journals Library; 2021 Aug. (Health Services and Delivery Research, No. 9.14.)

Family and health-care professionals managing medicines for patients with serious and terminal illness at home: a qualitative study.
Chapter 4 findings: participant demographics.
This chapter presents demographic information about participants in workstreams 1 and 2. Details of participants in workstream 3 are given in Chapter 8 .
- Recruitment
Workstream 1
Twenty-one BFCGs were recruited over a 15-month period between August 2017 and November 2018. Although we do not know how many information packs were subsequently given or sent to BFCGs, we have recorded that at least 94 information packs were given to 16 different HCP teams to distribute. Over half (12/21) of the participating BFCGs were referred via two hospices engaged with the study. Five were recruited through hospital teams across three hospitals. Two more were recruited via community nursing teams and the final two via community organisations.
Forty HCPs were recruited through a variety of routes over a 16-month period between June 2017 and October 2018. Information about the study was sent directly to key professional contacts, primarily via e-mail, and they were asked to distribute this to their networks. We also employed a snowball sampling technique by asking those interviewed to suggest other suitable HCPs or pass on information packs to colleagues. The Clinical Research Network representative supported engagement with general practices and several were visited to explain the study and leave information packs for both workstreams. We particularly targeted SPC teams, GPs, pharmacists and community nursing teams to obtain a range of perspectives from different professional roles.
Workstream 2
Over 136 patient information packs and 103 FCG information packs were provided to 16 different HCP sources. We do not know how many of these were given out. Twenty-two patient cases were recruited over a 15-month period between August 2017 and November 2018. Two patients were subsequently withdrawn: one died before sufficient data could be collected and it emerged, during interview, that the other did not meet the eligibility criteria for the study. Twenty patient case studies were completed. Nine further patients or FCGs expressed an interest in the study, but subsequently died or became too ill to take part. In addition, we are aware of 26 potential patients identified by HCPs and discussed with the research team for eligibility who did not go on to engage with the study. In some instances, the HCP subsequently decided that it was not appropriate to discuss the study, often because of a deterioration in the patient’s condition or family circumstances. In other instances, the patients or FCGs who received information packs decided against taking part and did not contact the researchers.
The 20 completed case study participants were recruited through one of three hospices (9/20), a hospital team such as respiratory (5/20), their GP (3/20), a CN (2/20) or a community organisation (1/20). The central participant in the case could be a patient or FCG, who could then go on to nominate other key people. Nominated individuals were then given information about the study and were asked to take part. We utilised a range of key contacts to recruit patients with conditions other than cancer and, where possible, from diverse or disadvantaged groups.
- Study participation
Workstream 1: bereaved family caregiver interviews
Twenty-one BFCGs were interviewed (15 women and six men). All were single interviews conducted face to face. Nineteen took place in the participant’s own home and two took place at the university. In relation to the patient, 11 identified as a wife, three as husbands, six as sons or daughters (including two in-laws) speaking about a mother, one was a father and one was a friend. The patients referred to in the BFCG interviews were 12 males and nine females and had a range of diagnoses ( Table 1 ), with many having multiple comorbidities. Of the 13 patients with cancer, there was considerable variety in the types of the disease, and for one patient cancer was not the primary diagnosis. Reflecting the high prevalence of cancer in this group, the majority ( n = 16) of patients in the BFCG accounts had been in receipt of SPC.
Bereaved family caregiver reports of patient diagnoses
Workstream 1: health-care practitioner interviews
We completed 27 interviews with HCPs, including one joint interview and three group interviews, with a total of 40 different participants from different professional roles ( Table 2 ). These were single interviews, usually taking place face to face at participants’ place of work, with four conducted by telephone.
Health-care practitioner participation by role
Workstream 2: case studies
The workstream 2 case studies included several different data sources ( Table 3 ).
Case study data collection
Fifteen of the 20 cases had patient participants who took part to a variable extent, depending on their health and abilities. Each of the 20 cases had between one and five participants, involving 48 separate participants ( Table 4 ). In four cases, the patient was the only participant, and in two other cases the FCG was the only participant. Fourteen HCPs were involved in 11 cases, with 11 of those participating in interviews and three in observations. Patients and FCGs reported that patients often had a number of conditions and it was not always possible to identify their primary diagnosis ( Table 5 ).
Case study participants
Main diagnoses of patients reported in case studies
Across all 20 case studies, a total of 54 interviews were conducted with patients, family members, friends and HCPs. FCGs were predominantly wives, but also husbands, siblings, sons, daughters and, in one case, a friend. HCP participation in the case studies included six HCWs/support workers, four consultants, two GPs, two clinical nurse specialists (CNSs) and one specialist palliative care nurse (SPCN). All interviews with case patients and FCGs took place at their home. Fifteen interviews were conducted as joint interviews with the patient and FCG (four with two FCGs present). In one interview, a HCW was also present. One patient chose to participate solely by telephone throughout their engagement with the study. Two follow-up FCG interviews were conducted by telephone. Three of the 11 interviews with HCPs were carried out by telephone. All others took place at the participants’ place of work. The average duration of data collection for each case, excluding review of medical records, was just under 4 months.
- Observations
Over the period of case study data collection, 11 observations were carried out with 10 case participants. The specific length of time spent on each observation was not recorded, but ranged from a few minutes to almost 2 hours. Observations were predominantly undertaken during visits to interview participants. Two were undertaken at outpatient appointments. These included observations of:
- general care and feeding via a PEG
- storage and management of medications in the home
- pain management consultation
- renal clinic outpatient appointment observation
- support worker administering medications
- a specialist nurse visit to discuss injections and ACP documents
- the delivery of medications from the pharmacy
- equipment (including hospital bed, hoist, lift, wheelchairs, walking aids, non-invasive ventilation, oxygen machine and wheelchair adaptation for digital tablet use).
- Photographs
With consent, photographs were taken as part of data collection in 13 of the cases. Participants gave an overarching written consent to allow photographs to be taken, and verbal consent was then used to check for each individual photograph once an item of interest was identified. Where a participant expressed an interest, they were also shown the photograph to check that they were happy with the content. These were visual ways to document medication storage, medication devices and equipment and to illustrate their impact on the space and environment of the home. They include pictures of dosette boxes, blister packs, personal storage boxes, locations of medication storage (e.g. kitchen cupboard), mobile telephone applications, alarms, non-invasive ventilation and oxygen machines, and equipment such as hospital beds, hoists and a lift installed in a living room. All photographs (see Figures 7 – 17 and Box 2 ) used in this report have been anonymised to remove both patient- and pharmacy-identifying data and each has been reproduced with permission from copyright holders Eleanor Wilson and Glenys Caswell (University of Nottingham, 21 January 2021).
Case study participant ecograms were completed during interviews, where appropriate, and subsequently included in diagrammatic summaries of each case compiled by the researchers on the basis of all data available within each case (see Appendix 1 ).
The largest network included 32 links and the smallest network included nine links (mean, n = 22 links; median, n = 20 links). Key caregivers or links were identified as individuals or services of particular emotional and/or practical importance to the case participants (indicated by dark colours in the ecograms in each case summary in Code Key – for case summaries). The number of key informal caregivers within each network was between zero and two, with two cases recording no informal caregivers as ‘key’. Key formal caregivers ranged from zero to four, with seven cases considered to have no key formal caregivers. Thirteen out of 20 cases had accessed SPC by the end of the study. In five cases, SPC links featured as key caregivers eight times. The most frequent formal caregiver link was with GPs ( n = 7) and the most cited informal caregiver link was a wife ( n = 10). Spouses in total were the most common key informal caregivers (13/20). Where HCWs were in place, they were always seen as key to care (four cases). Disease-specific CNSs were identified as key in three cases and DNs in two.
- Records review
Given the considerable time that sometimes elapsed between the completion of case interviews and the records review, we felt that it was appropriate to check consent given by patient participants at the start of the study. In five of the cases, there was no patient participant and in one other case the patient had refused consent. We deemed three further patient cases unsuitable to contact, either because we were aware that the patient had died or because the research team felt that it was inappropriate to make further contact. This left 11 cases for which consent had originally been given that needed to be checked. We endeavoured to follow up on each of these cases and to establish GP contact details to undertake reviews. Where the patient had died (in three cases), we discussed this with their FCG and each wanted to support the patient’s initial decision. One patient decided to revoke his initial consent. In two further cases, we were not able to contact the participant to confirm consent. This left eight participants with records that to review, two of whom also had hospice records that we were able to access ( Figure 6 ).
Process of reviewing patient records.
- Specialist palliative care
Initially, we had intended to search for differences in the experience of care between patients who had and patients who had not been referred to SPC. For the purposes of the study, the term SPC covers access to SPCNs, consultants and hospices. In practice, however, we found that this distinction was not particularly useful. In part, this was because of the very diverse conditions affecting patients, as well as the different stages of their illness and natural variation between these. All but three of the patient cases survived past the end of the study and would likely be referred for palliative care at a later stage of their illness. Consequently, we have not considered quality or experience of care in terms of a contrast between ‘specialist’ and ‘generalist’. All patients in workstreams 1 and 2 who were affected by cancer and half of those with other conditions had been referred for SPC. Some patients without cancer reported having prolonged input from specialist teams, including those dealing with long-term conditions, respiratory specialists and disease-specific nurses who had considerable experience and expertise in palliative and end-of-life care [e.g. nurses specialising in Parkinson’s disease, COPD, renal disease and motor neuron disease (MND)]. The most significant resource available to patients was the establishment of a key caregiver relationship with a particular HCP, and this resource was drawn from a wide range of general and specialist roles.
However, the established association between cancer and palliative care was apparent in the study data. Based on primary diagnosis, all four cancer patients in the case studies had prolonged hospice stays, outpatient appointments and SPC nursing at home. In comparison, for respiratory disease (e.g. COPD and emphysema), only one of four patients reported access to SPC. Several of these patients described a pattern of bypassing community professionals and contacting emergency and ambulance services directly when they recognised onset of an acute exacerbation of their condition that they anticipated from past experience would require hospitalisation. Three patients with MND had some access to SPC. The experiences of those with conditions such as heart failure, progressive supranuclear palsy (PSP), renal failure or multiple comorbidities all varied. In total, 13 out of our 20 cases reported accessing SPC at some level during their involvement in the study. The majority of BFCGs reported SPC input for their relatives prior to death in a group in which most patients were affected by cancer. The care of some patients in both workstreams had been predominantly managed by GP and DN teams.
- Do not attempt cardiopulmonary resuscitation
Do not attempt cardiopulmonary resuscitation (DNACPR) status was not routinely discussed during the interviews and it was not often mentioned in the BFCG interviews (6/21). However, from the cases for which we have been able to draw on multiple sources of data, we know that 12 patients had a DNACPR in place and two had some form of advanced directive (one with and one without a DNACPR).
- Anticipatory medications
Thirteen of the BFCGs reported that the patient had been prescribed AMs. In six instances, these were reported to have been used. Six of the case study participants also reported that AMs were in place. This number is likely to have increased as more of the participating case patients reached the end of life. A further four case study patients held a prescription for ‘just in case’ or ‘rescue’ antibiotics for chest infections. Across both groups, there did not appear to be any association between having anticipatory medications in place and having access to SPC.
- Syringe drivers
Ten of the patients described in the BFCG interviews had a syringe driver put in place for symptom management at the end of life. All but two of these patients had a diagnosis of cancer. Three of the case study patients also had syringe drivers in place. Two of these patients had cancer and one had renal failure. Across both groups, all of the patients recorded to have syringe drivers also had SPC input.
- Medications
Patient case studies
Within the 20 patient cases a total of 222 prescribed items were identified. Appendix 2 provides a list of all of the medication prescribed, as reported by participants or found in medical notes. We were able to review the medical notes of eight patients and in the other 12 we relied on participants’ accounts. Supporting information was also obtained in some interviews with HCPs who had immediate access to medical notes. On average, case patients had just under 12 prescribed medications and this ranged from 6 to 20. These include equipment and machines, such as oxygen concentrators, non-invasive ventilation and nebulisers, creams, eye drops, supplements (e.g. vitamins and fortifying drinks), laxatives and anticoagulants. There were a range of formulations prescribed, including tablets, liquids, injections, inhalers and patches. Complementary medicines were mentioned by participants, but not often and we did not enquire specifically about their use.
The aim of Appendix 2 is to illustrate the number, complexity and range of prescribed items across a relatively small cohort of 20 patients. The data show that participants frequently had more than one medication prescribed from a certain group of medications. Opioid analgesics were the most frequently prescribed medications, followed by proton pump inhibitors, which reduce acid in the stomach and are often prescribed to manage the side effects of other medications in palliative care.
Bereaved family caregiver interviews
We do not have extensive knowledge of the medications used by the relatives of BFCG participants and we have not attempted to collate this. Information was accessed during a single interview and based on participants’ recall of past events. However, some BFCGs gave very detailed accounts based on meticulous records maintained during the patient’s illness. The researchers were sometimes shown prescription and medicine lists. We cannot, of course, know how medications were taken by patients and whether or not they were taken as prescribed.
This chapter has presented background information on the participants who took part in the study and the different sources and extent of data collected. It includes a summary of the process of recruitment, characteristics of participants in workstreams 1 and 2, their involvement with SPC and the medications prescribed for case study patients. The next chapter gives a detailed account of a single case as a way of understanding the experience of treatment and decision-making at the end of life and to provide context for the key themes and issues presented in the chapters that follow.
- Cite this Page Pollock K, Wilson E, Caswell G, et al. Family and health-care professionals managing medicines for patients with serious and terminal illness at home: a qualitative study. Southampton (UK): NIHR Journals Library; 2021 Aug. (Health Services and Delivery Research, No. 9.14.) Chapter 4, Findings: participant demographics.
- PDF version of this title (80M)
In this Page
Other titles in this collection.
- Health Services and Delivery Research
Recent Activity
- Findings: participant demographics - Family and health-care professionals managi... Findings: participant demographics - Family and health-care professionals managing medicines for patients with serious and terminal illness at home: a qualitative study
Your browsing activity is empty.
Activity recording is turned off.
Turn recording back on
Connect with NLM
National Library of Medicine 8600 Rockville Pike Bethesda, MD 20894
Web Policies FOIA HHS Vulnerability Disclosure
Help Accessibility Careers

In order to continue enjoying our site, we ask that you confirm your identity as a human. Thank you very much for your cooperation.
- SUGGESTED TOPICS
- The Magazine
- Newsletters
- Managing Yourself
- Managing Teams
- Work-life Balance
- The Big Idea
- Data & Visuals
- Reading Lists
- Case Selections
- HBR Learning
- Topic Feeds
- Account Settings
- Email Preferences
Case Study: Are the Right People in the Right Seats?
- Nitin Nohria

A new CEO considers changes to her top team.
The newly appointed CEO of Highstreet Properties has doubts about several members of the top team she has inherited. She’s trying to drive a turnaround, the company has a complicated matrix structure, and some team members seem opposed to her strategy. She’s debating replacing several of them, but she’s worried about making too many changes too quickly, upsetting her board, and bringing in too many former colleagues.
Shannon Levy, the new CEO of Highstreet Properties, stared out the window of the company’s London headquarters, wondering whether she should call Justin Mooney and fire him. A once-thriving developer of retail malls, Highstreet had been battered by consumers’ shift to e-commerce, Covid-19 mall closures, and internal discord over strategy. Shannon had been brought in to turn the business around. She was fast approaching her 100-day mark and already feeling behind on selecting, aligning, and motivating her senior leadership team.
- Nitin Nohria is the George F. Baker Jr. and Distinguished Service University Professor. He served as the 10th dean of Harvard Business School, from 2010 to 2020.
Partner Center
Official websites use .gov
A .gov website belongs to an official government organization in the United States.
Secure .gov websites use HTTPS
A lock ( ) or https:// means you've safely connected to the .gov website. Share sensitive information only on official, secure websites.
The Untreated Syphilis Study at Tuskegee Timeline
In 1932, the USPHS, working with the Tuskegee Institute, began a study to record the natural history of syphilis. It was originally called the “Tuskegee Study of Untreated Syphilis in the Negro Male” (now referred to as the “USPHS Untreated Syphilis Study at Tuskegee”). The study initially involved 600 Black men – 399 with syphilis, 201 who did not have the disease. Participants’ informed consent was not collected. Researchers told the men they were being treated for “bad blood,” a local term used to describe several ailments, including syphilis, anemia, and fatigue. In exchange for taking part in the study, the men received free medical exams, free meals, and burial insurance.
The U.S. Public Health Service (USPHS) engages the Tuskegee Institute in Macon, AL in the USPHS Tuskegee Untreated Syphilis Study. 2
Penicillin becomes treatment of choice for syphilis , but men in study are not treated.
First news article about the study.
The study ends , on recommendation of an Ad Hoc Advisory Panel convened by the Assistant Secretary for Health and Scientific Affairs.
President Clinton issues a formal Presidential apology .
By 1943, penicillin was the treatment of choice for syphilis and becoming widely available , but the participants in the study were not offered treatment.
In 1972, an Associated Press story about the study was published. As a result, the Assistant Secretary for Health and Scientific Affairs appointed an Ad Hoc Advisory Panel to review the study. The advisory panel concluded that the study was “ethically unjustified”; that is, the “results [were] disproportionately meager compared with known risks to human subjects involved.” In October 1972, the panel advised stopping the study. A month later, the Assistant Secretary for Health and Scientific Affairs announced the end of the study. In March 1973, the panel also advised the Secretary of the Department of Health, Education, and Welfare (HEW) (now known as the Department of Health and Human Services) to instruct the USPHS to provide all necessary medical care for the survivors of the study. 1 The Tuskegee Health Benefit Program (THBP) was established to provide these services. In 1975, participants’ wives, widows and children were added to the program. In 1995, the program was expanded to include health, as well as medical, benefits. The last study participant died in January 2004. The last widow receiving THBP benefits died in January 2009. Participants’ children (10 at present) continue to receive medical and health benefits.
Later in 1973, a class-action lawsuit was filed on behalf of the study participants and their families, resulting in a $10 million, out-of-court settlement in 1974.
On May 16, 1997, President Bill Clinton issued a formal Presidential Apology for the study.
1 “HEW News” Office of the Secretary, March 5, 1973; Memorandum “USPHS Study of Untreated Syphilis (the Tuskegee Study; Authority to Treat Participants Upon Termination of the Study,” from Wilmot R Hastings to the secretary, March 5, 1973.
2 Vonderlehr, R.A., Clark, T., Wenger, O.C., Heller, J.R., Untreated Syphilis in the Male Negro, Journal of Venereal Disease Information. 17:260-265, (1936).
The content here can be syndicated (added to your web site).
Related Links
- Facts & Information about Syphilis
- STD Health Equity
- The National Archives – USPHS Untreated Syphilis Study at Tuskegee
- Tuskegee University National Center for Bioethics in Research and Health Care
Exit Notification / Disclaimer Policy
- The Centers for Disease Control and Prevention (CDC) cannot attest to the accuracy of a non-federal website.
- Linking to a non-federal website does not constitute an endorsement by CDC or any of its employees of the sponsors or the information and products presented on the website.
- You will be subject to the destination website's privacy policy when you follow the link.
- CDC is not responsible for Section 508 compliance (accessibility) on other federal or private website.
- Open access
- Published: 19 June 2024
Efficacy and safety of eliapixant in endometriosis-associated pelvic pain: the randomized, placebo-controlled phase 2b SCHUMANN study
- Susanne Parke 1 ,
- Kerstin Gude 2 ,
- Katrin Roth 1 &
- Fabrizio Messina 3
BMC Women's Health volume 24 , Article number: 353 ( 2024 ) Cite this article
Metrics details
The SCHUMANN study evaluated the efficacy and safety of the selective P2 × 3 antagonist eliapixant in patients with endometriosis-associated pelvic pain (EAPP).
SCHUMANN was a randomized, placebo- and active comparator-controlled, double-blind to placebo and open-label to comparator, parallel-group, multicenter, dose-finding phase 2b study. The participants were women with surgically diagnosed endometriosis who fulfilled defined EAPP criteria. Participants were randomized 1:1:1:1 to twice daily (BID) 25 mg, 75 mg, or 150 mg oral eliapixant or a placebo for 12 weeks. An exploratory once-daily elagolix 150 mg treatment group was also included. The primary endpoint was the absolute change in mean worst EAPP from baseline to the end of intervention (EOI).
Overall, 215 participants were randomized for treatment (44 to eliapixant 25 mg, 44 to eliapixant 75 mg, 43 to eliapixant 150 mg, 43 to a placebo, and 41 to elagolix 150 mg). For safety reasons, the study was terminated early; both treatment and enrollment stopped immediately, producing less than 50% of the planned number of completers. The study found no significant differences in EAPP reduction from baseline between groups and no significant dose-response model. The elagolix 150 mg group showed better pain reduction than any of the other groups. No new safety signals were observed, relative to the previously known safety profile of eliapixant, which was generally well tolerated. However, one case of moderate and probably drug-induced liver injury in a participant receiving eliapixant 150 mg BID supported the association between eliapixant and a potential increase in liver function values, defined before the start of the phase 2 program.
Conclusions
This study did not meet its primary objective as no statistically significant or clinically relevant differences in changes of mean worst EAPP from baseline were observed between treatment groups. The single observed case of moderate, probably drug-induced liver injury was the second case in the eliapixant phase 2 program conducted in the following indications: refractory or unexplained chronic cough, diabetic neuropathic pain, overactive bladder, and EAPP. Due to this, the benefit-risk ratio for the study was no longer considered to be positive.
Clinical trial registration
ClinicalTrials.gov identifier NCT04614246; registered November 3, 2020.
Peer Review reports
Introduction
Endometriosis is a chronic, benign, sex-hormone-dependent inflammatory disease, characterized by the presence of endometrium-like tissue outside the uterine cavity [ 1 , 2 , 3 , 4 ] and causing a substantial burden to individuals and society [ 5 , 6 , 7 ]. Although its precise prevalence is unknown, estimates range from 1% to 15% within the general female population [ 5 , 8 , 9 , 10 ]. Endometriosis is characterized by pain (primarily chronic, non-menstrual pelvic pain [NMPP]), pain during menstruation (dysmenorrhea) and sexual intercourse (dyspareunia), and infertility. The burden of the disease and the limitations of currently available treatment modalities have created a serious unmet medical need.
P2X3 is a non-selective cation channel activated by adenosine triphosphate (ATP); it has been described as a prominent mediator of pain [ 11 ]. P2X3 receptors are predominantly localized on primary sensory afferent neurons in small-to-medium diameter C and Aδ fibers throughout the body [ 11 , 12 , 13 ], which carry nerve impulses from sensory stimuli toward the central nervous system [ 12 , 14 ]. Afferent neuronal hypersensitization via P2X3 receptor signaling plays a significant role in the pathways that trigger cough and bladder urgency and may influence the pathophysiology of endometriosis-associated chronic pain, overactive bladder (OAB), and diabetic neuropathic pain (DNP) [ 13 , 15 , 16 , 17 , 18 , 19 ].
The expression of P2X3 receptors is upregulated during inflammation; this has been shown to sensitize peripheral nerves and contribute to central sensitization. Sensory nerve fibers within endometriotic lesions can themselves release inflammatory mediators (such as ATP, substance P, and nerve-growth factor) through a process known as neurogenic inflammation, which is thought to play a critical role in endometriosis-associated pain [ 20 , 21 , 22 , 23 , 24 ]. P2X3 expression in both endometriotic endometrium and lesions has been shown to be significantly higher than that found in control endometrium and positively correlated with pain. P2X3 channels are activated by ATP, which is released in response to various stimuli, including tissue injury, mechanical stress such as movement and distension, and the inflammation present in endometriotic lesions, particularly during menstruation [ 25 , 26 , 27 , 28 ]. Antagonizing P2X3 receptors was expected to combine high levels of pain relief with disease modification via an innovative mechanism.
Eliapixant is a potent P2X3 receptor antagonist that preferentially blocks the homomeric P2X3 channel. In a rat neurogenic inflammation model, eliapixant demonstrated robust efficacy in blocking inflammation of the skin evoked by an injection of mustard oil into the uterus and a concomitant transduction of the inflammation via the nervous system (i.e., neurogenic inflammation). It showed significant efficacy on visceral pain in a rat dyspareunia model and was still present 1 week after treatment, suggesting that eliapixant demonstrated efficacy beyond direct analgesic effects.
The goal of this project was to develop eliapixant as non-hormonal treatment for moderate-to-severe pain associated with endometriosis in women of reproductive age. Eliapixant was expected to provide clinical efficacy in the long-term management of pain associated with endometriosis, including a clinically meaningful reduction in chronic pelvic pain, dysmenorrhea, and dyspareunia, with a safety profile suitable for long-term use. It was also expected to improve patients’ quality of life substantially. At the start of this phase 2 study, these anticipated beneficial effects had not yet been demonstrated in patients with endometriosis-related pain. The phase 2b SCHUMANN study aimed to identify the optimal dose of eliapixant in patients with EAPP, to further assess efficacy, and to characterize the safety and tolerability profile of eliapixant over 12 weeks. Eliapixant has been developed in the indications OAB, refractory or unexplained chronic cough (RUCC), and DNP.
Study design
SCHUMANN (ClinicalTrials.gov NCT04614246) was a randomized, double-blind, open for active comparator, parallel-group, multicenter, phase 2b study to assess the efficacy and safety of three different doses of eliapixant (BAY 1817080) vs. placebo twice daily (BID) and elagolix 150 mg once daily in women with symptomatic endometriosis conducted at 144 centers in 20 countries across Europe, North America, China, and Japan (see Supplemental Methods for further details). The study comprised a 28-day screening phase, a 35-day pre-intervention period, an 84 + 3-day period of double-blind intervention, and a follow-up period lasting 38/90 days. When the protocol amendment was filed due to study termination, a comprehensive safety follow-up was implemented; this consisted of two additional visits for laboratory safety testing and a required 90-day follow-up for all participants, apart from those randomized to elagolix 150 mg (Supplemental Fig. S1 ). The latest version of the study protocol, including amendments and the statistical analysis plan, is available on ClinicalTrials.gov.
Eligible participants were centrally randomized 1:1:1:1:1 by the sponsor, using an interactive web response system (IWRS), to receive one of three oral doses of eliapixant BID (25 mg, 75 mg, or 150 mg; Bayer AG, Berlin, Germany), a placebo, or elagolix 150 mg once daily (QD). No participants from Japan or China were randomized to elagolix. To maintain blinding, tablets containing the placebo were identical in size, color, and shape to those containing eliapixant.
In addition to the study drugs described above, participants were only allowed to use standardized rescue medication (i.e., ibuprofen, acetaminophen, and tramadol) for treatment of endometriosis-associated pain. Women interested in participating in the study had to stop intake of hormonal treatments. Long-acting hormonal contraceptives as well as gonadotrophin-releasing hormone (GnRH) agonists, - GnRH antagonists, and progesterone receptor modulators had to be stopped at least 28 days before screening; all other hormonal therapies had to be stopped at Visit 1.
Ethics approval and consent to participate
The Institutional Review Board/Independent Ethics Committee at each center approved the protocol. The study was carried out in accordance with Good Clinical Practice guidelines, the Declaration of Helsinki, and the International Ethical Guidelines of the Council for International Organizations of Medical Sciences. All participants provided written informed consent.
Participants
The investigators enrolled women aged ≥ 18 years with endometriosis surgically diagnosed between 10 years and ≥ 8 weeks before screening and with self-reported moderate-to-severe pain. During the screening period, potential participants made at least 24 daily entries in an endometriosis symptom diary (ESD [ 29 ]), using the daily numeric rating scale (NRS) to indicate ESD item 1a (“worst pain”) summing up to ≥ 98. In Japan, inclusion by clinical diagnosis as opposed to surgical diagnoses was acceptable for up to 50% of the participants. Full inclusion and exclusion criteria are included in the Supplementary Methods .
Patient-reported outcome (PRO) assessments were collected using an electronic handheld diary (eDiary); pain catastrophizing and painDETECT scores were collected via paper questionnaires (see Supplemental Methods ). Safety was monitored throughout the study (adverse events [AEs], clinical laboratory, and vital signs). Two questions on hair and eye color, previously associated with endometriosis and potentially useful in marking genetic subpopulations, were included among the baseline characteristics [ 30 , 31 ].
Study endpoints
The primary efficacy endpoint was the absolute change in mean worst EAPP (i.e., ESD item 1, measured daily via the NRS) from baseline to end of intervention (EOI). Other pre-specified endpoints included absolute change in mean worst EAPP from baseline to the first 4/8 weeks of intervention; absolute change in mean worst EAPP on bleeding and non-bleeding days from baseline to the first 4/8 weeks of intervention/EOI (measured via NRS by items 1 and 4 [i.e., intensity of vaginal bleeding, measured daily using a categorical response scale with five levels of increasing intensity: none, spotting, light, normal, heavy] of the ESD), item level, total and/or domain scores at screening, baseline, and Weeks 4/8/EOI and absolute change from baseline, as applicable, using the data collected by PROs (see Supplemental Methods ) and absolute change in mean worst EAPP (overall, during, and outside days with vaginal bleeding) from EOI to non-overlapping, consecutive 28-day intervals during follow-up.
Treatment-emergent AEs (TEAEs) and serious AEs were recorded in line with the Medical Dictionary for Regulatory Activities (MedDRA), version 25.0. Additional safety assessments are described in the Supplemental Methods . At the end of the study, participants who spontaneously reported a taste-related AE completed an assessment on taste disturbances.
Statistical analysis
A multiple comparison procedure modeling (MCP-Mod) approach [ 32 ] was used to complete a pre-specified analysis of the primary efficacy endpoint. As SCHUMANN was a phase 2b dose-finding study, the MCP-Mod approach, a well-accepted dose-finding method that uses available data more efficiently than traditional pairwise comparisons, was adopted [ 33 , 34 ]. The MCP-Mod approach makes it possible to estimate a dose response and to select an optimum dose for further phase 3 trials [ 34 ].
To detect a dose–response signal, four candidate dose–response models were tested with a single contrast test using the generalized MCP-Mod approach. The null hypothesis (“the response to all doses is equal”) was tested against the alternative (“there is a dose–response relationship”). If at least one of the four individual model tests was statistically significant (adjusted p of one-sided test ≤ 0.1), a dose–response signal would be established. The model with the best fit would then be used to estimate the dose–response curve and minimum effective dose. For further information on the primary endpoint analysis, see the Supplementary Methods .
Sample size calculations were conducted to establish evidence of a drug effect across the doses. A sample size of 50 evaluable participants per dose group was predicted to have approximately 90% power to demonstrate a dose–response relationship for the primary efficacy endpoint, using a one-sided test at a type I error rate of α = 0.10 (see Supplementary Methods for more details).
The secondary endpoint analyses and definitions of the per-protocol as well as the full- and safety-analysis sets are described in the Supplementary Methods .
A statistical evaluation was performed using SAS software version 9.4 (SAS Institute, USA) and ValidR software version 3.5.2 or higher (Mango Solutions, UK). Confirmatory p -values are reported for the analysis of the primary endpoint. The study was not powered to make individual pairwise comparisons between dose groups. The analyses of secondary endpoints, sensitivity, and AEs should be viewed as exploratory.
In total, 504 participants were screened between 01/29/2021 and 01/27/2022, of whom 215 were randomized to eliapixant 25 mg ( n = 44), 75 mg ( n = 44), 150 mg ( n = 43), placebo ( n = 43) (all BID), or elagolix 150 mg ( n = 41) (Fig. 1 ). Follow-up continued until 05/03/2022. Although all 215 randomized participants were included in the full-analysis set, 25 did not receive a study intervention; thus, the safety-analysis set includes 190 participants. A total of 183 participants were included in the per-protocol set (for a definition see Supplemental Methods ). In total, 120 participants (42.7%) completed the treatment period.

Participant disposition. BID twice daily, FAS full-analysis set, SAS safety-analysis set
Table 1 presents an overview of per-protocol set demographic characteristics. Similar results were obtained in the full-analysis set. Age and body mass index (BMI) were generally well balanced across the treatment arms (Table 1 ). The mean (standard deviation [SD]) age was 34.64 (7.22) years, with 26.8% of participants aged < 30 years, 50.3% aged 30–40 years, and 23.0% aged > 40–55 years. The mean (SD) BMI was 25.54 (5.89) kg/m 2 . In the per-protocol set, 73.8% of participants were white. In the elagolix 150 mg arm, however, 94.6% were white, as participants from Japan and China could not be randomized to elagolix (Table 1 ).
Baseline demographics and clinical characteristics were generally well balanced across the treatment groups (Table 1 ). Slight imbalances between the treatment groups were observed in relation to mean worst pain, NMPP, and dysmenorrhea. The mean (SD) of mean worst pain, NMPP, and dysmenorrhea was higher in the eliapixant 150 mg BID arm (6.63 [1.62], 6.45 [1.78], and 7.38 [1.39]), respectively, and in the elagolix 150 mg arm (6.38 [1.78], 6.18 [1.86], and 7.29 [1.84]), respectively, than in other arms (Table 1 ).
Slight imbalances between the treatment groups were also observed with regard to hair and eye color. As expected, given the participating countries, about half of the population (48.6%) had brown eyes. However, the proportion ranged between 35.1% in the elagolix 150 mg arm, which had predominantly white participants (no women from China or Japan were randomized to this treatment group), and 62.2% in the eliapixant 25 mg group. The full-analysis set results were similar.
The data for the primary efficacy endpoint, absolute change in mean worst EAPP from baseline to EOI (measured daily on the NRS as ESD item 1) are shown in Table 2 .
At Week 12, the end of the intervention, the mean (SD) changes in mean worst EAPP from baseline were − 1.56 (1.35) in the eliapixant 25 mg BID arm, − 2.12 (2.66) in the eliapixant 75 mg BID arm, − 1.88 (2.03) in the eliapixant 150 mg BID arm, and − 1.89 (1.91) in the placebo arm, based on the primary per-protocol set. The full-analysis set results were similar. For elagolix, the open-label comparator arm, the mean (SD) of mean worst EAPP was 6.38 (1.76) at baseline and 3.39 (2.57) at Week 12 (EOI), resulting in a mean (SD) change from baseline of − 2.83 (2.38), based on the full-analysis set.
The least squares (LS) mean (standard error [SE]) of change obtained via mixed-model repeated measures from baseline to Week 12 (EOI) was − 1.63 (0.38) in the eliapixant 25 mg BID arm, − 2.13 (0.41) in the eliapixant 75 mg BID arm, − 1.96 (0.41) in the eliapixant 150 mg BID arm, and − 1.94 (0.38) in the placebo arm. Overall, no significant differences were observed across the various treatment arms.
At Week 12 (EOI) the adjusted p -values for the Emax 1, Emax 2, SigEmax 1, and SigEmax 2 candidate models were 0.5266, 0.4679, 0.4052, and 0.4082, respectively. No significant dose-response model was found (Fig. 2 ).

The dose response model and target dose for the change over 28 days indicate the change in worst EAPP from baseline to Week 12 (with 80% CI). Left: Emax model; right: SigEmax model. CI confidence interval, EAPP endometriosis-associated pelvic pain
A secondary analysis on the primary per-protocol set, including the elagolix arm, resulted in similar LS mean changes from baseline for the placebo and eliapixant arms, but showed an LS mean change (SD) from baseline of − 2.69 (0.41) for the elagolix arm, resulting in a difference of approximately − 0.8 from the placebo. This difference is comparable to the effect of elagolix 150 mg QD observed in previous studies [ 35 ]. The mean EAPP on bleeding days was higher at baseline than the overall EAPP, with a mean (SD) of 7.19 (1.62) across all treatment groups and a change from baseline ranging from − 1.07 (1.7) to − 1.73 (2.1), a smaller effect than those observed for overall EAPP. Mean EAPP on non-bleeding days was slightly lower at baseline than overall EAPP, with a mean (SD) of 6.00 (1.87) across all treatment groups and a change from baseline similar to that observed for overall EAPP. No relevant dose–response relationship was observed for any of these endpoints.
Throughout the entire study, participants were asked to report pain at its worst during the past 24 h on a 0–10 NRS on the eDiary to assess dyschezia and dysuria. At baseline, dyschezia mean worst pain was present across all treatment groups with a mean (SD) of 3.78 (2.89), and the means (SDs) in the different treatment groups ranging from 3.04 (2.83) in the eliapixant 25 mg BID group to 4.15 (2.68) in the elagolix 150 mg group. After 12 weeks of treatment, a reduction was observed in all treatment groups, with mean (SD) changes from baseline of − 0.65 (0.81) in the eliapixant 25 mg BID group up to − 2.18 (1.95) in the elagolix 150 mg group. Dysuria mean worst pain at baseline was observed with an overall mean (SD) of 2.80 (2.79), ranging from 2.37 (2.82) in the eliapixant 25 mg BID group to 3.40 (2.78) in the elagolix 150 mg group. After 12 weeks of treatment, a reduction was observed in all treatment groups, with mean (SD) changes from baseline of − 0.67 (1.11) in the eliapixant 25 mg BID group up to − 1.67 (2.01) in the elagolix 150 mg group. No dose-response trend and no relevant difference to placebo were observed for dyschezia or dysuria in the different eliapixant groups.
A higher percentage of participants experienced TEAEs in the eliapixant 150 mg BID arm (76.3% [ n = 29]), placebo arm (73.0% [ n = 27]), and elagolix 150 mg arm (73.7% [ n = 28]) than did so in the other two lower-dose eliapixant treatment arms (eliapixant 25 mg BID (59.0% [ n = 23]) and eliapixant 75 mg BID (55.3% [ n = 21]). The same pattern was seen in the percentage of participants with TEAEs attributed to study intervention (Table 3 ). No AEs/TEAEs had death as an outcome. Few participants had serious TEAEs (Table 3 ). Two serious TEAEs were assessed as related to the study intervention: one in the eliapixant 150 mg BID arm (MedDRA preferred term [PT] “Liver function test increased”) and one in the elagolix 150 mg arm (MedDRA PT “Hypertensive crisis”). A low percentage of participants stopped using the study drug permanently due to a TEAE (Table 3 ).
Table 4 summarizes the most commonly reported TEAEs in each treatment arm, with headache being the most frequently reported TEAE across all treatment arms. In almost all safety-analysis set participants, the maximum intensity of TEAEs was assessed as either mild (43.7%) or moderate (20.0%). Only 7 participants reported severe TEAEs: 3 participants in the eliapixant 150 mg arm, 2 participants in the placebo arm, and 2 participants in the elagolix 150 mg arm.
Most TEAEs attributed to study intervention affected single participants and were assessed as having either mild or moderate maximum intensity. The TEAEs attributed to study intervention and reported by at least 2 participants were as follows:
In the eliapixant 75 mg BID arm: 2 participants reported intermenstrual bleeding.
In the eliapixant 150 mg BID arm: 2 participants reported dysgeusia, 2 participants reported taste disorder, and 3 participants reported hypogeusia.
In the elagolix 150 mg arm: 2 participants reported headache and 6 participants reported hot flushes.
For 1 participant in the placebo arm (PT: “Nightmares”) and 2 participants in the elagolix 150 mg arm (PT: “Hot flushes”, PT: “Hypertensive crisis”), TEAEs attributed to study intervention were assessed as severe at maximum intensity.
Ten taste-related AEs were reported in the eliapixant 150 mg BID (6 participants, 15.8%; one of those reported three events) and placebo arms (2 participants, 5.4%). Eight of the 10 taste-related AEs were attributed to the study medication and impacted participants in the eliapixant 150 mg arm. No participant discontinued treatment due to a taste-related AE.
Increases in liver function parameters and antithrombin III activity were described as potential risks in the study protocol and monitored closely across the study period. No mean increase over time or relevant differences were observed across the treatment arms in relation to the mean values of aspartate aminotransferase (AST), alanine aminotransferase (ALT), gamma-glutamyl transferase (GGT), or bilirubin. However, a dose-dependent increase in the mean values of alkaline phosphatase (AP) was seen at Week 2 after the start of the study intervention. From Week 4 onward, the values remained relatively stable until the end of intervention at Week 12. All changes were reversible and returned to baseline levels by the safety follow-up visit—approximately 38 days after the end of the intervention. The clinical relevance of this finding and the origins of the increase (i.e., liver vs. bone) are unclear in the absence of a concurrent increase in the mean ALT, AST, bilirubin, or GGT values. In total, 4 participants (10.3%) in the eliapixant 25 mg BID arm, 3 participants (7.9%) in the eliapixant 75 mg BID arm, 1 participant (2.6%) in the eliapixant 150 mg BID arm, 2 participants (5.4%) in the placebo arm, and 3 participants in the elagolix 150 mg arm reported TEAEs associated with the Standardized MedDRA Query “Drug-related hepatic disorders”. One participant in the eliapixant 150 mg BID arm experienced a drug-induced liver injury, reported as a suspected unexpected serious adverse reaction (SUSAR). The 44-year-old study participant was diagnosed with “liver function test increased” (AST 4.8-fold upper limit of normal [ULN], ALT 7.5-fold ULN) at her regular visit after 4 weeks of treatment with the study drug; normal values had been recorded at screening and baseline and slightly increased values at Week 2 after the start of treatment. Unblinding revealed that the patient had received a dose of 150 mg BID eliapixant. Other liver parameters remained within normal ranges. Treatment with the study drug was discontinued immediately. This patient reported malaise and edema starting 3 weeks after the treatment began. She recovered from her symptoms and her transaminases returned to normal 2 weeks (AST) and 4 weeks (ALT) after the treatment ended. A detailed analysis of alternative causes of this patient’s moderate liver injury, based on the Food and Drug Administration 2009 Drug-Induced Liver Injury Guideline [ 36 ], found that she was reactive for anti-hepatitis A virus (HAV) immunoglobulin M (IgM) antibodies at baseline and at 2, 4, and 6 weeks thereafter as a potential alternative cause. However, no HAV ribonucleic acid or anti-HAV immunoglobulin G (IgG) antibodies were detected in serum at any point in time (unfortunately, no stool sample was collected or analyzed). Furthermore, no anti-HAV IgG antibodies were detected until 10 weeks after the first anti-HAV IgM antibodies were found; earlier detection would be expected during the course of a typical HAV infection. The totality of data gathered to clarify the causality of this case did not support a recent acute infection by HAV. In conclusion, the case was assessed by an external hepatology expert as an acute hepatocellular liver injury of moderate severity, based on the associated symptoms. The suggestive chronology and the absence of any other causes with clear potential made eliapixant the most likely cause of the liver injury.
A mean and median increase in antithrombin activity was observed in the eliapixant arms but not in the placebo arm, in line with similar findings from other phase 1 and 2 studies with eliapixant [ 37 , 38 , 39 ]. However, no differences were observed in TEAEs involving bleeding between the treatment arms.
A mean increase in fibrinogen was seen in the eliapixant arms, but not in the placebo arm, confirming a similar finding in other phase 2 studies with eliapixant [ 38 , 39 ]. The clinical relevance remains unclear, as there was no difference between eliapixant and the placebo regarding potential clinical manifestations of such changes, e.g., frequency of TEAEs involving bleeding or thromboembolic events.
Six participants reported a pregnancy during this study: 2 in the placebo arm and 1 in the elagolix 150 mg arm. Three participants reported pregnancies conceived during treatment: 2 in the eliapixant 25 mg arm and 1 in the eliapixant 150 mg arm.
Endometriosis, a debilitating gynecologic condition affecting millions of women worldwide, poses significant challenges in both diagnosis and management. A general lack of awareness by women and healthcare providers results in a significant delay from when a woman first experiences symptoms until she eventually is diagnosed and treated. Despite its prevalence and profound impact on quality of life, there remains a critical need for further research and the development of more effective treatment options [ 40 ].
To date, none of the medical treatments have been able to cure the disease, most treatments are not suitable for long-term use due to side effects [ 41 ], and symptoms recur as soon as the medication is stopped. Hormone treatments for endometriosis include combined contraceptives, progestogens, GnRH agonists, GnRH antagonists, and aromatase inhibitors. All treatments lead to a clinically significant reduction in pain with a similar magnitude of the treatment effect. Symptoms return after cessation of treatment and all hormones used to manage endometriosis have unwanted side effects [ 42 ]. Non-steroidal anti-inflammatory drugs are effective in reducing EAPP, but also have significant side effects, including gastric ulceration. Surgery can be effective to remove endometriosis lesions and scar tissue, but success rates are dependent on the extent of disease and the surgeon’s skills.
The SCHUMANN study aimed to assess the efficacy and safety of three different doses of eliapixant, in comparison with a placebo and elagolix 150 mg, in women with symptomatic endometriosis. The study was terminated prematurely, due to liver safety concerns; as a result, less than 50% of the planned number of participants completed the study.
Reductions in mean worst EAPP were observed at Week 12 in all treatment groups; no significant differences were observed across the treatment arms or compared with a placebo. At end of the intervention (Week 12), the mean (SD) changes in mean worst EAPP from baseline were − 1.56 (1.35) in the eliapixant 25 mg BID arm, − 2.21 (2.66) in the eliapixant 75 mg BID arm, − 1.88 (2.03) in the eliapixant 150 mg BID arm, and − 1.89 (1.91) in the placebo arm (primary per-protocol set). Only the elagolix 150 mg arm achieved better pain reduction than the other treatment arms of − 2.83 (2.38) (full-analysis set). The observed efficacy of elagolix was in line with expectations.
No significant dose-response model was found at any time. As no statistically significant or clinically relevant differences were observed between the treatment groups, this study did not meet its primary objective.
Due to the low percentage of women who completed the study, it was not considered meaningful to carry out subgroup analyses, for example with regard to hair and eye color.
No new safety signals were observed in relation to the known safety profile of eliapixant, which was generally well tolerated in this study. More participants experienced TEAEs in the eliapixant 150 mg BID, placebo, and elagolix 150 mg arms than in the two lower-dosed eliapixant treatment arms; no specific event caused this difference between the treatment arms.
Increases were observed in mean antithrombin activity, fibrinogen, and AP, confirming similar findings from other phase 2 studies with eliapixant [ 37 , 38 , 39 ]. The clinical relevance of these findings remains unclear, as there was no difference between eliapixant and the placebo with regard to clinical manifestations of these changes (e.g., TEAEs related to bleeding or thromboembolic events). Taste-related AEs were observed only in the eliapixant 150 mg BID (6 participants, 15.8%) and placebo (2 participants, 5.4%) arms.
The one observed case of moderate, probably drug-induced liver injury in a SCHUMANN participant receiving eliapixant 150 mg BID was the second case in the eliapixant phase 2 program with the following indications: RUCC, DNP, OAB, and EAPP. The first case occurred in a 26-year-old female participant after 4 weeks of exposure to eliapixant 150 mg BID in the phase 2b study of patients suffering from RUCC [ 38 ]. Although increases in liver function parameters with eliapixant treatment had been defined as a potential risk before the start of phase 2 studies, the benefit-risk ratio for the SCHUMANN study was no longer considered positive following two cases of moderate drug-induced liver injury of hepatocellular origin in participants exposed to eliapixant for 8–12 weeks of treatment during the phase 2 program in all indications and considering the totality of liver-safety data from the phase 2 program. The study was therefore put on clinical hold with an immediate stop of treatment and enrollment.
The strengths of SCHUMANN include its baseline demographics, which largely reflect those seen in a typical endometriosis population. The recruitment of participants across 20 countries meant that the results were likely to reflect the global population of patients with endometriosis. The limitations of the study include its premature termination, which resulted in less than 50% of the planned number of completers.
In summary, the SCHUMANN study did not show any significant differences in mean worst EAPP reductions at Week 12 across various treatment arms or compared with a placebo. The tolerability profile of eliapixant was consistent with that observed in other phase 2 studies of the program. However, the benefit-risk ratio for this study was no longer considered positive following the second case of a moderate, probably drug-induced liver injury in a participant receiving eliapixant 150 mg BID in the phase 2 program. The study was terminated prematurely; subsequently, Bayer AG discontinued the entire development program in all indications.
Overall, there is need for further research in the fields of new treatment options and early diagnosis of endometriosis, particularly in young patients.
Availability of data and materials
The availability of data underpinning this publication will be determined in accordance with Bayer’s commitment to the European Federation of Pharmaceutical Industries and Associations and Pharmaceutical Research and Manufacturers of America and their principles for responsible clinical trial data sharing, pertaining to scope, timepoint, and the data access process. At the request of qualified scientific and medical researchers, Bayer commits to sharing patient-level clinical trial data, study-level clinical trial data, and protocols from patient clinical trials of medicines and indications approved in the US and European Union, as required for legitimate research. This commitment applies to data on new medicines and indications approved by the European Union and US regulatory agencies on or after January 1, 2014. Interested researchers can use www.clinicalstudydatarequest.com to request access to anonymized patient-level data and supporting documents from clinical studies in order to carry out further research to advance medical science or improve patient care. Alternatively, researchers may contact Susanne Parke (Research and Development, Bayer AG, Berlin, Germany; [email protected]). Information on the Bayer criteria for listing studies and other relevant information is provided in the study sponsors section of the portal. Access will be granted to anonymized patient-level data, protocols, and clinical study reports following approval by an independent scientific review panel. Bayer is not involved in the decisions made by the independent review panel and will implement all necessary measures to safeguard patient privacy.
Data availability
No datasets were generated or analyzed during the current study.
Abbreviations
Adverse event
Alanine aminotransferase
Alkaline phosphatase
Aspartate aminotransferase
Adenosine triphosphate
Twice daily
Body mass index
Confidence interval
Diabetic neuropathic pain
- Endometriosis-associated pelvic pain
Electronic handheld diary
End of intervention
Endometriosis symptom diary
Gamma-glutamyl transferase
Gonadotrophin-releasing hormone
Anti-hepatitis A virus
Immunoglobulin G
Immunoglobulin M
Least squares
Multiple comparison procedure modeling
Medical Dictionary for Regulatory Activities
Non-menstrual pelvic pain
Numeric rating scale
Overactive bladder
Patient-reported outcome
Preferred term
Refractory or unexplained chronic cough
Serious adverse event
Standard deviation
Treatment-emergent adverse event
Upper limit of normal
Zondervan KT, Becker CM, Koga K, Missmer SA, Taylor RN. Viganò P. Endometriosis. Nat Rev Dis Primers. 2018;4(1):9.
Article PubMed Google Scholar
Bulun SE, Yilmaz BD, Sison C, Miyazaki K, Bernardi L, Liu S, et al. Endometriosis. Endocr Rev. 2019;40(4):1048–79.
Article Google Scholar
Marquardt RM, Kim TH, Shin JH, Jeong JW. Progesterone and estrogen signaling in the endometrium: what goes wrong in endometriosis? Int J Mol Sci. 2019;20(15):3822.
Article CAS PubMed PubMed Central Google Scholar
Chapron C, Marcellin L, Borghese B, Santulli P. Rethinking mechanisms, diagnosis and management of endometriosis. Nat Rev Endocrinol. 2019;15(11):666–82.
Nnoaham KE, Hummelshoj L, Webster P, D’Hooghe T, de Cicco Nardone F, de Cicco Nardone C, et al. Impact of endometriosis on quality of life and work productivity: a multicenter study across ten countries. Fertil Steril. 2011;96(2):366–e738.
Article PubMed PubMed Central Google Scholar
Simoens S, Dunselman G, Dirksen C, Hummelshoj L, Bokor A, Brandes I, et al. The burden of endometriosis: costs and quality of life of women with endometriosis and treated in referral centres. Hum Reprod. 2012;27(5):1292–9.
Soliman AM, Yang H, Du EX, Kelley C, Winkel C. The direct and indirect costs associated with endometriosis: a systematic literature review. Hum Reprod. 2016;31(4):712–22.
Eisenberg VH, Weil C, Chodick G, Shalev V. Epidemiology of endometriosis: a large population-based database study from a healthcare provider with 2 million members. BJOG. 2018;125(1):55–62.
Article CAS PubMed Google Scholar
Dunselman GA, Vermeulen N, Becker C, Calhaz-Jorge C, D’Hooghe T, De Bie B, et al. ESHRE guideline: management of women with endometriosis. Hum Reprod. 2014;29(3):400–12.
Parasar P, Ozcan P, Terry KL. Endometriosis: epidemiology, diagnosis and clinical management. Curr Obstet Gynecol Rep. 2017;6(1):34–41.
Ford AP. In pursuit of P2X3 antagonists: novel therapeutics for chronic pain and afferent sensitization. Purinergic Signal. 2012;8(Suppl 1):3–26.
Fabbretti E. ATP P2X3 receptors and neuronal sensitization. Front Cell Neurosci. 2013;7:236.
Bonvini SJ, Belvisi MG. Cough and airway disease: the role of ion channels. Pulm Pharmacol Ther. 2017;47:21–8.
North RA. P2X receptors. Philos Trans R Soc Lond B Biol Sci. 2016;371(1700):20150427.
Cockayne DA, Dunn PM, Zhong Y, Rong W, Hamilton SG, Knight GE, et al. P2X2 knockout mice and P2X2/P2X3 double knockout mice reveal a role for the P2X2 receptor subunit in mediating multiple sensory effects of ATP. J Physiol. 2005;567(Pt 2):621–39.
Burnstock G. Purinergic signalling: therapeutic developments. Front Pharmacol. 2017;8:661.
Yuan M, Ding S, Meng T, Lu B, Shao S, Zhang X, et al. Effect of A-317491 delivered by glycolipid-like polymer micelles on endometriosis pain. Int J Nanomed. 2017;12:8171–83.
Article CAS Google Scholar
Bernier LP, Ase AR, Séguéla P. P2X receptor channels in chronic pain pathways. Br J Pharmacol. 2018;175(12):2219–30.
Ryan NM, Vertigan AE, Birring SS. An update and systematic review on drug therapies for the treatment of refractory chronic cough. Expert Opin Pharmacother. 2018;19(7):687–711.
Laux-Biehlmann A, D’Hooghe T, Zollner TM. Menstruation pulls the trigger for inflammation and pain in endometriosis. Trends Pharmacol Sci. 2015;36(5):270–6.
McKinnon B, Bersinger NA, Wotzkow C, Mueller MD. Endometriosis-associated nerve fibers, peritoneal fluid cytokine concentrations, and pain in endometriotic lesions from different locations. Fertil Steril. 2012;97(2):373–80.
McKinnon BD, Bertschi D, Bersinger NA, Mueller MD. Inflammation and nerve fiber interaction in endometriotic pain. Trends Endocrinol Metab. 2015;26(1):1–10.
Wang G, Tokushige N, Russell P, Dubinovsky S, Markham R, Fraser IS. Hyperinnervation in intestinal deep infiltrating endometriosis. J Minim Invasive Gynecol. 2009;16(6):713–9.
Barcena de Arellano ML, Mechsner S. The peritoneum—an important factor for pathogenesis and pain generation in endometriosis. J Mol Med (Berl). 2014;92(6):595–602.
CAS PubMed Google Scholar
Bodin P, Burnstock G. Increased release of ATP from endothelial cells during acute inflammation. Inflamm Res. 1998;47(8):351–4.
Bodin P, Burnstock G. Evidence that release of adenosine triphosphate from endothelial cells during increased shear stress is vesicular. J Cardiovasc Pharmacol. 2001;38(6):900–8.
Burnstock G. Purinergic receptors and pain. Curr Pharm Des. 2009;15(15):1717–35.
Lazarowski ER, Shea DA, Boucher RC, Harden TK. Release of cellular UDP-glucose as a potential extracellular signaling molecule. Mol Pharmacol. 2003;63(5):1190–7.
Seitz C, Lanius V, Lippert S, Gerlinger C, Haberland C, Oehmke F, et al. Patterns of missing data in the use of the endometriosis symptom diary. BMC Womens Health. 2018;18(1):88.
Vitonis AF, Vincent K, Rahmioglu N, Fassbender A, Buck Louis GM, Hummelshoj L, et al. World endometriosis research foundation endometriosis phenome and biobanking harmonization project: II. Clinical and covariate phenotype data collection in endometriosis research. Fertil Steril. 2014;102(5):1223–32.
Missmer SA, Spiegelman D, Hankinson SE, Malspeis S, Barbieri RL, Hunter DJ. Natural hair color and the incidence of endometriosis. Fertil Steril. 2006;85(4):866–70.
Bretz F, Pinheiro JC, Branson M. Combining multiple comparisons and modeling techniques in dose-response studies. Biometrics. 2005;61(3):738–48.
European Medicines Agency (EMA). ICH E4 Dose response information to support drug registration. 1994. https://www.ema.europa.eu/en/ich-e4-dose-response-information-support-drug-registration . Accessed October 31, 2022.
European Medicines Agency (EMA). Qualification of MCP Mod as an efficient statistical methodology for model-based design and analysis of phase II dose finding studies under model uncertainty. 2014. https://www.ema.europa.eu/en/documents/regulatory-procedural-guideline/qualification-opinion-mcp-mod-efficient-statistical-methodology-model-based-design-analysis-phase-ii_en.pdf . Accessed June 14, 2022.
Taylor HS, Giudice LC, Lessey BA, Abrao MS, Kotarski J, Archer DF, et al. Treatment of endometriosis-associated pain with elagolix, an oral GnRH antagonist. N Engl J Med. 2017;377(1):28–40.
US Food and Drug Administration. Drug-induced liver injury: premarketing clinical evaluation. 2009. https://www.fda.gov/regulatory-information/search-fda-guidancedocuments/drug-induced-liver-injury-premarketing-clinical-evaluation
Ewerton F, Cruz F, Kapp M, Klein S, Roehm P, Chapple C. Efficacy and safety of eliapixant in overactive bladder: the 12-week, randomised, placebo-controlled phase 2a OVADER study. Eur Urol Focus. 2023;S2405–4569(23):00182–7.
Google Scholar
Dicpinigaitis PV, Morice AH, Smith JA, Sher MR, Vaezi M, Guilleminault L, et al. Efficacy and safety of eliapixant in refractory chronic cough: the randomized, placebo-controlled phase 2b PAGANINI study. Lung. 2023;201(3):255–66.
Bouhassira D, Tesfaye S, Sarkar A, Soisalon-Soininen S, Stemper B, Baron R. Efficacy and safety of eliapixant in patients with diabetic neuropathic pain: the randomized, placebo-controlled, double-blind, phase 2a PUCCINI study. Pain. 2024;165(4):785–95.
Becker CM, Bokor A, Heikinheimo O, Horne A, Jansen F, Kiesel L et al. ESHRE guidelines: endometriosis. Hum Reprod Open. 2022:1–26.
Rogers PAW, Adamson GD, Al-Jefout M, Becker CM, D’Hooghe TM, Dunselman GAJ, et al. Research priorities for endometriosis. Recommendations from a global consortium of investigators in endometriosis. Reprod Sci. 2017;24(2):202–26.
Horne A, Missmer S. Pathophysiology, diagnosis, and management of endometriosis. BMJ. 2022;379:e070750.
Download references
Acknowledgements
The authors would like to thank the investigators and participants for their involvement in the study.
Bayer AG funded the SCHUMANN study.
Author information
Authors and affiliations.
Research and Development, Bayer AG, Berlin, Germany
Susanne Parke & Katrin Roth
Pharmacovigilance, Bayer AG, Berlin, Germany
Kerstin Gude
Data Science UK, Bayer PLC, UK
Fabrizio Messina
You can also search for this author in PubMed Google Scholar
Contributions
FM, KG, KR, and SP designed the study. All authors interpreted the data, wrote the manuscript, and approved the final version for publication.
Corresponding author
Correspondence to Susanne Parke .
Ethics declarations
The Institutional Review Board/Independent Ethics Committee at each center approved the protocol. The study was carried out in accordance with Good Clinical Practice guidelines, the Declaration of Helsinki, and the Council for International Organizations of Medical Sciences International Ethical Guidelines. All participants provided written informed consent.
Consent for publication
All authors have reviewed the final version of this manuscript and consented to its publication.
Competing interests
All authors are employees of Bayer AG.
Additional information
Publisher’s note.
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Supplementary Material 1
Rights and permissions.
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/ . The Creative Commons Public Domain Dedication waiver ( http://creativecommons.org/publicdomain/zero/1.0/ ) applies to the data made available in this article, unless otherwise stated in a credit line to the data.
Reprints and permissions
About this article
Cite this article.
Parke, S., Gude, K., Roth, K. et al. Efficacy and safety of eliapixant in endometriosis-associated pelvic pain: the randomized, placebo-controlled phase 2b SCHUMANN study. BMC Women's Health 24 , 353 (2024). https://doi.org/10.1186/s12905-024-03188-8
Download citation
Received : 05 December 2023
Accepted : 06 June 2024
Published : 19 June 2024
DOI : https://doi.org/10.1186/s12905-024-03188-8
Share this article
Anyone you share the following link with will be able to read this content:
Sorry, a shareable link is not currently available for this article.
Provided by the Springer Nature SharedIt content-sharing initiative
- P2X3 receptor antagonist
- Phase 2b clinical trial
BMC Women's Health
ISSN: 1472-6874
- Submission enquiries: [email protected]
- General enquiries: [email protected]

IMAGES
VIDEO
COMMENTS
Participant observation is a type of observational study. Like most observational studies, these are primarily qualitative in nature, used to conduct both explanatory research and exploratory research. Participant observation is also often used in conjunction with other types of research, like interviews and surveys.
Case study research requires researchers to purposefully select information-rich cases, as they will allow researchers an in-depth understanding of relevant and critical issues under investigation (Patton, 1990, 2002).To gain such insights, purposive sampling is generally believed to contribute to the richness in the range of data collected and help increase the possibilities of uncovering ...
Although case studies have been discussed extensively in the literature, little has been written about the specific steps one may use to conduct case study research effectively (Gagnon, 2010; Hancock & Algozzine, 2016).Baskarada (2014) also emphasized the need to have a succinct guideline that can be practically followed as it is actually tough to execute a case study well in practice.
A case study is one of the most commonly used methodologies of social research. This article attempts to look into the various dimensions of a case study research strategy, the different epistemological strands which determine the particular case study type and approach adopted in the field, discusses the factors which can enhance the effectiveness of a case study research, and the debate ...
A case study is a research method that involves an in-depth examination and analysis of a particular phenomenon or case, such as an individual, organization, community, event, or situation. It is a qualitative research approach that aims to provide a detailed and comprehensive understanding of the case being studied.
Abstract. A case study enables s ocial researchers to examine the experiences of participants within a specific context in. detail. It involves a study of a particular case from which general ...
This study on an innovative secondary school in England shows how a "low-profile" participant-observer case study was crucial to the initial observation, the testing of hypotheses, the interpretive approach, and the grounded theory. Gilgun, J. F. (1994). A Case for Case Studies in Social Work Research. Social Work, 39, 4, 371-381.
"Participant observation is the central research method of ethnography. It requires a researcher to engage with people in as many different situations as possible to look at what people actually do as well as what they say they do (as in interviews or documents) in their everyday lives" - from EAR Training Handbook
A case study is an in-depth analysis of one individual or group. Learn more about how to write a case study, including tips and examples, and its importance in psychology. ... Participant observation: When the researcher serves as a participant in events and observes the actions and outcomes, it is called participant observation.
This SAGE case study demonstrates the practical application of participant observation as a research methodology, in its use by students during an undergraduate field-trip module. Participant observation requires researchers to engage in the "systematic description" of events and interactions, as framed by a research question, to produce a ...
A case study enables social researchers to examine the experiences of participants within a specific context in detail. It involves a study of a particular case from which general principles and rules can be drawn while relying on the analysis of the social context that reflects everyday experience. This article provides practical guidance on
The case study was designed as an ongoing participant observation and documentation of the design process and of the underlaying learning process, in which the author was engaged as reflective ...
As in the case of covert and passive participant observation, researchers do not run the risk that their presence alters the behavior of the groups they study through their interactions with them. However, the guinea pig effect is a problem for this form of observation, unlike the case of covert and passive participant observation, because the ...
Case Studies and Qualitative Reports Case studies and qualitative reports may have only a few participants or even a single participant. If there is space to do so, you can write a brief background of each participant in the "Participants" section and include relevant information on the participant's birthplace, current place of residence ...
The essential topics related to the selection of participants for a health research are: 1) whether to work with samples or include the whole reference population in the study (census); 2) the sample basis; 3) the sampling process and 4) the potential effects nonrespondents might have on study results. We will refer to each of these aspects ...
The above four examples of participant observation studies are all taken from Bryman's (2016) research methods book. Bryman ranges several studies (23 in total) on a scale ranging from 'full member' through to 'partially participating observer' down to 'non-participating observer with interaction'. Students might find it ...
Single-case (SC) studies have been identified by a variety of names, including "n of 1 studies" and "single-subject" studies. The term "single-case study" is preferred over the previously mentioned terms because previous terms suggest these studies include only one participant.
Case study research is an "…intensive study of a single ... documents included participant curriculum vitas and demographic information. Field notes written by the researcher provide a source of additional data noting observations of the setting and the participant.10 Observations of the university, the participant's office ...
As demonstrated above, there are various advantages to both idiographic and nomothetic single case study analyses - notably the empirically-rich, context-specific, holistic accounts that they have to offer, and their contribution to theory-building and, to a lesser extent, that of theory-testing.
The inclusion criteria should also address the capacity of the participant to understand the study goals, the study requirements (such as completing questionnaires and taking study medications), and the potential risks associated with joining a study. ... a case to exclude pregnant women could be made for studies with known risks to maternal ...
Participant and Nonparticipant Observation: A Study of Instructional Support Liaisons. The case presents an example of a research project in which the researchers struggled to negotiate participant and nonparticipant observation roles. The project and the data collection efforts including mistakes and eventual solutions are described.
The 20 completed case study participants were recruited through one of three hospices (9/20), a hospital team such as respiratory (5/20), their GP (3/20), a CN (2/20) or a community organisation (1/20). The central participant in the case could be a patient or FCG, who could then go on to nominate other key people.
Participant observation is a qualitative research method in which the researcher observes members of the group or community being researched and participates with them in their activities. This ...
Amy C. Edmondson is the Novartis Professor of Leadership and Management at Harvard Business School, a chair established to support the study of human interactions that lead to the creation of successful enterprises that contribute to the betterment of society.She has pioneered the concept of psychological safety for over 20 years and was recognized in 2021 as #1 on the Thinkers50 global ...
The newly appointed CEO of Highstreet Properties has doubts about several members of the top team she has inherited. She's trying to drive a turnaround, the company has a complicated matrix ...
The study initially involved 600 Black men - 399 with syphilis, 201 who did not have the disease. Participants' informed consent was not collected. ... In 1995, the program was expanded to include health, as well as medical, benefits. The last study participant died in January 2004. The last widow receiving THBP benefits died in January ...
Objective: There is a lack of evidence guiding treatment guidelines for individuals with comorbid complex posttraumatic stress disorder (C-PTSD) and binge eating disorder (BED). The article aims to present preliminary evidence to support the proof of concept of novel therapy termed meta-emotion therapy (MET) that targeted maladaptive beliefs about emotions, for this specific comorbidity ...
Section 10-4o - [Effective 10/1/2024] (Formerly Sec. 17-605). Family resource center program. Guidelines for programs. Study. Grants (a) The Department of Education, in conjunction with the Department of Social Services, shall coordinate a family resource center program to provide comprehensive child care services, remedial educational and literacy services, families-in-training programs and ...
The first case occurred in a 26-year-old female participant after 4 weeks of exposure to eliapixant 150 mg BID in the phase 2b study of patients suffering from RUCC . Although increases in liver function parameters with eliapixant treatment had been defined as a potential risk before the start of phase 2 studies, the benefit-risk ratio for the ...
Four blood samples were collected from each participant aseptically. The first blood sample (2 mL) was collected into a gel vacutainer tube and was centrifuged at 4000 r/min for 20 min after complete blood clotting. ... This case-control study included 100 adult HCV-positive patients with HCC and 100 matching adult HCV-positive patients with ...